Enzymes
... particular configuration of the active site into which the substrate molecules fit like a key, giving rise to the lock and key hypothesis. This hypothesis has been modified to the induced fit hypothesis, where it is thought that when a Substrate combines with an enzyme, it induces the enzyme structu ...
... particular configuration of the active site into which the substrate molecules fit like a key, giving rise to the lock and key hypothesis. This hypothesis has been modified to the induced fit hypothesis, where it is thought that when a Substrate combines with an enzyme, it induces the enzyme structu ...
Cardiff International School Dhaka (CISD) Lost Class Make Up
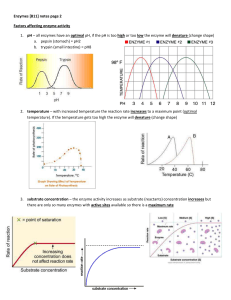
... (b) Heavy metals (Ag, Hg, TI), ions react with one or more sulfhydryl groups, replacing the hydrogen atom with a metal ion. Q2. (a) At temperatures between 0o C and about 40 o C the rate of enzyme activity increases with temperature. Enzyme activity decreases markedly above 40 o C. (b) As temperatur ...
... (b) Heavy metals (Ag, Hg, TI), ions react with one or more sulfhydryl groups, replacing the hydrogen atom with a metal ion. Q2. (a) At temperatures between 0o C and about 40 o C the rate of enzyme activity increases with temperature. Enzyme activity decreases markedly above 40 o C. (b) As temperatur ...
BIO 330 Cell Biology Lecture Outline Spring 2011 Chapter 6
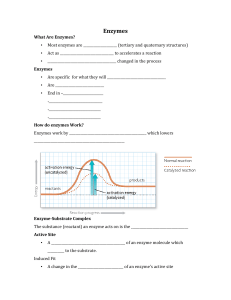
... Products are released following reaction IV. Enzyme Kinetics A. Reaction rates are dependent upon… Substrate concentration Enzyme concentration Product concentration Inhibitor concentration B. Michaelis-Menten kinetics Initial reaction velocity (v) Substrate concentration ([S]) Vmax = maximum veloci ...
... Products are released following reaction IV. Enzyme Kinetics A. Reaction rates are dependent upon… Substrate concentration Enzyme concentration Product concentration Inhibitor concentration B. Michaelis-Menten kinetics Initial reaction velocity (v) Substrate concentration ([S]) Vmax = maximum veloci ...
Control of Metabolic Pathways (2)
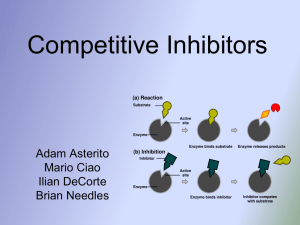
... • These compete with molecules of substrate for the active sites on an enzyme • Molecular structure similar to the substrate • Rate of reaction is reduced ...
... • These compete with molecules of substrate for the active sites on an enzyme • Molecular structure similar to the substrate • Rate of reaction is reduced ...
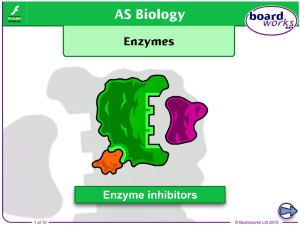
Boardworks Enzyme Inhibitors
... whether they compete with the substrate for binding at the active site or not. ...
... whether they compete with the substrate for binding at the active site or not. ...
key
... 1. pH – all enzymes have an optimal pH, if the pH is too high or too low the enzyme will denature (change shape) a. pepsin (stomach) = pH2 ...
... 1. pH – all enzymes have an optimal pH, if the pH is too high or too low the enzyme will denature (change shape) a. pepsin (stomach) = pH2 ...
CLS 431 – midterm exam
... Lactate dehydrogenase (LDH) Alanine transaminase (ALT) Creatine kinase (CK) Amylase (AMY) ...
... Lactate dehydrogenase (LDH) Alanine transaminase (ALT) Creatine kinase (CK) Amylase (AMY) ...
Enzyme_Activity_and_Regulation_Internet_Activity updated 1
... Directions: You are going to view a series of on-line animations to help you better understand enzymes. Log onto http://www.4teachers.org/ and click onto Track Star icon along the right margin. View Track # 240347. The password is “ferg”. You should view the track in text. Visit each of the sites; t ...
