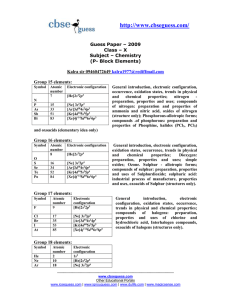
Slide 1
... • Raising the temperature of this system requires the addition of heat, which shifts the equilibrium to the left and reduces the concentration of hydrogen chloride. • Thus, the value of Keq decreases. • Lowering the temperature of the system means that heat is removed, so the equilibrium relieves th ...
... • Raising the temperature of this system requires the addition of heat, which shifts the equilibrium to the left and reduces the concentration of hydrogen chloride. • Thus, the value of Keq decreases. • Lowering the temperature of the system means that heat is removed, so the equilibrium relieves th ...
content - Thesis Scientist
... P or As, the dopant atom forms four covalent bonds like a Si or Ge atom but the fifth electron, not used in bonding, becomes delocalised and contribute its share towards electrical conduction. Thus silicon or germanium doped with P or As is called n-type semiconductor, n indicative of negative, sinc ...
... P or As, the dopant atom forms four covalent bonds like a Si or Ge atom but the fifth electron, not used in bonding, becomes delocalised and contribute its share towards electrical conduction. Thus silicon or germanium doped with P or As is called n-type semiconductor, n indicative of negative, sinc ...
No Slide Title
... Calculate standard enthalpy changes using bond enthalpy values Calculate standard enthalpy changes using enthalpies of formation and combustion Know simple calorimetry methods for measuring enthalpy changes Calculate enthalpy changes from calorimetry measurements ...
... Calculate standard enthalpy changes using bond enthalpy values Calculate standard enthalpy changes using enthalpies of formation and combustion Know simple calorimetry methods for measuring enthalpy changes Calculate enthalpy changes from calorimetry measurements ...
Chemistry - SSA Punjab
... of 1.83g mL-1 (i) Calculate the molarity of the solution (ii) volume of concentrated acid required to prepare 3.5L of 0.50 M H2SO4 ...
... of 1.83g mL-1 (i) Calculate the molarity of the solution (ii) volume of concentrated acid required to prepare 3.5L of 0.50 M H2SO4 ...
enthalpy changes
... Calculate standard enthalpy changes using bond enthalpy values Calculate standard enthalpy changes using enthalpies of formation and combustion Know simple calorimetry methods for measuring enthalpy changes Calculate enthalpy changes from calorimetry measurements ...
... Calculate standard enthalpy changes using bond enthalpy values Calculate standard enthalpy changes using enthalpies of formation and combustion Know simple calorimetry methods for measuring enthalpy changes Calculate enthalpy changes from calorimetry measurements ...
DELTAHPP
... Calculate standard enthalpy changes using bond enthalpy values Calculate standard enthalpy changes using enthalpies of formation and combustion Know simple calorimetry methods for measuring enthalpy changes Calculate enthalpy changes from calorimetry measurements ...
... Calculate standard enthalpy changes using bond enthalpy values Calculate standard enthalpy changes using enthalpies of formation and combustion Know simple calorimetry methods for measuring enthalpy changes Calculate enthalpy changes from calorimetry measurements ...
Unit 3: 1 Equilibrium and the Constant, K
... principle, to infer the relative rates of the forward and reverse reactions. [See SP 7.2; Essential knowledge 6.A.3] Learning objective 6.4 The student can, given a set of initial conditions (concentrations or partial pressures) and the equilibrium constant, K, use the tendency of Q to approach K to ...
... principle, to infer the relative rates of the forward and reverse reactions. [See SP 7.2; Essential knowledge 6.A.3] Learning objective 6.4 The student can, given a set of initial conditions (concentrations or partial pressures) and the equilibrium constant, K, use the tendency of Q to approach K to ...
Kinetic Modeling Of Methanol Synthesis From Carbon Monoxide
... (∆H = -41.2 kJ/mol; ∆G = -28.60 kJ/mol) ...
... (∆H = -41.2 kJ/mol; ∆G = -28.60 kJ/mol) ...
deltahpps
... Calculate standard enthalpy changes using bond enthalpy values Calculate standard enthalpy changes using enthalpies of formation and combustion Know simple calorimetry methods for measuring enthalpy changes Calculate enthalpy changes from calorimetry measurements ...
... Calculate standard enthalpy changes using bond enthalpy values Calculate standard enthalpy changes using enthalpies of formation and combustion Know simple calorimetry methods for measuring enthalpy changes Calculate enthalpy changes from calorimetry measurements ...
