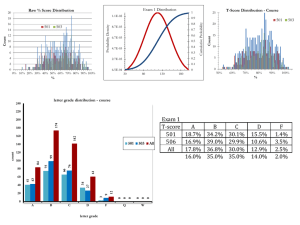
EXCESS HEAT DURING DIFFUSION OF DEUTERIUM THROUGH PALLADIUM
... Input power is measured accurately since the heater is driven by a DC power supply. Output power is measured via the temperature difference given by two thermistors, one at the inlet, and the other one at the outlet with a precision of +/- 0.01 K. De-ionized water mass flow rate is measured with acc ...
... Input power is measured accurately since the heater is driven by a DC power supply. Output power is measured via the temperature difference given by two thermistors, one at the inlet, and the other one at the outlet with a precision of +/- 0.01 K. De-ionized water mass flow rate is measured with acc ...
Chapter 5 Thermochemistry - Byron Senior High School
... discharged by running the fan, its E is the same. But q and w are different in the two cases. Thermochemistry ...
... discharged by running the fan, its E is the same. But q and w are different in the two cases. Thermochemistry ...
Thermodynamics and Kinetics
... •SI unit for specific heat is joules per gram-1 Kelvin-1 (J/g-K) Calorie - The specific heat of water = 4.184 J/g-K Molar heat capacity - The amount of heat required to raise the temperature of one mole of a substance by 1 C (or 1 K) •SI unit for molar heat capacity is joules per mole-1 Kelvin-1 (J/ ...
... •SI unit for specific heat is joules per gram-1 Kelvin-1 (J/g-K) Calorie - The specific heat of water = 4.184 J/g-K Molar heat capacity - The amount of heat required to raise the temperature of one mole of a substance by 1 C (or 1 K) •SI unit for molar heat capacity is joules per mole-1 Kelvin-1 (J/ ...
CHEM 1411 Exam #2 - HCC Learning Web
... _____16. A hot metal pan at 90°C weighing 363 g was immersed in 1000 g of water at 22.0°C. The final temperature of the pan and water is 26.9 °C. Calculate the specific heat of the metal. The specific heat of water is 4.184 J/g-°C. A. 2.82 J/g·oC ...
... _____16. A hot metal pan at 90°C weighing 363 g was immersed in 1000 g of water at 22.0°C. The final temperature of the pan and water is 26.9 °C. Calculate the specific heat of the metal. The specific heat of water is 4.184 J/g-°C. A. 2.82 J/g·oC ...
Lecture notes
... Enthalpy Enthalpy (H) is a term used to express the changes of energy in a system. It can be determined through measurement of the temperature change during a chemical reaction or a physical change at constant pressure. ∆U = q + -p∆V can be rearranged to: ∆U + p∆V = q = ∆H Reactions that evolve or ...
... Enthalpy Enthalpy (H) is a term used to express the changes of energy in a system. It can be determined through measurement of the temperature change during a chemical reaction or a physical change at constant pressure. ∆U = q + -p∆V can be rearranged to: ∆U + p∆V = q = ∆H Reactions that evolve or ...
Thermochemistry Thermochemistry
... of matter and deduces a few general laws – It does not require any knowledge/assumptions of molecules CHEM 1000A 3.0 ...
... of matter and deduces a few general laws – It does not require any knowledge/assumptions of molecules CHEM 1000A 3.0 ...
Why is S(H2O(l) > S(H20(g)? It is better to speak of entropy as a
... temperature (S = Q/T), and the change in entropy represents the amount of energy input to the system which does not participate in mechanical work done by the system (S = (Q - W)/T). In statistical mechanics, the interpretation is more general perhaps, where the entropy becomes a function of statist ...
... temperature (S = Q/T), and the change in entropy represents the amount of energy input to the system which does not participate in mechanical work done by the system (S = (Q - W)/T). In statistical mechanics, the interpretation is more general perhaps, where the entropy becomes a function of statist ...
Basic Thermodynamics - Text of NPTEL IIT Video Lectures
... That means, this work and heat quantities interactions are equal in magnitude, but opposite in direction (Refer Slide Time: 06:07). That is the characteristic feature of the reversible heat engine. But if we reverse the heat engine, HE, it will work as a heat pump. Do not consider that as it is an i ...
