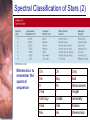
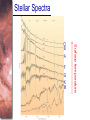
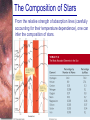
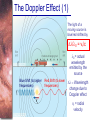
* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter6-7
Lovell Telescope wikipedia , lookup
Allen Telescope Array wikipedia , lookup
Optical telescope wikipedia , lookup
James Webb Space Telescope wikipedia , lookup
Spitzer Space Telescope wikipedia , lookup
International Ultraviolet Explorer wikipedia , lookup
Reflecting telescope wikipedia , lookup
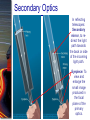
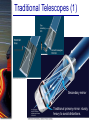
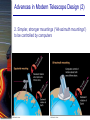
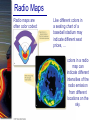
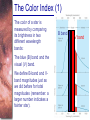
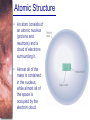
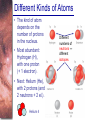
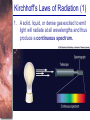
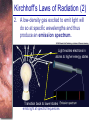
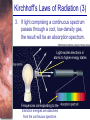
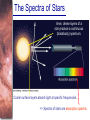
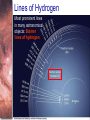
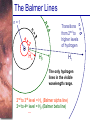
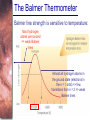
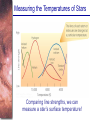
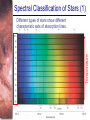
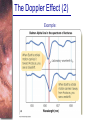
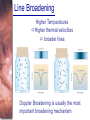
Chapter 6 Light and Telescopes Outline I. Radiation: Information from Space A. Light as a Wave and a Particle B. The Electromagnetic Spectrum II. Optical Telescopes A. Two Kinds of Telescopes B. The Powers of a Telescope C. Buying a Telescope D. New-Generation Telescopes E. Interferometry III. Special Instruments A. Imaging Systems B. The Spectrograph Outline (continued) IV. Radio Telescopes A. Operation of a Radio Telescope B. Limitations of the Radio Telescope C. Advantages of Radio Telescopes V. Astronomy from Space A. The Ends of the Visual Spectrum B. Telescopes in Space C. Cosmic Rays Light and Other Forms of Radiation • The Electromagnetic Spectrum In astronomy, we cannot perform experiments with our objects (stars, galaxies, …). The only way to investigate them, is by analyzing the light (and other radiation) which we observe from them. Light as a Wave (1) l c = 300,000 km/s = 3*108 m/s • Light waves are characterized by a wavelength l and a frequency f. • f and l are related through f = c/l Light as a Wave (2) • Wavelengths of light are measured in units of nanometers (nm) or Ångström (Å): 1 nm = 10-9 m 1 Å = 10-10 m = 0.1 nm Visible light has wavelengths between 4000 Å and 7000 Å (= 400 – 700 nm). Wavelengths and Colors Different colors of visible light correspond to different wavelengths. Light as Particles • Light can also appear as particles, called photons (explains, e.g., photoelectric effect). • A photon has a specific energy E, proportional to the frequency f: E = h*f h = 6.626x10-34 J*s is the Planck constant. The energy of a photon does not depend on the intensity of the light!!! The Electromagnetic Spectrum Wavelength Frequency Need satellites to observe High flying air planes or satellites Optical Telescopes Astronomers use telescopes to gather more light from astronomical objects. The larger the telescope, the more light it gathers. Refracting/Reflecting Telescopes Focal length Focal length Refracting Telescope: Lens focuses light onto the focal plane Reflecting Telescope: Concave Mirror focuses light onto the focal plane Almost all modern telescopes are reflecting telescopes. Secondary Optics In reflecting telescopes: Secondary mirror, to redirect the light path towards the back or side of the incoming light path. Eyepiece: To view and enlarge the small image produced in the focal plane of the primary optics. Seeing Weather conditions and turbulence in the atmosphere set further limits to the quality of astronomical images. Bad seeing Good seeing The Best Location for a Telescope Far away from civilization – to avoid light pollution The Best Location for a Telescope (2) Paranal Observatory (ESO), Chile On high mountain-tops – to avoid atmospheric turbulence ( seeing) and other weather effects Traditional Telescopes (1) Secondary mirror Traditional primary mirror: sturdy, heavy to avoid distortions. Traditional Telescopes (2) The 4-m Mayall Telescope at Kitt Peak National Observatory (Arizona) Advances in Modern Telescope Design (1) Modern computer technology has made possible significant advances in telescope design: Segmented mirror 1. Lighter mirrors with lighter support structures, to be controlled dynamically by computers Floppy mirror Adaptive Optics Computer-controlled mirror support adjusts the mirror surface (many times per second) to compensate for distortions by atmospheric turbulence Advances in Modern Telescope Design (2) 2. Simpler, stronger mountings (“Alt-azimuth mountings”) to be controlled by computers Examples of Modern Telescope Design (1) Design of the Large Binocular Telescope (LBT) The control room of the 4-m Mayall Telescope on Kitt Peak. Examples of Modern Telescope Design (2) The Very Large Telescope (VLT) 8.1-m mirror of the Gemini Telescopes The Spectrograph Using a prism (or a grating), light can be split up into