CHM 423 Coordination Chemistry
... from the nature of chemical bond that leads to the formation of these compounds. This chemical bond called coordinate covalent bond involves donation of electron pair(s) by a molecule or negatively charged ion, a Lewis base, to a neutral metal or positively charged ion, a Lewis acid. These compounds ...
... from the nature of chemical bond that leads to the formation of these compounds. This chemical bond called coordinate covalent bond involves donation of electron pair(s) by a molecule or negatively charged ion, a Lewis base, to a neutral metal or positively charged ion, a Lewis acid. These compounds ...
enjoy chemistry
... (xxiv)Pentahalides of group 15 are more Covalent than trihalides Ans:Higher the positive oxidation state of central atom, more will be its polarizing power which, in turn, increases the covalent character of bond formed between the central atom and the other atom. (xxv)N2 is less reactive at room te ...
... (xxiv)Pentahalides of group 15 are more Covalent than trihalides Ans:Higher the positive oxidation state of central atom, more will be its polarizing power which, in turn, increases the covalent character of bond formed between the central atom and the other atom. (xxv)N2 is less reactive at room te ...
Brønsted Acidity in Metal−Organic Frameworks
... containing several different kinds of metal ions within one SBU83 is highly desirable for developing stronger Brønsted acids based on bridging hydroxyl groups. Water molecules bound to metal sites could also result in Brønsted acidity, as exemplified by MIL-100.84,85 Mediumstrength Brønsted acidic sit ...
... containing several different kinds of metal ions within one SBU83 is highly desirable for developing stronger Brønsted acids based on bridging hydroxyl groups. Water molecules bound to metal sites could also result in Brønsted acidity, as exemplified by MIL-100.84,85 Mediumstrength Brønsted acidic sit ...
Review of N and Metal co-Doped TiO for Water Purification under
... nitrogen [7], sulfur [8], fluorine [9], iodine [10] and boron [11] into the TiO2 bulk to successfully cause the red shift of the absorption onset of TiO2 to the visible region and enhance the photocatalytic activity. Among these elements, nitrogen was thought as the most effective substitution doping ...
... nitrogen [7], sulfur [8], fluorine [9], iodine [10] and boron [11] into the TiO2 bulk to successfully cause the red shift of the absorption onset of TiO2 to the visible region and enhance the photocatalytic activity. Among these elements, nitrogen was thought as the most effective substitution doping ...
AP Chemistry - Siva Kodali
... at Fusion Learning Center and Fusion Academy. There, he enjoys convincing students that biology and chemistry are, in fact, fascinating journeys, not entirely designed to inflict pain on hapless teenagers. His military training occasionally aids him in this effort. He is the author of AP Biology For ...
... at Fusion Learning Center and Fusion Academy. There, he enjoys convincing students that biology and chemistry are, in fact, fascinating journeys, not entirely designed to inflict pain on hapless teenagers. His military training occasionally aids him in this effort. He is the author of AP Biology For ...
MULTIPLY CHOICE QUESTIONS ON MEDICAL CHEMISTRY
... 1.49. The daily ration of fats for an adult is: А. 70–80 g B. 110–120 g C. 30–50 g D. 90-110 g E. 60–70 g 1.50. The daily ration of carbohydrates for an adult is: А. 380–390 g B. 250–280 g C. 60–70 g D. 90–100 g E. 300–370 g 1.51. Energy which is accumulated in the organism may be released under the ...
... 1.49. The daily ration of fats for an adult is: А. 70–80 g B. 110–120 g C. 30–50 g D. 90-110 g E. 60–70 g 1.50. The daily ration of carbohydrates for an adult is: А. 380–390 g B. 250–280 g C. 60–70 g D. 90–100 g E. 300–370 g 1.51. Energy which is accumulated in the organism may be released under the ...
Chemistry 110 Oxidation Reduction Reactions Oxidation Number
... We can see that the oxidation number of C increases from -4 to +4 in this reaction, so C is oxidized. We can also see that the oxidation number of O decreases from zero (0) to -2, so O is reduced. Notice that the oxidation number of hydrogen does not change. It is always the case that if any element ...
... We can see that the oxidation number of C increases from -4 to +4 in this reaction, so C is oxidized. We can also see that the oxidation number of O decreases from zero (0) to -2, so O is reduced. Notice that the oxidation number of hydrogen does not change. It is always the case that if any element ...
B.Sc. (Hons.) Chemistry
... of Kapustinskii expression for lattice energy. Madelung constant, Born-Haber cycle and its application, Solvation energy. (ii) Covalent bond: Lewis structure, Valence Bond theory (Heitler-London approach). Energetics of hybridization, equivalent and non-equivalent hybrid orbitals. Bent’s rule, Reson ...
