* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download 06. urinary embryo
Survey
Document related concepts
Transcript
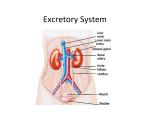
Development of Urinary System The urogenital system develops from the intermediate mesoderm which extends along the dorsal body wall of the embryo During folding of the embryo in the horizontal plane this mesoderm is carried ventrally and loses its connection with somites. A longitudinal elevation of mesoderm forms on each side of the dorsal aorta – The Urogenital Ridge. This ridge gives rise to parts of the urinary system which is the nephrogenic cord or ridge. Development of the Kidneys 3 sets are formed. The first set is the pronephroi: These transitory, nonfunctional structures appear in the 4th week. The pronephroi are represented by a few cell clusters and tubular structures in the neck region. The pronephric ducts run caudally and open into the cloaca. The rudimentary pronephroi soon degenerate. Most of the pronephric ducts persist and are utilized by the next set of the kidneys. Mesonephroi These large; elongated; excretory organs appear late in the 4th week caudal to the rudimentary pronephroi. They are well developed and function as inteim kidneys for about 4 weeks. It consists of glomeruli and mesonephric tubules. These tubules open into the mesonephric ducts. The mesonephric ducts open into the cloaca. The mesonephroi degenerate toward the end of the first trimester. In male, the tubules become the Efferent ductules of the testes. In female, it become the Epoophoron and Paroophoron. The duct gives rise in male to paradidymis; appendix of epididymis; duct of epididymis; ductus deferens; ureter; pelvis; calices; collecting tubules; ejaculatory duct and seminal gland. The duct in female gives rise to appendix vesiculosa; duct of epoophron; duct of Gartner; ureter ; pelvis; calices and collecting tubules. From 5th to 8th week Metanephroi The primordia of permanent kidneys begin to develop early in the 5th week and start to function about 4 weeks later. The permanent kidneys develop from 2 sources, the metanephreic diverticulum ( ureteric bud ) and the metanephric mass of intermediate mesoderm ( metanephrogenic blastema ). The metanephric diverticulum is an outgrowth from the mesonephric duct near its entrance into the cloaca. The metanephric mass of the intermediate mesoderm is derived from the caudal part of the nephrogenic cord. Both primordia of the metanephros are mesodermal origin. Metanephric diverticulum; It is the primordium of the ureter; renal pelvis; calices and collecting tubules. As it elongates, it penetrates the metanephric mass of the intermediate mesoderm. The stalk of it becomes the ureter and its expanded cranial end forms the renal pelvis. The straight collecting tubules undergo repeated branching, forming generations of tubules. The first 4 generations enlarge and become confluent to form the major calices. The 2nd four generations coalesce to form the minor calices. The remaining generations of tubules form the collecting tubules. The end of each arched collecting tubule induces clusters of mesenchymal cells in the metanephric mass of mesoderm to form metanephric vesicles. These vesicles elongate and become metanephric tubules. The proximal ends of these tubules are invaginated by glomeruli. Between the 10th & 18th weeks of gestation, the number of the glomeruli increases until the 32 week, when an upper limit is reached. Their filtration begins around the 9th fetal week and the rate of filtration increases after birth. The renal corpuscle ( glomerulus and glomerular capsule ) and its proximal convoluted tubules; nephron loop ( of Henle ) and distal convoluted tubule constitute a nephron. Each distal convoluted tubules contacts an arched collecting tubule. A uriniferous tubule: It consists of 2 different parts 1.Nephron from the metanephron mass of intermediate mesoderm. The nephron formation is complete at birth. 2- Collecting tubule from the metanephric diverticulum. The metanephric diverticulum and the metanephric mass of intermediate mesoderm interact and induce each other by a process known as reciprocal induction to form the permanent kidneys. Transformation of the metanephric mesenchyme to epithelial cells of the nephon ( mesenchymal- epithelial transition ) is regulated by factors secreted by the metanephric diverticulum. Fetal kidneys They are divided into lobes. This lobulation diminishes toward the end of fetal period, but the lobes are still inducated in the kidneys of a newborn infant. These lobulation disappears by the end of the first postnatal year during infancy as the nephrons increase and grow. At term, each kidney contains 800,000 to 1000,000 nephrons. The increase in kidney size after birth results mainly from the elongation of the proximal convoluted tubules as well as an increase of interstitial tissue. Functional maturation of the kidneys occurs after birth. Positional Changes of Kidneys Initially the metanephric kidneys lie close to each other in the pelvis ventral to the sacrum. As the abdomen and pelvis grow, caudal to the kidneys, so that they occupy more cranial level. They gradually come to lie in the abdomen and move farther apart and attain their adult position by 9th week. Initially the hilum faces ventrally, however, as the kidney ascends it rotates medially 90 degrees. By the 9th week the hilum is directed anteromedially. Changes in Blood Supply of kidneys Initially, the renal arteries are branches of the common iliac arteries. As they ascend, they receive their blood supply from the distal end of the aorta. Then, they receive new branches from the aorta. When they come into contact with the suprarenal glands in the 9th week their ascend stops. Normally, the caudal branches undergo involution and disappear. The right renal artery is longer and often more superior. Cystic kidney Disease Polycystic kidney is an autosomal recessive disorder ( ARPKD ) that is diagnosed at birth or in utero by ultrasonography. Both kidneys contains many hundred small cysts which results in renal insufficiency. Gene mutations have been implicated. Death of the infant usually occurs shortly after birth. These infants may survive by using postnatal dialysis and kidney transplantation. Multicystic dysplastic kidney disease (MDK) results from dysmorphology during development of the renal system. The outcome for children with MKD is generally good since, in 75% of cases, the disease is unilateral. In it, fewer cysts are seen than in ARPKD and can range in size from a few millimeters to many centimeters in the same kidney. Now, it is believed that the cystic structures are wide dilations of parts of the otherwise continuous nephrons, particularly the nephron loops ( of Henle) . 