BSc in Chemistry-CUCBCSS UG 2014-Scheme
... With the pace that the world keeps and the speed with which technology advances, an understanding of science is inevitable in our day-to-day life. To make the study of science interesting and enjoyable, the creation of a scientific temper in society is a must which could be achieved through proper e ...
... With the pace that the world keeps and the speed with which technology advances, an understanding of science is inevitable in our day-to-day life. To make the study of science interesting and enjoyable, the creation of a scientific temper in society is a must which could be achieved through proper e ...
edexcel_u1_2010_2013..
... of hydrogen produced was 66 cm3, measured at room temperature and pressure. (i) Draw a labelled diagram of the apparatus you would use to carry out this experiment, showing how you would collect the hydrogen produced and ...
... of hydrogen produced was 66 cm3, measured at room temperature and pressure. (i) Draw a labelled diagram of the apparatus you would use to carry out this experiment, showing how you would collect the hydrogen produced and ...
9 SHS CH 9 LECTURE shs_ch_9_lecture
... not in mole units convert it to moles using the molecular mass as a conversion factor. Step 3. Now convert from the moles of starting substance to the moles of the desired substance by using a mole ratio from the chemical reaction. reaction. Step 4. If the problem is to find grams of the substance, ...
... not in mole units convert it to moles using the molecular mass as a conversion factor. Step 3. Now convert from the moles of starting substance to the moles of the desired substance by using a mole ratio from the chemical reaction. reaction. Step 4. If the problem is to find grams of the substance, ...
Adsorption and Reaction of SO2 with a Polycrystalline UO2 Film
... of physisorbed SO2 and chemisorbed SO3, as shown by the indicated curve fitting. Heating to 175 K causes a rapid decrease in the intensity of the S 2p peak; a small shift of the spectrum centroid to ∼167.5 eV is observed. This is explained by desorption of the physisorbed SO2 molecules at ∼133 K. Ho ...
... of physisorbed SO2 and chemisorbed SO3, as shown by the indicated curve fitting. Heating to 175 K causes a rapid decrease in the intensity of the S 2p peak; a small shift of the spectrum centroid to ∼167.5 eV is observed. This is explained by desorption of the physisorbed SO2 molecules at ∼133 K. Ho ...
Chapter 5 | Molecular Orbitals
... As mentioned previously, nonbonding molecular orbitals have energies essentially equal to that of atomic orbitals. These can form in larger molecules, for example when there are three atomic orbitals of the same symmetry and similar energies, a situation that requires the formation of three molecula ...
... As mentioned previously, nonbonding molecular orbitals have energies essentially equal to that of atomic orbitals. These can form in larger molecules, for example when there are three atomic orbitals of the same symmetry and similar energies, a situation that requires the formation of three molecula ...
The Concept of Collision Strength and Its Applications
... uniform expression. The new theory provides a simple and exact method for describing the equilibrium plasma collision rate, and a precise calculation of the equilibrium relaxation time. It generalizes bimolecular collision reaction rate theory to a reaction rate theory for plasmas. A simple formula ...
... uniform expression. The new theory provides a simple and exact method for describing the equilibrium plasma collision rate, and a precise calculation of the equilibrium relaxation time. It generalizes bimolecular collision reaction rate theory to a reaction rate theory for plasmas. A simple formula ...
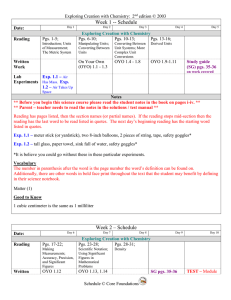
Week 1 -- Schedule
... ** Before you begin this science course please read the student notes in the book on pages i-iv. ** ** Parent – teacher needs to read the notes in the solutions / test manual ** Reading has pages listed, then the section names (or partial names). If the reading stops mid-section then the reading has ...
... ** Before you begin this science course please read the student notes in the book on pages i-iv. ** ** Parent – teacher needs to read the notes in the solutions / test manual ** Reading has pages listed, then the section names (or partial names). If the reading stops mid-section then the reading has ...
Novel Systems and Methods for Quantum
... quantum simulation with ultracold alkaline-earth atoms in optical lattices. We show how multiple qubits can be encoded in individual alkaline-earth atoms and harnessed for quantum computing and precision measurements applications. We also demonstrate that alkaline-earth atoms can be used to simulate ...
... quantum simulation with ultracold alkaline-earth atoms in optical lattices. We show how multiple qubits can be encoded in individual alkaline-earth atoms and harnessed for quantum computing and precision measurements applications. We also demonstrate that alkaline-earth atoms can be used to simulate ...
Entanglement in many body quantum systems Arnau Riera Graells
... els valors esperats d’alguns observables, també requerirà un nombre exponencial de passos. Aquesta és la raó per la qual no podem simular clàssicament qualsevol sistema quàntic de moltes partícules, i en particular, alguns interessants sistemes de la ...