different wavelengths (colors!) to produce a spectrum. Spectral lines in a spectrum tell us about the chemical composition and other properties of the observed object Radio Astronomy Recall: Radio waves of l ~ 1 cm – 1 m also penetrate the Earth’s atmosphere and can be observed from the ground. Radio Telescopes Large dish focuses the energy of radio waves onto a small receiver (antenna) Amplified signals are stored in computers and converted into images, spectra, etc. Radio Maps Radio maps are often color coded: Like different colors in a seating chart of a baseball stadium may indicate different seat prices, … colors in a radio map can indicate different intensities of the radio emission from different locations on the sky. Radio Interferometry Just as for optical telescopes, the resolving power of a radio telescope is amin = 1.22 l/D. For radio telescopes, this is a big problem: Radio waves are much longer than visible light Use interferometry to improve resolution! Radio Interferometry (2) The Very Large Array (VLA): 27 dishes are combined to simulate a large dish of 36 km in diameter. Even larger arrays consist of dishes spread out over the entire U.S. (VLBA = Very Long Baseline Array) or even the whole Earth (VLBI = Very Long Baseline Interferometry)! The Largest Radio Telescopes The 300-m telescope in Arecibo, Puerto Rico The 100-m Green Bank Telescope in Green Bank, WVa. Science of Radio Astronomy Radio astronomy reveals several features, not visible at other wavelengths: • Neutral hydrogen clouds (which don’t emit any visible light), containing ~ 90 % of all the atoms in the Universe. • Molecules (often located in dense clouds, where visible light is completely absorbed). • Radio waves penetrate gas and dust clouds, so we can observe regions from which visible light is heavily absorbed. Infrared Astronomy Most infrared radiation is absorbed in the lower atmosphere. NASA infrared telescope on Mauna Kea, Hawaii Infrared cameras need to be cooled to very low temperatures, usually using liquid nitrogen. However, from high mountain tops or high-flying air planes, some infrared radiation can still be observed. NASA’s Space Infrared Telescope Facility (SIRTF) Infrared light with wavelengths much longer than visible light (“Far Infrared”) can only be observed from space. The Hubble Space Telescope • Launched in 1990; maintained and upgraded by several space shuttle service missions throughout the 1990s and early 2000’s • Avoids turbulence in the Earth’s atmosphere • Extends imaging and spectroscopy to (invisible) infrared and ultraviolet Chapter 7 Starlight and Atoms Outline I. Starlight A. Temperature and Heat B. The Origin of Starlight C. Two Radiation Laws II. Atoms A. A Model Atom B. Different Kinds of Atoms C. Electron Shells III. The Interaction of Light and Matter A. The Excitation of Atoms B. The Formation of a Spectrum Outline (continued) IV. Stellar Spectra A. The Balmer Thermometer B. Spectral Classification C. The Composition of the Stars D. The Doppler Effect E. How the Doppler Shift Works F. Calculating the Doppler Velocity G. The Shapes of Spectral Lines The Amazing Power of Starlight Just by analyzing the light received from a star, astronomers can retrieve information about a star’s 1. Total energy output 2. Surface temperature 3. Radius 4. Chemical composition 5. Velocity relative to Earth 6. Rotation period Color and Temperature Stars appear in different colors, from blue (like Rigel) Orion Betelgeuse via green / yellow (like our sun) to red (like Betelgeuse). These colors tell us about the star’s temperature. Rigel Black Body Radiation (1) The light from a star is usually concentrated in a rather narrow range of wavelengths. The spectrum of a star’s light is approximately a thermal spectrum called a black body spectrum. A perfect black body emitter would not reflect any radiation. Thus the name “black body”. Two Laws of Black Body Radiation 1. The hotter an object is, the more energy it emits: Energy Flux F = s*T4 where F = Energy Flux = = Energy given off in the form of radiation, per unit time and per unit surface area [J/s/m2]; s = Stefan-Boltzmann constant Two Laws of Black Body Radiation 2. The peak of the black body spectrum shifts towards shorter wavelengths when the temperature increases. Wien’s displacement law: lmax ≈ 3,000,000 nm / TK (where TK is the temperature in Kelvin). The Color Index (1) The color of a star is measured by comparing its brightness in two different wavelength bands: The blue (B) band and the visual (V) band. We define B-band and Vband magnitudes just as we did before for total magnitudes (remember: a larger number indicates a fainter star). B band V band The Color Index (2) We define the Color Index B–V (i.e., B magnitude – V magnitude). The bluer a star appears, the smaller the color index B – V. The hotter a star is, the smaller its color index B – V. Light and Matter Spectra of stars are more complicated than pure blackbody spectra. characteristic lines, called absorption lines. To understand those lines, we need to understand atomic structure and