Chemistry Appendixes
... Shaded circles indicate the parts you are expected to complete in a particular investigation report. One or more parts of an Evaluation may be required, as indicated by the numbers. ...
... Shaded circles indicate the parts you are expected to complete in a particular investigation report. One or more parts of an Evaluation may be required, as indicated by the numbers. ...
chemistry - University of Malaya
... the first institution of higher learning in Malaysia to receive the prestigious Royal Society of Chemistry, UK accreditation for its BSc (Chemistry) and BSc (Applied Chemistry) programme since August 2012. One of the objectives of the Department is to provide a centre of excellence in chemical educa ...
... the first institution of higher learning in Malaysia to receive the prestigious Royal Society of Chemistry, UK accreditation for its BSc (Chemistry) and BSc (Applied Chemistry) programme since August 2012. One of the objectives of the Department is to provide a centre of excellence in chemical educa ...
8 Momentum - mrfosterscience
... 8.4 Conservation of Momentum The force or impulse that changes momentum must be exerted on the object by something outside the object. • Molecular forces within a basketball have no effect on the momentum of the basketball. • A push against the dashboard from inside does not affect the momentum of a ...
... 8.4 Conservation of Momentum The force or impulse that changes momentum must be exerted on the object by something outside the object. • Molecular forces within a basketball have no effect on the momentum of the basketball. • A push against the dashboard from inside does not affect the momentum of a ...
Applied Physics Letters
... the studies from Xu et al., where they also find reduced modulation strength of the graphene Fermi level with electric field in ambient conditions.1,16 The experimental measurements find strong characteristics of CE enhancement for graphene. One possible explanation for the observed trend is that th ...
... the studies from Xu et al., where they also find reduced modulation strength of the graphene Fermi level with electric field in ambient conditions.1,16 The experimental measurements find strong characteristics of CE enhancement for graphene. One possible explanation for the observed trend is that th ...
Transition Metal-Modified Zirconium Phosphate Electrocatalysts for
... intercalated systems and that the metal‐adsorbed catalyst had retained α‐ZrP characteristics. In the metal-intercalated systems and that the metal-adsorbed catalyst had retained α-ZrP characteristics. case of pure α‐ZrP (Figure 3) two weight losses are observed, dehydration of water w ...
... intercalated systems and that the metal‐adsorbed catalyst had retained α‐ZrP characteristics. In the metal-intercalated systems and that the metal-adsorbed catalyst had retained α-ZrP characteristics. case of pure α‐ZrP (Figure 3) two weight losses are observed, dehydration of water w ...
Aligned AlN Nanorods with Multi-tipped Surfaces
... were used in the present study. The silicon wafers were cleaned chemically by the standard RCA cleaning process. The wafers were dipped in a dilute HF solution (HF/H2O = 1:50) for 2 min immediately before loading into a thermal-evaporation system. An ultrathin Ni film (3 nm thick) was deposited at ∼ ...
... were used in the present study. The silicon wafers were cleaned chemically by the standard RCA cleaning process. The wafers were dipped in a dilute HF solution (HF/H2O = 1:50) for 2 min immediately before loading into a thermal-evaporation system. An ultrathin Ni film (3 nm thick) was deposited at ∼ ...
Stoichiometry - Milton
... scientific revolution, very little was known about the process of combustion. In attempting to explain this common phenomenon, chemists of the 18th century developed one of the first universally accepted theories in their field. But as one man would show, scientific theories do not always stand the ...
... scientific revolution, very little was known about the process of combustion. In attempting to explain this common phenomenon, chemists of the 18th century developed one of the first universally accepted theories in their field. But as one man would show, scientific theories do not always stand the ...
Define resistance V splitting between components V
... PD from potential divider calc V=IR calc graph skills Explain why V changes more at low light intensity than high for LDR effect of rising resistance of one component on a circuit advantages of a data logger 5. Waves [16 marks] state a property of EM waves that other’s can’t do Name parts of the EM ...
... PD from potential divider calc V=IR calc graph skills Explain why V changes more at low light intensity than high for LDR effect of rising resistance of one component on a circuit advantages of a data logger 5. Waves [16 marks] state a property of EM waves that other’s can’t do Name parts of the EM ...
8 Momentum
... 8.4 Conservation of Momentum The force or impulse that changes momentum must be exerted on the object by something outside the object. • Molecular forces within a basketball have no effect on the momentum of the basketball. • A push against the dashboard from inside does not affect the momentum of a ...
... 8.4 Conservation of Momentum The force or impulse that changes momentum must be exerted on the object by something outside the object. • Molecular forces within a basketball have no effect on the momentum of the basketball. • A push against the dashboard from inside does not affect the momentum of a ...
Class XI worksheet - Indian School Muscat
... 10. Show that the wavelength related to a 250g ball moving with a speed of 100 m/s is too short to be observed. 11. i. State Heisenberg’s uncertainty principle. ii. The speed of an electron moving at 600m/s is measured to an accuracy of 0.005%. What would be the minimum error in determining its posi ...
... 10. Show that the wavelength related to a 250g ball moving with a speed of 100 m/s is too short to be observed. 11. i. State Heisenberg’s uncertainty principle. ii. The speed of an electron moving at 600m/s is measured to an accuracy of 0.005%. What would be the minimum error in determining its posi ...
Cookies and Chemistry…Huh!?!?
... Makes 3 dozen If we had the specified amount of all ingredients listed, could we make 4 dozen cookies? What if we had 6 eggs and twice as much of everything else, could we make 9 dozen cookies? What if we only had one egg, could we make 3 dozen cookies? ...
