Chemistry 3719L – Week 9 Reduction of Benzil with Sodium
... The use of metal hydride reagents (e.g. NaBH4 and LiAlH4) for the reduction of aldehydes and ketones is the most widely used method for conversion to primary and secondary alcohols respectively. This experiment will give you experience in using the milder NaBH4 reagent that readily converts the dike ...
... The use of metal hydride reagents (e.g. NaBH4 and LiAlH4) for the reduction of aldehydes and ketones is the most widely used method for conversion to primary and secondary alcohols respectively. This experiment will give you experience in using the milder NaBH4 reagent that readily converts the dike ...
The carbonyl functional group Formation of the C=O group π
... either side of the carbonyl group. • A symmetric ketone like propanone, CH3COCH3, will only produce a single product - not a mixture of isomers. The product doesn't have four different groups around the central carbon atom, and so won't have optical isomers ...
... either side of the carbonyl group. • A symmetric ketone like propanone, CH3COCH3, will only produce a single product - not a mixture of isomers. The product doesn't have four different groups around the central carbon atom, and so won't have optical isomers ...
Carboxylates/esters vs ketones/aldehydes
... BH3 becomes B(OC2H5)3 by reacting with ethanol, then, when heated with water, becomes B(OH)3. The mechanism of the NaBH4 reduction in a protic solvent such as ethanol, methanol, and water is known to be quite complex since NaBH4 reacts with the solvent, e.g., NaBH4 + C2H5OH → NaBH3(OC2H5) + H2 Becau ...
... BH3 becomes B(OC2H5)3 by reacting with ethanol, then, when heated with water, becomes B(OH)3. The mechanism of the NaBH4 reduction in a protic solvent such as ethanol, methanol, and water is known to be quite complex since NaBH4 reacts with the solvent, e.g., NaBH4 + C2H5OH → NaBH3(OC2H5) + H2 Becau ...
18 Important and sometimes forgotten) organic transformations
... •Tertiary alcohols are prone to elimination (Chugaev reaction) •Thionoformates may be used to derivatise and deoxygenate tertiary alcohols without competing elimination ...
... •Tertiary alcohols are prone to elimination (Chugaev reaction) •Thionoformates may be used to derivatise and deoxygenate tertiary alcohols without competing elimination ...
$doc.title
... http://www.chem.wisc.edu/areas/clc (Resource page) Reactions of Alcohols #8: Reaction of a 1° Alcohol with Hydrogen Halides ...
... http://www.chem.wisc.edu/areas/clc (Resource page) Reactions of Alcohols #8: Reaction of a 1° Alcohol with Hydrogen Halides ...
Molybdenum based catalysts as Oxidants and Reductants
... molar ratio in THF for about 18 hours as depicted in scheme 1. The completion of the reaction may be ascertained by measuring the requisite amount of hydrogen gas released. Generally these compounds exist in solution phase and can be recovered in solid form by the partial evaporation of THF and addi ...
... molar ratio in THF for about 18 hours as depicted in scheme 1. The completion of the reaction may be ascertained by measuring the requisite amount of hydrogen gas released. Generally these compounds exist in solution phase and can be recovered in solid form by the partial evaporation of THF and addi ...
Chap Thirteen: Alcohols
... ii. Reduction of Esters and Carboxylic Acids with LiAlH4 only LEARNING OUTCOMES: Understand the formation of alcohols through use of organometallics in carboncarbon bond forming reactions with electrophiles Predict the stereochemistry and optical activity of a product from an understanding of it ...
... ii. Reduction of Esters and Carboxylic Acids with LiAlH4 only LEARNING OUTCOMES: Understand the formation of alcohols through use of organometallics in carboncarbon bond forming reactions with electrophiles Predict the stereochemistry and optical activity of a product from an understanding of it ...
Slide 1
... • Nucleophiles that form unstable addition products form conjugated addition products, because the conjugate addition is not reversible ...
... • Nucleophiles that form unstable addition products form conjugated addition products, because the conjugate addition is not reversible ...
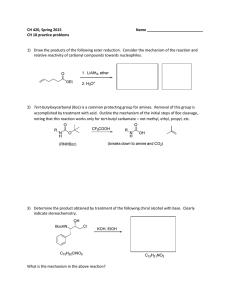
CH 420, Spring 2015 Name ___________________________ CH 18 practice problems
... 7) Rank the following compounds according to their relative acidity: cyclohexanol, phenol, pmethoxyphenol, p-nitrophenol. ...
... 7) Rank the following compounds according to their relative acidity: cyclohexanol, phenol, pmethoxyphenol, p-nitrophenol. ...
reactions of the carbonyl group in aldehydes and ketones
... atoms attracted to an electron-deficient centre, where it donates a pair of electrons to form a new covalent bond A curly arrow is a symbol used in reaction mechanisms to show the movement of an electron pair in the braking or forming of a covalent bond ...
... atoms attracted to an electron-deficient centre, where it donates a pair of electrons to form a new covalent bond A curly arrow is a symbol used in reaction mechanisms to show the movement of an electron pair in the braking or forming of a covalent bond ...
Wolff–Kishner reduction

The Wolff–Kishner reduction is a reaction used in organic chemistry to convert carbonyl functionalities into methylene groups. In the context of complex molecule synthesis, it is most frequently employed to remove a carbonyl group after it has served its synthetic purpose of activating an intermediate in a preceding step. As such, there is no obvious retron for this reaction. Originally reported by Nikolai Kischner in 1911 and Ludwig Wolff in 1912, it has been applied to the total synthesis of scopadulcic acid B, aspidospermidine and dysidiolide.In general, the reaction mechanism first involves the in situ generation of a hydrazone by condensation of hydrazine with the ketone or aldehyde substrate. Sometimes it is however advantageous to use a pre-formed hydrazone as substrate (see modifications). The hydrazone is deprotonated by alkoxide base followed by a concerted, rate-determining step in which a diimide anion is formed. Collapse of this alkyldiimde with loss of N2 leads to formation of an alkylanion which can be protonated by solvent to give the desired product.Because the Wolff–Kishner reduction requires highly basic conditions, it is unsuitable for base-sensitive substrates. However, this method can be superior over the related Clemmensen reduction for acid-sensitive compounds such as pyrroles and for high-molecular weight compounds.