the interactions between light and atoms. Atomic Structure • An atom consists of an atomic nucleus (protons and neutrons) and a cloud of electrons surrounding it. • Almost all of the mass is contained in the nucleus, while almost all of the space is occupied by the electron cloud. Nuclear Density If you could fill a teaspoon just with material as dense as the matter in an atomic nucleus, it would weigh ~ 2 billion tons!! Different Kinds of Atoms • The kind of atom depends on the number of protons in the nucleus. • Most abundant: Hydrogen (H), with one proton (+ 1 electron). • Next: Helium (He), with 2 protons (and 2 neutrons + 2 el.). Helium 4 Different numbers of neutrons ↔ different isotopes Electron Orbits • Electron orbits in the electron cloud are restricted to very specific radii and energies. r3, E3 r2, E2 r1, E1 • These characteristic electron energies are different for each individual element. Atomic Transitions • An electron can be kicked into a higher orbit when it absorbs a photon with exactly the right energy. Eph = E3 – E1 Eph = E4 – E1 Wrong energy • The photon is absorbed, and the electron is in an excited state. (Remember that Eph = h*f) • All other photons pass by the atom unabsorbed. Kirchhoff’s Laws of Radiation (1) 1. A solid, liquid, or dense gas excited to emit light will radiate at all wavelengths and thus produce a continuous spectrum. Kirchhoff’s Laws of Radiation (2) 2. A low-density gas excited to emit light will do so at specific wavelengths and thus produce an emission spectrum. Light excites electrons in atoms to higher energy states Transition back to lower states emits light at specific frequencies Kirchhoff’s Laws of Radiation (3) 3. If light comprising a continuous spectrum passes through a cool, low-density gas, the result will be an absorption spectrum. Light excites electrons in atoms to higher energy states Frequencies corresponding to the transition energies are absorbed from the continuous spectrum. The Spectra of Stars Inner, dense layers of a star produce a continuous (blackbody) spectrum. Cooler surface layers absorb light at specific frequencies. => Spectra of stars are absorption spectra. Analyzing Absorption Spectra • Each element produces a specific set of absorption (and emission) lines. • Comparing the relative strengths of these sets of lines, we can study the composition of gases. By far the most abundant elements in the Universe Lines of Hydrogen Most prominent lines in many astronomical objects: Balmer lines of hydrogen The Balmer Lines n=1 Transitions from 2nd to higher levels of hydrogen Ha Hb Hg The only hydrogen lines in the visible wavelength range. 2nd to 3rd level = Ha (Balmer alpha line) 2nd to 4th level = Hb (Balmer beta line) … Observations of the H-Alpha Line Emission nebula, dominated by the red Ha line. Absorption Spectrum Dominated by Balmer Lines Modern spectra are usually recorded digitally and represented as plots of intensity vs. wavelength The Balmer Thermometer Balmer line strength is sensitive to temperature: Most hydrogen atoms are ionized => weak Balmer lines Almost all hydrogen atoms in the ground state (electrons in the n = 1 orbit) => few transitions from n = 2 => weak Balmer lines Measuring the Temperatures of Stars Comparing line strengths, we can measure a star’s surface temperature! Spectral Classification of Stars (1) Temperature Different types of stars show different characteristic sets of absorption lines. Spectral Classification of Stars (2) Mnemonics to remember the spectral sequence: Oh Oh Only Be Boy, Bad A An Astronomers Fine F Forget Girl/Guy Grade Generally Kiss Kills Known Me Me Mnemonics Stellar Spectra F G K M Surface temperature O B A The Composition of Stars From the relative strength of absorption lines (carefully accounting for their temperature dependence), one can infer the composition of stars. The Doppler Effect (1) The light of a moving source is blue/red shifted by Dl/l0 = vr/c l0 = actual wavelength emitted by the source Blue Shift (to higher frequencies) vr Red Shift (to lower frequencies) Dl = Wavelength change due to Doppler effect vr = radial velocity The Doppler Effect (2) The Doppler effect allows us to measure the source’s radial velocity. The Doppler Effect (2) Example: The Doppler Effect (4) Take l0 of the Ha (Balmer alpha) line: l0 = 656 nm Assume, we observe a star’s spectrum with the Ha line at l = 658 nm. Then, Dl = 2 nm. We find Dl/l0 = 0.003 = 3*10-3 Thus, vr/c = 0.003, or vr = 0.003*300,000 km/s = 900 km/s. The line is red shifted, so the star is receding from us with a radial velocity of 900 km/s. Doppler Broadening In principle, line absorption should only affect a very unique wavelength. In reality, also slightly different wavelengths are absorbed. ↔ Lines have a finite width; we say: Blue shifted abs. Red shifted abs. vr vr Atoms in random thermal motion they are broadened. One reason for broadening: The Doppler effect! Observer Line Broadening Higher Temperatures Higher thermal velocities broader lines Doppler Broadening is usually the most important broadening mechanism.