... That means, this work and heat quantities interactions are equal in magnitude, but opposite in direction (Refer Slide Time: 06:07). That is the characteristic feature of the reversible heat engine. But if we reverse the heat engine, HE, it will work as a heat pump. Do not consider that as it is an i ...
Thermodynamic temperature
... perature is the null or zero point. At this point, absolute a substance. More specifically, the thermodynamic temzero, the particle constituents of matter have minimal perature of any bulk quantity of matter is the measure motion and can become no colder.[1][2] In the quantum- of the average kinetic ...
... perature is the null or zero point. At this point, absolute a substance. More specifically, the thermodynamic temzero, the particle constituents of matter have minimal perature of any bulk quantity of matter is the measure motion and can become no colder.[1][2] In the quantum- of the average kinetic ...
Principles of Chemical Thermodynamics and Kinetics
... thermal equilibrium with each other. To summarize, should two systems be separate from each other but both be in thermal equilibrium with a third, then the first two are also in thermal equilibrium with each other. For example, if in a closed system you have three solid metal cubes all at different ...
... thermal equilibrium with each other. To summarize, should two systems be separate from each other but both be in thermal equilibrium with a third, then the first two are also in thermal equilibrium with each other. For example, if in a closed system you have three solid metal cubes all at different ...
Thermal Diffusion and Partial Molar Enthalpy Variations of n
... One aim of this work is to study transport properties which so far, to the best of our knowledge, were not studied in zeolites, namely, the coupling between heat and mass transfer. We have earlier found that large temperature gradients can arise in the material due to the relatively large heats of a ...
... One aim of this work is to study transport properties which so far, to the best of our knowledge, were not studied in zeolites, namely, the coupling between heat and mass transfer. We have earlier found that large temperature gradients can arise in the material due to the relatively large heats of a ...
Transport Phenomena - Hailie Nene Foundation
... the permeability Lp. This is often used in pressure driven membrane processes where a pressure difference across a membrane induces a volume flux through the membrane. Proportionality between the applied pressure and the flux is seen when pure water permeates through the membrane. A pressure differe ...
... the permeability Lp. This is often used in pressure driven membrane processes where a pressure difference across a membrane induces a volume flux through the membrane. Proportionality between the applied pressure and the flux is seen when pure water permeates through the membrane. A pressure differe ...
Physical Properties of Food Materials
... The study of food engineering focuses on the analysis of equipment and systems used to process food on a commercial production scale. Engineering of systems for food materials can be more thorough if there is an understanding of the changes that occur in food as it is processed by the system. Raw fo ...
... The study of food engineering focuses on the analysis of equipment and systems used to process food on a commercial production scale. Engineering of systems for food materials can be more thorough if there is an understanding of the changes that occur in food as it is processed by the system. Raw fo ...
Poultry House Temperature Control Using Fuzzy-PID
... is below the thermal neutral zone of the bird, energy from the feed will be is used to generate heat rather than for growth and development. If the temperature is above this zone, heat must be lost in some way; this tends to result in reduced feed conversion efficiency [5]. Traditional ways of contr ...
... is below the thermal neutral zone of the bird, energy from the feed will be is used to generate heat rather than for growth and development. If the temperature is above this zone, heat must be lost in some way; this tends to result in reduced feed conversion efficiency [5]. Traditional ways of contr ...
No Slide Title
... The negative sign in the above equation occurs because we are measuring the value of q for the surroundings, and qsyst = - qsurr. Since the combustion process occurs under conditions of constant volume, q = E, the change in internal energy. Example: 1.412 g of carbon (M = 12.01 g/mol) are burned in ...
... The negative sign in the above equation occurs because we are measuring the value of q for the surroundings, and qsyst = - qsurr. Since the combustion process occurs under conditions of constant volume, q = E, the change in internal energy. Example: 1.412 g of carbon (M = 12.01 g/mol) are burned in ...
Heat transfer

Heat transfer is the exchange of thermal energy between physical systems, depending on the temperature and pressure, by dissipating heat. The fundamental modes of heat transfer are conduction or diffusion, convection and radiation.Heat transfer always occurs from a region of high temperature to another region of lower temperature. Heat transfer changes the internal energy of both systems involved according to the First Law of Thermodynamics. The Second Law of Thermodynamics defines the concept of thermodynamic entropy, by measurable heat transfer.Thermal equilibrium is reached when all involved bodies and the surroundings reach the same temperature. Thermal expansion is the tendency of matter to change in volume in response to a change in temperature.