Chemical Redox Agents for Organometallic
... solutions, the usual media for organometallic ET reactions. It is hoped that the information in this review will also be useful to those interested in ET reactions of organic and inorganic systems, which have more often involved the use of labile metal ions such as M(III), M ) Fe, Tl1 (but see ref 2 ...
... solutions, the usual media for organometallic ET reactions. It is hoped that the information in this review will also be useful to those interested in ET reactions of organic and inorganic systems, which have more often involved the use of labile metal ions such as M(III), M ) Fe, Tl1 (but see ref 2 ...
Chemical Redox Agents for Organometallic
... solutions, the usual media for organometallic ET reactions. It is hoped that the information in this review will also be useful to those interested in ET reactions of organic and inorganic systems, which have more often involved the use of labile metal ions such as M(III), M ) Fe, Tl1 (but see ref 2 ...
... solutions, the usual media for organometallic ET reactions. It is hoped that the information in this review will also be useful to those interested in ET reactions of organic and inorganic systems, which have more often involved the use of labile metal ions such as M(III), M ) Fe, Tl1 (but see ref 2 ...
Experimental details
... pressure as well as the very intense ionizing radiation emitted. The relevant materials in nuclear technological applications are required to be stable and durable after having withstood these extreme conditions. The materials stability under high temperature and pressure is primarily controlled by ...
... pressure as well as the very intense ionizing radiation emitted. The relevant materials in nuclear technological applications are required to be stable and durable after having withstood these extreme conditions. The materials stability under high temperature and pressure is primarily controlled by ...
BSc in Chemistry-CUCBCSS UG 2014-Scheme
... and developments of the modern society from time to time. To achieve this goal, the curriculum should be restructured by giving emphasis on various aspects such as the creativity of students, knowledge of current developments in the discipline, awareness of environmental impacts due to the developme ...
... and developments of the modern society from time to time. To achieve this goal, the curriculum should be restructured by giving emphasis on various aspects such as the creativity of students, knowledge of current developments in the discipline, awareness of environmental impacts due to the developme ...
design synthesis and functionalization of self assembled
... Supramolecular chemistry is a broad field, owing to the vast number of diverse structures that can be formed by using a variety of noncovalent intermolecular interactions. Notable examples include biologically relevant enzyme mimics11, molecular devices including light harvesters12, sensors13, wires1 ...
... Supramolecular chemistry is a broad field, owing to the vast number of diverse structures that can be formed by using a variety of noncovalent intermolecular interactions. Notable examples include biologically relevant enzyme mimics11, molecular devices including light harvesters12, sensors13, wires1 ...
Chemistry Final Exam Review
... ____ 24. A reversible chemical reaction means that the reaction can travel forwards or backwards. ____ 25. Subscripts are used to balance chemical reactions. ____ 26. A synthesis reaction contains two products. ____ 27. A decomposition reaction contains at least two products. ____ 28. A combustion r ...
... ____ 24. A reversible chemical reaction means that the reaction can travel forwards or backwards. ____ 25. Subscripts are used to balance chemical reactions. ____ 26. A synthesis reaction contains two products. ____ 27. A decomposition reaction contains at least two products. ____ 28. A combustion r ...
Chemistry.of Organic Compounds
... in this text considerable emphasis is placed on the explanation of physical properties and on the mechanism of organic reactions. Where such material is given, an effort has been made to keep the discussion as simple as possible even at the risk of being quantitatively inaccurate, for "except ye utt ...
... in this text considerable emphasis is placed on the explanation of physical properties and on the mechanism of organic reactions. Where such material is given, an effort has been made to keep the discussion as simple as possible even at the risk of being quantitatively inaccurate, for "except ye utt ...
master ap chemistry - NelnetSolutions.com
... ALL RIGHTS RESERVED. No part of this work covered by the copyright herein may be reproduced or used in any form or by any means—graphic, electronic, or mechanical, including photocopying, recording, taping, Web distribution, or information storage and retrieval systems—without the prior written perm ...
... ALL RIGHTS RESERVED. No part of this work covered by the copyright herein may be reproduced or used in any form or by any means—graphic, electronic, or mechanical, including photocopying, recording, taping, Web distribution, or information storage and retrieval systems—without the prior written perm ...
Solving Problems: A Chemistry Handbook
... A scientific method is a systematic approach used to answer a question or study a situation. It is both an organized way for scientists to do research and a way for scientists to verify the work of other scientists. A typical scientific method includes making observations, forming a hypothesis, perf ...
... A scientific method is a systematic approach used to answer a question or study a situation. It is both an organized way for scientists to do research and a way for scientists to verify the work of other scientists. A typical scientific method includes making observations, forming a hypothesis, perf ...