... of Kapustinskii expression for lattice energy. Madelung constant, Born-Haber cycle and its application, Solvation energy. (ii) Covalent bond: Lewis structure, Valence Bond theory (Heitler-London approach). Energetics of hybridization, equivalent and non-equivalent hybrid orbitals. Bent’s rule, Reson ...
Document
... vacancy defect. This results the decrease in density of the substance. This defect develops when a substance is heated. # Interstitial defect- When some constituent particles occupy an interstitial site, the crystal is said to have interstitial defect. This defect increases the density of the substa ...
... vacancy defect. This results the decrease in density of the substance. This defect develops when a substance is heated. # Interstitial defect- When some constituent particles occupy an interstitial site, the crystal is said to have interstitial defect. This defect increases the density of the substa ...
x - SharpSchool
... two different acids (or bases) can have the same [ ] but have different strengths eg) 1 M CH3COOH(aq) and 1 M HCl(aq) will react in the same way but not to the same degree the stronger the acid, the more electricity it conducts, ...
... two different acids (or bases) can have the same [ ] but have different strengths eg) 1 M CH3COOH(aq) and 1 M HCl(aq) will react in the same way but not to the same degree the stronger the acid, the more electricity it conducts, ...
The Chemistry and Applications of Metal
... octahedral SBUs each linked by six chelating 1,4-benzenedicarboxylate (BDC2–) units to give a cubic framework (Fig. 2, figs. S2 and S3, and tables S1 and S2). The architectural robustness of MOF-5 allowed for gas sorption measurements, which revealed 61% porosity and a BrunauerEmmett-Teller (BET) su ...
... octahedral SBUs each linked by six chelating 1,4-benzenedicarboxylate (BDC2–) units to give a cubic framework (Fig. 2, figs. S2 and S3, and tables S1 and S2). The architectural robustness of MOF-5 allowed for gas sorption measurements, which revealed 61% porosity and a BrunauerEmmett-Teller (BET) su ...
Synthetic Organic Chemistry - Name
... 1.3 Applications of organolithium compounds Organolithium compounds are the most versatile reagents in all fields of chemistry . Thus some of the important applications of these compounds are as follows: 1. Organolithium compounds are highly reactive nucleophiles and strong bases due to the presenc ...
... 1.3 Applications of organolithium compounds Organolithium compounds are the most versatile reagents in all fields of chemistry . Thus some of the important applications of these compounds are as follows: 1. Organolithium compounds are highly reactive nucleophiles and strong bases due to the presenc ...
Ozone decomposition
... above 105 °C. The gaseous ozone is characterized by different times of half-life, depending on the temperature (Table 1). The ozone structure is resonance stabilized, which is one of the reasons for its resistance against decomposition at low temperatures (Figure 2). In most reactions with inorganic ...
... above 105 °C. The gaseous ozone is characterized by different times of half-life, depending on the temperature (Table 1). The ozone structure is resonance stabilized, which is one of the reasons for its resistance against decomposition at low temperatures (Figure 2). In most reactions with inorganic ...
chemical equilibrium type 1
... technique that is used to determine whether a reaction it truly at equilibrium is to approach equilibrium starting with reactants in one experiment and starting with products in another. If the same value of the reaction quotient is observed when the concentrations stop changing in both experiments, ...
... technique that is used to determine whether a reaction it truly at equilibrium is to approach equilibrium starting with reactants in one experiment and starting with products in another. If the same value of the reaction quotient is observed when the concentrations stop changing in both experiments, ...
RUMPLE-DISSERTATION-2014 - SMARTech Home
... Eckert. The opportunity to work with such skilled scientists and kind mentors is a rare one, and I am extremely glad I had the opportunity to learn from them. I have always been in awe of their brilliance (I’m pretty sure they have each forgotten more than I’ll ever learn, and they still know so muc ...
... Eckert. The opportunity to work with such skilled scientists and kind mentors is a rare one, and I am extremely glad I had the opportunity to learn from them. I have always been in awe of their brilliance (I’m pretty sure they have each forgotten more than I’ll ever learn, and they still know so muc ...
Types of Chemical Reactions
... – One of the chemical properties of acids and bases is that they neutralize one another. – A neutralization reaction is a reaction of an acid and a base that results in an ionic compound (salt) and water. A + B salt + H2O – The ionic compound that is the product of a neutralization reaction is cal ...
... – One of the chemical properties of acids and bases is that they neutralize one another. – A neutralization reaction is a reaction of an acid and a base that results in an ionic compound (salt) and water. A + B salt + H2O – The ionic compound that is the product of a neutralization reaction is cal ...