... Makes 3 dozen If we had the specified amount of all ingredients listed, could we make 4 dozen cookies? What if we had 6 eggs and twice as much of everything else, could we make 9 dozen cookies? What if we only had one egg, could we make 3 dozen cookies? ...
Chemistry - A Quantitative Science
... done the same as converting dozens to items. 1.5 doz = (1.5 doz)(12 items.doz-1) = 18 items and 1.5 mol = (1.5 mol)( 6.0x1023 atoms.mol-1) = 9.0x1023 atoms. The mole is used simply because it is much easier to discuss the number of atoms in moles than it is as individual items - 0.10 mol H2O is a mu ...
... done the same as converting dozens to items. 1.5 doz = (1.5 doz)(12 items.doz-1) = 18 items and 1.5 mol = (1.5 mol)( 6.0x1023 atoms.mol-1) = 9.0x1023 atoms. The mole is used simply because it is much easier to discuss the number of atoms in moles than it is as individual items - 0.10 mol H2O is a mu ...
Chapter+12
... Makes 3 dozen If we had the specified amount of all ingredients listed, could we make 4 dozen cookies? What if we had 6 eggs and twice as much of everything else, could we make 9 dozen cookies? What if we only had one egg, could we make 3 dozen cookies? ...
... Makes 3 dozen If we had the specified amount of all ingredients listed, could we make 4 dozen cookies? What if we had 6 eggs and twice as much of everything else, could we make 9 dozen cookies? What if we only had one egg, could we make 3 dozen cookies? ...
The General Relativistic Two Body Problem and the Effective One
... and the coalescence of binary neutron stars. Much physics remains to be explored in these systems, especially during and after the merger of the neutron stars (which involves a much more complex physics than the pure-gravity merger of two black holes). Recently, a new source of information on the ge ...
... and the coalescence of binary neutron stars. Much physics remains to be explored in these systems, especially during and after the merger of the neutron stars (which involves a much more complex physics than the pure-gravity merger of two black holes). Recently, a new source of information on the ge ...
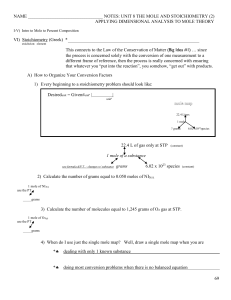
NAME NOTES: UNIT 8 THE MOLE AND STOICHIOMETRY (2
... that whatever you “put into the reaction”, you somehow, “get out” with products. A) How to Organize Your Conversion Factors 1) Every beginning to a stoichiometry problem should look like: ...
... that whatever you “put into the reaction”, you somehow, “get out” with products. A) How to Organize Your Conversion Factors 1) Every beginning to a stoichiometry problem should look like: ...
Review Problems for Introductory Physics 1
... The problems in this review guide are provided as is without any guarantee of being correct! That’s not to suggest that they are all broken – on the contrary, most of them are well-tested and have been used as homework, quiz and exam problems for decades if not centuries. It is to suggest that they ...
... The problems in this review guide are provided as is without any guarantee of being correct! That’s not to suggest that they are all broken – on the contrary, most of them are well-tested and have been used as homework, quiz and exam problems for decades if not centuries. It is to suggest that they ...
Answers to SelectedTextbook Questions
... (b) In the Bohr model, a hydrogen atom consists of an electron in a circular orbit about a proton. (c) A conical flask used in chemistry labs to carry out reactions. (d) van der Waals equation is a relation between the pressure, temperature and volume of a gas that accounts for the non‐zero ...
... (b) In the Bohr model, a hydrogen atom consists of an electron in a circular orbit about a proton. (c) A conical flask used in chemistry labs to carry out reactions. (d) van der Waals equation is a relation between the pressure, temperature and volume of a gas that accounts for the non‐zero ...
Chapter 1: Matter and Measurements
... (a) “Compounds containing carbon atoms are molecular” is usually true. Carbon, being a nonmetal can combine with other non-metals to form molecular compounds such as those mentioned in Section 2.4 (indeed, a whole field of chemistry, called organic chemistry, is dedicated to the study of such compou ...
... (a) “Compounds containing carbon atoms are molecular” is usually true. Carbon, being a nonmetal can combine with other non-metals to form molecular compounds such as those mentioned in Section 2.4 (indeed, a whole field of chemistry, called organic chemistry, is dedicated to the study of such compou ...
CHAPTER 21 ELECTROCHEMISTRY: CHEMICAL CHANGE AND
... No, one half-reaction cannot take place independent of the other because there is always a transfer of electrons from one substance to another. If one substance loses electrons (oxidation half-reaction), another substance must gain those electrons (reduction half-reaction). ...
... No, one half-reaction cannot take place independent of the other because there is always a transfer of electrons from one substance to another. If one substance loses electrons (oxidation half-reaction), another substance must gain those electrons (reduction half-reaction). ...
Rh(acac)(CO)(PR1R2R3) - University of the Free State
... [Rh(acac)(CO)(PPh3)] (1), [Rh(acac)(CO)(PCyPh2)] (2), [Rh(acac)(CO)(PCy2Ph)] (3) and [Rh(acac)(CO)(PCy3)] (4) which were successfully completed. Selected crystallographic parameters are listed in the table below. ...
... [Rh(acac)(CO)(PPh3)] (1), [Rh(acac)(CO)(PCyPh2)] (2), [Rh(acac)(CO)(PCy2Ph)] (3) and [Rh(acac)(CO)(PCy3)] (4) which were successfully completed. Selected crystallographic parameters are listed in the table below. ...
Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.The word atom comes from the Ancient Greek adjective atomos, meaning ""uncuttable"". 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. While seemingly apropos, around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called ""uncuttable atom"" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all. Since atoms were found to be divisible, physicists later invented the term ""elementary particles"" to describe the ""uncuttable"", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.