Copyright 2010 Scott R
... Na(H3BNMe2BH3), in tetrahydrofuran produces the new complex Th(H3BNMe2BH3)4. The thorium center forms bonds with fifteen hydrogen atoms; accordingly, this is the first example of a fifteen-coordinate atom of any kind. As determined by both single crystal X-ray and single crystal neutron diffraction ...
... Na(H3BNMe2BH3), in tetrahydrofuran produces the new complex Th(H3BNMe2BH3)4. The thorium center forms bonds with fifteen hydrogen atoms; accordingly, this is the first example of a fifteen-coordinate atom of any kind. As determined by both single crystal X-ray and single crystal neutron diffraction ...
questions based on high order thinking skill
... of end face centred (monoclinic) lattice with a molecule at each lattice. Ans. 2. Q. 3. What is the coordination number of carbon, in diamond ? Ans. 4 and its unit cell has 8 atoms. [The space lattice of diamond is FCC] Q. 4. Name the solid which has weakest intermolecular force ? Ans. Ice Q. 5. Arr ...
... of end face centred (monoclinic) lattice with a molecule at each lattice. Ans. 2. Q. 3. What is the coordination number of carbon, in diamond ? Ans. 4 and its unit cell has 8 atoms. [The space lattice of diamond is FCC] Q. 4. Name the solid which has weakest intermolecular force ? Ans. Ice Q. 5. Arr ...
questions based on high order thinking skill - Entrance
... of end face centred (monoclinic) lattice with a molecule at each lattice. Ans. 2. Q. 3. What is the coordination number of carbon, in diamond ? Ans. 4 and its unit cell has 8 atoms. [The space lattice of diamond is FCC] Q. 4. Name the solid which has weakest intermolecular force ? Ans. Ice Q. 5. Arr ...
... of end face centred (monoclinic) lattice with a molecule at each lattice. Ans. 2. Q. 3. What is the coordination number of carbon, in diamond ? Ans. 4 and its unit cell has 8 atoms. [The space lattice of diamond is FCC] Q. 4. Name the solid which has weakest intermolecular force ? Ans. Ice Q. 5. Arr ...
Metallocene Organoactinide Complexes
... match with ligand-based orbitals, spatial overlap is poor, precluding strong metal–ligand bonding [44]. Thorium lies early in the actinide series and the relatively high energy of the 5f orbitals (before the increasing effective nuclear charge across the series drops the energy of these orbitals) ha ...
... match with ligand-based orbitals, spatial overlap is poor, precluding strong metal–ligand bonding [44]. Thorium lies early in the actinide series and the relatively high energy of the 5f orbitals (before the increasing effective nuclear charge across the series drops the energy of these orbitals) ha ...
Late Transition Metal Amido Complexes: Electronic
... theory,[1] complexes of early, electron poor transition metals in high oxidation states are well stabilized by covalently bound -donating ligands, such as alkoxy, oxo, amido, imido or nitrido ligands which adapt for the electronic demand of the high-valent, Lewis acidic metal center.[2] In the 1960 ...
... theory,[1] complexes of early, electron poor transition metals in high oxidation states are well stabilized by covalently bound -donating ligands, such as alkoxy, oxo, amido, imido or nitrido ligands which adapt for the electronic demand of the high-valent, Lewis acidic metal center.[2] In the 1960 ...
Energetics
... Standard enthalpy changes CH4(g) + 2O2(g) CO2(g) + 2H2O(g) H = -802 kJ mol-1 at 373 K CH4(g) + 2O2(g) CO2(g) + 2H2O(l) H = -890 kJ mol-1 at 298 K ...
... Standard enthalpy changes CH4(g) + 2O2(g) CO2(g) + 2H2O(g) H = -802 kJ mol-1 at 373 K CH4(g) + 2O2(g) CO2(g) + 2H2O(l) H = -890 kJ mol-1 at 298 K ...
Mole-mole factor
... – If special conditions such as heat are required – The number of molecules, formula units, or atoms of each type of molecule involved in the reaction • Number can be in terms of single atoms, or moles of atoms – The relative number of moles of each reactant and product ...
... – If special conditions such as heat are required – The number of molecules, formula units, or atoms of each type of molecule involved in the reaction • Number can be in terms of single atoms, or moles of atoms – The relative number of moles of each reactant and product ...
Soln Chem 2008Nov(9746)
... Ka, the acid dissociation constant, is only affected by changes in temperature. It is not affected by changes in volume, V. (ans) © Step-by-Step ...
... Ka, the acid dissociation constant, is only affected by changes in temperature. It is not affected by changes in volume, V. (ans) © Step-by-Step ...
