Midterm Examination
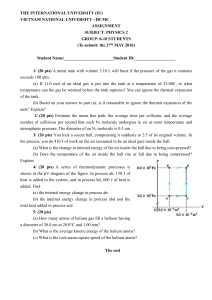
... temperature can the gas be warmed before the tank ruptures? You can ignore the thermal expansion of the tank. (b) Based on your answer to part (a), is it reasonable to ignore the thermal expansion of the tank? Explain? 2/ (20 pts) Estimate the mean free path, the average time per collision, and the ...
... temperature can the gas be warmed before the tank ruptures? You can ignore the thermal expansion of the tank. (b) Based on your answer to part (a), is it reasonable to ignore the thermal expansion of the tank? Explain? 2/ (20 pts) Estimate the mean free path, the average time per collision, and the ...
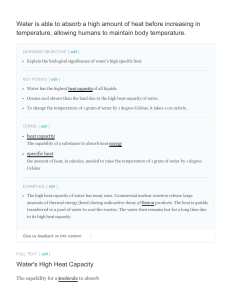
HONORS CHEMISTRY STUDY GUIDE Thermochemistry
... as well as the names of the heats associated with the phase changes sections. -Be able to do problems determining the heat where the temperature and phase of a substance changes. -Understand that in such problems each section of the heat curve needs its own calculation. The total heat can then be de ...
... as well as the names of the heats associated with the phase changes sections. -Be able to do problems determining the heat where the temperature and phase of a substance changes. -Understand that in such problems each section of the heat curve needs its own calculation. The total heat can then be de ...
A-Basic on Thermal Management
... electromagnetic waves that intervene from the hot surfaces to the cold one as electromagnetic waves. It does not require medium a conductor for its propagation. For these three modes, the heat transfer rates depend on the transport properties of the substances involved, the geometrical parameters, a ...
... electromagnetic waves that intervene from the hot surfaces to the cold one as electromagnetic waves. It does not require medium a conductor for its propagation. For these three modes, the heat transfer rates depend on the transport properties of the substances involved, the geometrical parameters, a ...
Energy - Winona State University
... particles, therefore related to kinetic energy of the system. Heat involves a transfer of energy between 2 objects due to a temperature difference ...
... particles, therefore related to kinetic energy of the system. Heat involves a transfer of energy between 2 objects due to a temperature difference ...
Themodynamic notes section 6.1
... Laws of Thermodynamics • The energy in the universe is constant • The entropy of a system not in thermal equilibrium will increase. • The entropy of a system approaches a constant value as the system approaches absolute zero. ...
... Laws of Thermodynamics • The energy in the universe is constant • The entropy of a system not in thermal equilibrium will increase. • The entropy of a system approaches a constant value as the system approaches absolute zero. ...
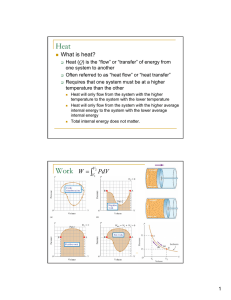
Heat Work
... may happen through heat transfer or through mechanical work First law is a statement of conservation of energy Change in internal energy of system equals the difference between the heat added to the system and the work done by the ...
... may happen through heat transfer or through mechanical work First law is a statement of conservation of energy Change in internal energy of system equals the difference between the heat added to the system and the work done by the ...
More Carnot Cycle March 4, 2010 Efficiency = W/Qin = Qin
... length (L) of the rod. Introducing a constant of proportionality K, called the thermal conductivity of the material. ...
... length (L) of the rod. Introducing a constant of proportionality K, called the thermal conductivity of the material. ...
Heat transfer

Heat transfer is the exchange of thermal energy between physical systems, depending on the temperature and pressure, by dissipating heat. The fundamental modes of heat transfer are conduction or diffusion, convection and radiation.Heat transfer always occurs from a region of high temperature to another region of lower temperature. Heat transfer changes the internal energy of both systems involved according to the First Law of Thermodynamics. The Second Law of Thermodynamics defines the concept of thermodynamic entropy, by measurable heat transfer.Thermal equilibrium is reached when all involved bodies and the surroundings reach the same temperature. Thermal expansion is the tendency of matter to change in volume in response to a change in temperature.