* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download regan-gas-10jul2010
Survey
Document related concepts
Transcript
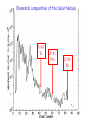
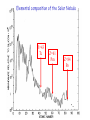
Making Gold : Nuclear Alchemy Prof. Paddy Regan Department of Physics University of Surrey Guildford, GU2 7XH [email protected] Medieval alchemist…trying to turn base metals into gold… Mistake…to try and use ‘chemistry’…needed nuclear physics Atoms (‘indivisible’) …… ~10-10 m, electrons (and their orbital structure) determine chemistry of the elements, e.g., NaCl Nuclei…..~10-14m across, protons determine the chemical element (Z); neutron number (N) determines the mass, (A = N+Z). > 99.9 % of the mass of the atom is in the nucleus. Nucleons (protons and neutrons ~10-15m) have a substructure, three quarks in each nucleon (‘ups’ and ‘downs’)…but they don’t exists on their own. Elemental composition of the Solar Nebula Z=43 Tc Z=61 Pm Z=84 Po What makes a nucleus ‘stable’? • There is an ongoing interplay and competition between coulomb repulsion and strong nuclear force interactions • The result is that only certain combinations of Z and N give rise to stable configurations (about 300 in total). • Other non-stable types can ‘radioactively decay’ (about 7,000 predicted). Radioactive species in the body Isotope Average amount by weight Activity U-Uranium 90μg 1.1Bq Th-Thorium 30 μg 0.11Bq 40K 17mg 4.4 kBq Ra 31pg 1.1Bq 14C 22ng 3.7kBq 3H-tritium 0.06pg 23Bq Po-Polonium 0.2pg 37Bq Some variation- for example smokers have 4-5 times more Po. 10 Mendeleyev Moseley’s Law….evidence for Atomic numbers…. ‘Characteristic’ X-rays…with a chemical (Z) dependence X-rays come from atomic ‘vacancies’ i.e. holes in the electron shells around the atom. Quantum mechanics means that the electron orbits are fixed in energy…. X-rays come from an electron ‘dropping’ from one energy level to a lower one X-rays come from atomic ‘vacancies’ i.e. holes in the electron shells around the atom. Quantum mechanics means that the electron orbits are fixed in energy…. X-rays come from an electron ‘dropping’ from one energy level to a lower one X-rays come from atomic ‘vacancies’ i.e. holes in the electron shells around the atom. Quantum mechanics means that the electron orbits are fixed in energy…. X-rays come from an electron ‘dropping’ from one energy level to a lower one X-ray emitted Spectral Maps of the Galaxy Ref http://adc.gsfc.nasa.gov/mw/mmw_images.html Radioactive 26Al around the Galaxy…. Diehl et al., Astron. Astrophys 97, 181 (1993) Full-sky Comptel map of 1.8 MeV gamma rays in 26Mg following 26Al b-decay. Elemental composition of the Solar Nebula Z=43 Tc Z=61 Pm Z=84 Po How it all starts….Hydrogen (Z=1) to Helium (Z=2) The Proton-Proton Chain Nuclear Fusion creates energy up to A~56 (Z=26 = Iron) If the star is hot enough, nuclear fusion will fuel the star and create elements up to A~56 Once you have 4He what next? • 4He + H→ 5Li not energetically allowed…neither is 4He+4He → 8Be…we’re stuck with A=1,2,3 and 4…. • BUT!!! 4He can fuse with 2 other 4He (8Be) stuck together for a short time (~10-16sec) to make 12C…complicated but understood. • Once we have made 12C (Z=6) nuclei can fuse together and gain energy (if the star is hot and massive enough) to make all elements up to Z=26 (Iron=Fe). DB ~1.5 MeV per A Once you have 56Fe what next? • Top of the binding energy per nucleon curve reached at A~56…fusion above this costs energy…bad news for the star supernova • BUT elements from 27-92 exist in nature – how are these made ? • Neutron Capture – neutrons have no charge – no electrostatic repulsion. A=N+Z = fixed Example of a mass parabola 125In 125Ba : Z=56; N=69 : Z=49 p→ n→ A=N+Z=125 p + b- + n 125Sn n + b+ + n 125Cs : Z=50; 125Xe Mass energy (mc2) : Z=55; N=70 125Sb : Z=51; 125Te 125I : Z=52; N=73 STABLE ISOBAR FOR A=125 : Z=54; N=71 : Z=53; N=72 Beta –radioactive decay, (consequence of E=mc2) 2 types: (i) Beta- plus proton changes to a neutron (Z ->Z-1) (ii) Beta – minus neutron changes to a proton (Z -> Z+1) SN1987a before and after !! Mass Parabolas and Radioactive Decays Super Heavies For a give fixed A (isobar), we have Fewer than 300 nuclei different combinations of Z and N. e.g., A=Z+N=125 can be from Proton Drip Line Z=52, N=73 →125Te73 ; or Z=51, N=74 → 125Sb74… A = constant Neutron Drip Line 36 The Natural Decay Chain for 238U Qa(210Pb) = 5.41 MeV Ea = 5.30 MeV E(206Pb) = 0.11 MeV T1/2 = 138 days. ‘Radium’ ‘218At =Radium B’ 210Po Radon =‘Emanation’ =Radium ‘F’ C’ E D C C’’ ‘218Po =Radium A’ Figure Wiescher, Regan & Aprahamian, Physics World Feb. 2002, page 33-38 Slow-neutron capture process allows formation of elements from A~56 to A=209 (Bi)... terminates at 209Bi...why? Neutron capture… no electrostatic barrier to nuclear fusion…. all you need are enough Neutrons… Nuclear reactions in Red giant stars create ‘spare’ neutrons Stellar neutron sources in the middle of Red Giant stars (e.g., Betelguese) 13C+4He→17O*→16O +n 22Ne+4He →26Mg*→25Mg + n So, how do you make Gold ? • Gold has 79 protons (i.e. Z=79) • Start with Z=78 protons (i.e. Platinum) • Specifically 196Pt ( Pt = Z=78, N=196-78=118) • Reaction is 196Pt + neutron to make 197Pt • 197Pt is radioactive and ‘beta-decays’ to make 197Au (i.e., normal ‘stable’ gold). S-process makes 209Bi from 208Pb+n → 209Pb (T1/2=3.2hr) → 209Bi Neutron capture on stable 210Po 209Bi → 210Bi (T1/2 =5 days) → 210Po. → a + 206Pb (stable nucleus, as is 207Pb and 208Pb). Summary What’s made where and how. – Hydrogen to Helium (in the sun, p-p chain CNO cycles). – Helium to Carbon, triple-alpha process, special fusion. – Carbon to iron: nuclear fusion reactions, if hot enough. – Above 56Fe, • (a) up to Z=92, 238U, supernova, rapid neutron captures...also spits out material for future neutron capture in 2nd / 3rd generation star • (b) can get up to 209Bi (210Po end-point) by slow neutron capture