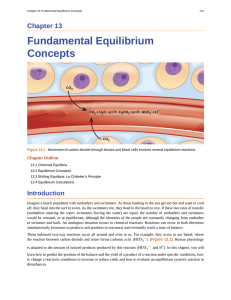
Fundamental Equilibrium Concepts
... Figure 13.2 A mixture of NO2 and N2O4 moves toward equilibrium. Colorless N2O4 reacts to form brown NO2. As the reaction proceeds toward equilibrium, the color of the mixture darkens due to the increasing concentration of NO2. ...
... Figure 13.2 A mixture of NO2 and N2O4 moves toward equilibrium. Colorless N2O4 reacts to form brown NO2. As the reaction proceeds toward equilibrium, the color of the mixture darkens due to the increasing concentration of NO2. ...
File
... Let's say the volume of the container is decreased. This increases the total pressure of the system. Increasing the pressure will increase the concentrations of all three species the same amount. Since there are more moles of gas (N2(g) 2(g)) on the left side, there is more "stuff" increased in the ...
... Let's say the volume of the container is decreased. This increases the total pressure of the system. Increasing the pressure will increase the concentrations of all three species the same amount. Since there are more moles of gas (N2(g) 2(g)) on the left side, there is more "stuff" increased in the ...
EVS - RSC - Developments in Microwave Chemistry
... include the synthesis of fine chemicals, organometallic, coordination, intercalation compounds, and nanoparticles. Microwave technology also enables chemists to achieve cleaner and more efficient chemical reactions with higher yields, compared to conventional heating methods. The microwave chemistry ...
... include the synthesis of fine chemicals, organometallic, coordination, intercalation compounds, and nanoparticles. Microwave technology also enables chemists to achieve cleaner and more efficient chemical reactions with higher yields, compared to conventional heating methods. The microwave chemistry ...
МЕТОДИЧЕСКИЕ УКАЗАНИЯ СТУДЕНТАМ
... 1. What volume (in mL) of 40% H3PO4 solution (ρ=1,25 g/cm3) is necessary to prepare 400mL of 0,25N of phosphoric acid solution (ρ=1g/cm3)? Calculate the mole fraction of H3PO4 in the obtained solution. 2. How many grams of Na2CO3·10H2O are necessary to prepare 100mL of 10% Na2CO3 solution (ρ=1,12 g/ ...
... 1. What volume (in mL) of 40% H3PO4 solution (ρ=1,25 g/cm3) is necessary to prepare 400mL of 0,25N of phosphoric acid solution (ρ=1g/cm3)? Calculate the mole fraction of H3PO4 in the obtained solution. 2. How many grams of Na2CO3·10H2O are necessary to prepare 100mL of 10% Na2CO3 solution (ρ=1,12 g/ ...
Chapter 15: Chemical Equilibrium
... The equilibrium concentrations of ammonia and hydrogen chloride do not depend on the amount of solid present as long as there is some solid present for the system to reach equilibrium. The concentration of a pure solid depends only on the density of the substance, a constant that can be incorporate ...
... The equilibrium concentrations of ammonia and hydrogen chloride do not depend on the amount of solid present as long as there is some solid present for the system to reach equilibrium. The concentration of a pure solid depends only on the density of the substance, a constant that can be incorporate ...
REVIEWS Environmental remediation by photocatalysis R. Vinu AND Giridhar Madras
... that, the oxidizing power of the VB hole or the hydroxyl radicals is always higher than that of the reducing power of the CB electrons. Moreover, these interfacial electron transfer steps compete with the electron-hole recombination reaction (10 ns) and hence, the practical efficiency or quantum yield ...
... that, the oxidizing power of the VB hole or the hydroxyl radicals is always higher than that of the reducing power of the CB electrons. Moreover, these interfacial electron transfer steps compete with the electron-hole recombination reaction (10 ns) and hence, the practical efficiency or quantum yield ...
Complete Solution Manual
... The zero point for standard reduction potentials (E) is the standard hydrogen electrode. The half-reaction is: 2 H+ + 2 e → H2. This half-reaction is assigned a standard potential of zero, and all other reduction half-reactions are measured relative to this zero point. Substances less easily reduc ...
... The zero point for standard reduction potentials (E) is the standard hydrogen electrode. The half-reaction is: 2 H+ + 2 e → H2. This half-reaction is assigned a standard potential of zero, and all other reduction half-reactions are measured relative to this zero point. Substances less easily reduc ...
Complete Solution Manual
... The zero point for standard reduction potentials (E) is the standard hydrogen electrode. The half-reaction is: 2 H+ + 2 e → H2. This half-reaction is assigned a standard potential of zero, and all other reduction half-reactions are measured relative to this zero point. Substances less easily reduc ...
... The zero point for standard reduction potentials (E) is the standard hydrogen electrode. The half-reaction is: 2 H+ + 2 e → H2. This half-reaction is assigned a standard potential of zero, and all other reduction half-reactions are measured relative to this zero point. Substances less easily reduc ...