5 week Development of Urinary Bladder Division of the cloaca by urorectal septum into dorsal rectum and a ventral urogenital sinus. The urogenital sinus is divided into 3 parts: 1- A cranial vesical part that forms most of the bladder and is continuous with the allantois. 2- A middle pelvic part that becomes the urethra in the bladder neck and the prostatic part of urethra in male and the entire urethra in females. 3- A caudal phallic part that grows toward the genital tubercle. 5 week Development of Urinary Bladder The bladder develops mainly from the vesical part of the urogenital sinus, but the trigon is derived from the caudal ends of the mesonephric ducts. The epithelium of the entire bladder is derived from the endoderm of the vesical part of the urogenital sinus. The other layers of its wall develop from adjacent splanchnic mesenchyme As the bladder enlarges, the distal parts of the mesonephric ducts are incorporated into its dorsal wall. These ducts contribute to the formation of the connective tissue in the trigone of the bladder. As the caudal ends mesonephric ducts are absorbed, the ureters come to open separately into the urinary bladder. 18 week 12 weeks Initially the bladder is continuous with the allantois. The allantois become constricted and form a thick fibrous cord, the urachus which extends from the apex of the bladder to the umbilicus. In the adult the urachus is represented by the median umbilical ligament which is attached to the apex of the bladder and extends along the posterior surface of the anterior abdominal wall . The median umbilical ligament lies between the medial umbilical ligaments which are the fibrous remnants of the umbilical arteries. Due to traction which is exerted by the kidneys during their ascent the orifices of the ureters move superolaterally and the ureters enter oblique through the base of the bladder. The distal ends of the mesonephric ducts in females degenerate. The caudal ends of mesonephric ducts become the ejaculatory ducts. The orifices of these ducts move close together and enter the prostatic part of the urethra. In infants and children the urinary bladder, is in the abdomen when it is empty. It begins to enter the greater pelvis at about 6 years of age. It enters lesser pelvis and become a pelvic organ at or even after puberty. Urachal Anomalies A- Urachal cysts Remnants of the epithelial lining of the urachus may give rise to cysts. These cysts can be observed in about one- third of cadavers, but are not usually detected on living persons unless they become infected. B- Urachal Sinus A remnant of the lumen usually persists in the inferior part of the urachus and in about 50 % of cases the lumen is continuous with the cavity of the bladder. The lumen in the superior part of the urachus may remain patent and form a urachal sinus that opens at the umbilicus. C- Urachal Fistula Very rarely the entire urachus remains patent and form a urachal fistula that allows urine to escape from its umbilical orifice. 11 weeks 12 weeks 14 weeks Development of Urethra The epithelium of most of the male urethra is derived from the endoderm of the phallic part of the urogenital sinus. The epithelium of the terminal part of the urethra is derived from the surface ectoderm. The entire female urethra is derived from endoderm of the urogenital sinus. The distal part of the spongy urethra in the glans of the penis is derived from a solid cord of ectodermal cells which is derived from the surface ectoderm. These cells grow from the tip of the glans and joins the rest of the spongy urethra. So, the spongy urethra has a dual origin. The connective tissue and smooth muscle of the urethra in both sexes are derived from splanchnic mesenchyme. Development of the Suprarenal Gland The medulla develops from neural crest cells that derived from an adjacent sympathetic ganglion. The cortex develops from mesodermal cells which is first indicated during the 6th weeks by an aggregation of mesenchymal cells on each side, between the root of the dorsal mesentery and the developing gonad. Initially the neural crest cells form a mass on the medial side of the fetal cortex. As they are surrounded by the fetal cortex, these cells differentiate into the secretory cells of the medulla. Later more mesenchymal cells arise from the mesothelium lining the posterior abdominal wall and enclose the fetal cortex. These cells give rise to the permanent cortex. Differentiation of the suprarenal cortical zones begin during the late fetal period. The zona glomerulosa and the zona fasciculata are pressent at birth, but the zona reticularis is not recognizable until the end of the third year. 54 days 28 weeks The suprarenal glands of the human fetus are 10 to 20 times larger than the adult glands relative to body weight and are large compared with the kidneys. These large glands result from the extensive size of the fetal cortex. The medulla remains relatively small until after birth. The suprarenal glands rapidly become smaller as the fetal cortex regresses during the first year. The glands lose about one- third to about half of their weight during the first 2 or 3 weeks after birth and do not regain their original weight until the end of the 2nd year.