... els valors esperats d’alguns observables, també requerirà un nombre exponencial de passos. Aquesta és la raó per la qual no podem simular clàssicament qualsevol sistema quàntic de moltes partícules, i en particular, alguns interessants sistemes de la ...
Composition and Evolution of Interstellar Clouds
... with the HIM, i.e., a tenuous, ionized coronal gas (McKee and Ostriker, 1977). Supernovae explosions generate the coronal gas (Spitzer, 1990) when their ejecta collide with and shock the surrounding medium. Expanding supernova ejecta sweep up ambient ISM material, compressing it into rapidly expandi ...
... with the HIM, i.e., a tenuous, ionized coronal gas (McKee and Ostriker, 1977). Supernovae explosions generate the coronal gas (Spitzer, 1990) when their ejecta collide with and shock the surrounding medium. Expanding supernova ejecta sweep up ambient ISM material, compressing it into rapidly expandi ...
BSc Honours chemistry CBCS Syllabus 2016-17
... Examiner will set nine questions in all, selecting two questions from section A, B, C, and D of 10 marks each and may contain more than one part. Section E will be of 10 marks and consists of objective type questions (MCQ/true and false / fill in the blanks etc.) of one mark each covering the entire ...
... Examiner will set nine questions in all, selecting two questions from section A, B, C, and D of 10 marks each and may contain more than one part. Section E will be of 10 marks and consists of objective type questions (MCQ/true and false / fill in the blanks etc.) of one mark each covering the entire ...
Methodological Advances in Theoretical - ETH E
... to deduce the desired information solely from spectroscopic measurements. Theoretical methods are a valuable complement to experiment since spectroscopic spectra can be calculated and predicted. In this way, not only the interpretation of the experimental results is facilitated but also additional i ...
... to deduce the desired information solely from spectroscopic measurements. Theoretical methods are a valuable complement to experiment since spectroscopic spectra can be calculated and predicted. In this way, not only the interpretation of the experimental results is facilitated but also additional i ...
Solving General Chemistry Problems 5e
... 7. Look for a relationship (a conceptual principle or a mathematical equation) between what is given and what is asked for. 8. Set up the problem in a concise, logical, stepwise manner, using units for all terms and factors. ...
... 7. Look for a relationship (a conceptual principle or a mathematical equation) between what is given and what is asked for. 8. Set up the problem in a concise, logical, stepwise manner, using units for all terms and factors. ...
CHAPTER 9 Notes
... theoretical yield: Amount of product one should get based on the chemical equation and the amount of reactants present -One generally calculates this in grams from info given Actual yield: Amount of produce one actually obtains -Generally smaller than the theoretical yield because of impurities and ...
... theoretical yield: Amount of product one should get based on the chemical equation and the amount of reactants present -One generally calculates this in grams from info given Actual yield: Amount of produce one actually obtains -Generally smaller than the theoretical yield because of impurities and ...
VOLUME 3 - ICHO 41-45 _opravené_
... the forty-five years of its existence. In the elaboration of this collection the editor had to face certain difficulties because the aim was not only to make use of past recordings but also to give them such a form that they may be used in practice and further chemical education. Consequently, it wa ...
... the forty-five years of its existence. In the elaboration of this collection the editor had to face certain difficulties because the aim was not only to make use of past recordings but also to give them such a form that they may be used in practice and further chemical education. Consequently, it wa ...
The de Broglie Wave as Evidence of a Deeper Wave Structure
... to vary sinusoidally in space as well as time. But for now, we will follow de Broglie in leaving the spatial variation of the wave “intentionally vague”. We want to know the form that this “periodic phenomenon” would take when observed from a frame of reference in which the particle is moving with v ...
... to vary sinusoidally in space as well as time. But for now, we will follow de Broglie in leaving the spatial variation of the wave “intentionally vague”. We want to know the form that this “periodic phenomenon” would take when observed from a frame of reference in which the particle is moving with v ...
National German Competition
... If you want to explain this particular reaction with the help of the Nernst equation you have to take into account a very low copper(II) concentration in both experiments. h) Calculate the limit of concentration of copper(II) ions below which the reaction should proceed theoretically - for the react ...
... If you want to explain this particular reaction with the help of the Nernst equation you have to take into account a very low copper(II) concentration in both experiments. h) Calculate the limit of concentration of copper(II) ions below which the reaction should proceed theoretically - for the react ...
Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.The word atom comes from the Ancient Greek adjective atomos, meaning ""uncuttable"". 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. While seemingly apropos, around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called ""uncuttable atom"" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all. Since atoms were found to be divisible, physicists later invented the term ""elementary particles"" to describe the ""uncuttable"", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.