... Directions: You are going to view a series of on-line animations to help you better understand enzymes. Log onto http://www.4teachers.org/ and click onto Track Star icon along the right margin. View Track # 240347. The password is “ferg”. You should view the track in text. Visit each of the sites; t ...
Interfering with enzymes (poisons and drugs)
... • “A non-protein organic molecule that forms a permanent part of a functioning protein molecule.” • E.g. zinc-based prosthetic group in carbonic anhydrase – where have we met this enzyme? ...
... • “A non-protein organic molecule that forms a permanent part of a functioning protein molecule.” • E.g. zinc-based prosthetic group in carbonic anhydrase – where have we met this enzyme? ...
Lehninger Principles of Biochemistry
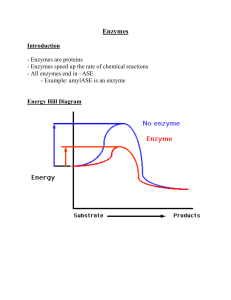
... There is an energy barrier between formation of product from substrate There is an activation energy for formation of the transition state ...
... There is an energy barrier between formation of product from substrate There is an activation energy for formation of the transition state ...
chapter 20
... 4. Discuss the difference between the “lock and key” and “induced fit” models of enzyme catalysis. 5. What are some factors that affect enzyme activity? What happens to the rate of the reaction as the available enzyme molecules become saturated with substrate? 6. Discuss differences between competit ...
... 4. Discuss the difference between the “lock and key” and “induced fit” models of enzyme catalysis. 5. What are some factors that affect enzyme activity? What happens to the rate of the reaction as the available enzyme molecules become saturated with substrate? 6. Discuss differences between competit ...
Enzymes
... Inhibitors that ______________________ the active site, but __________________________________________ of the enzyme causing the enzyme to _________________________________________, which in turn ...
... Inhibitors that ______________________ the active site, but __________________________________________ of the enzyme causing the enzyme to _________________________________________, which in turn ...
Competitive Inhibitors
... at the active site, where the substrates would normally bind. • This protein has a similar shape of the substrate that binds to the enzyme, it will cause the enzyme to stop working (inhibit). • An inhibitor can be reversible if the concentration of the substrate is increased enough. ...
... at the active site, where the substrates would normally bind. • This protein has a similar shape of the substrate that binds to the enzyme, it will cause the enzyme to stop working (inhibit). • An inhibitor can be reversible if the concentration of the substrate is increased enough. ...
Enzyme inhibitor

An enzyme inhibitor is a molecule that binds to an enzyme and decreases its activity. Since blocking an enzyme's activity can kill a pathogen or correct a metabolic imbalance, many drugs are enzyme inhibitors. They are also used in pesticides. Not all molecules that bind to enzymes are inhibitors; enzyme activators bind to enzymes and increase their enzymatic activity, while enzyme substrates bind and are converted to products in the normal catalytic cycle of the enzyme.The binding of an inhibitor can stop a substrate from entering the enzyme's active site and/or hinder the enzyme from catalyzing its reaction. Inhibitor binding is either reversible or irreversible. Irreversible inhibitors usually react with the enzyme and change it chemically (e.g. via covalent bond formation). These inhibitors modify key amino acid residues needed for enzymatic activity. In contrast, reversible inhibitors bind non-covalently and different types of inhibition are produced depending on whether these inhibitors bind to the enzyme, the enzyme-substrate complex, or both.Many drug molecules are enzyme inhibitors, so their discovery and improvement is an active area of research in biochemistry and pharmacology. A medicinal enzyme inhibitor is often judged by its specificity (its lack of binding to other proteins) and its potency (its dissociation constant, which indicates the concentration needed to inhibit the enzyme). A high specificity and potency ensure that a drug will have few side effects and thus low toxicity.Enzyme inhibitors also occur naturally and are involved in the regulation of metabolism. For example, enzymes in a metabolic pathway can be inhibited by downstream products. This type of negative feedback slows the production line when products begin to build up and is an important way to maintain homeostasis in a cell. Other cellular enzyme inhibitors are proteins that specifically bind to and inhibit an enzyme target. This can help control enzymes that may be damaging to a cell, like proteases or nucleases. A well-characterised example of this is the ribonuclease inhibitor, which binds to ribonucleases in one of the tightest known protein–protein interactions. Natural enzyme inhibitors can also be poisons and are used as defences against predators or as ways of killing prey.