* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download PPT
Gastroenteritis wikipedia , lookup
Globalization and disease wikipedia , lookup
Urinary tract infection wikipedia , lookup
Childhood immunizations in the United States wikipedia , lookup
Common cold wikipedia , lookup
Hygiene hypothesis wikipedia , lookup
Schistosomiasis wikipedia , lookup
Hepatitis B wikipedia , lookup
Eradication of infectious diseases wikipedia , lookup
Marburg virus disease wikipedia , lookup
Hepatitis C wikipedia , lookup
Sociality and disease transmission wikipedia , lookup
Transmission (medicine) wikipedia , lookup
Neonatal infection wikipedia , lookup
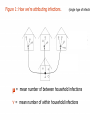
Preparedness for an Emerging Infection Niels G Becker National Centre for Epidemiology and Population Health Australian National University This presentation outlines the modelling and results in: Becker NG, Glass K, Li Z, Aldis G (2005). Controlling emerging infectious diseases like SARS. Mathematical Biosciences 193, 205-221 1 We have no vaccine to control an emerging infection, like SARS. It is necessary to resort to basic control measures such as • Isolating cases following diagnosis • Quarantining households with cases • Quarantining traced contacts of cases • Providing advice on how to reduce exposure • Closing schools How well do such measures work? How can models be used to assess the measures? 2 Structure of the model A household structure is needed because we want to assess the effect of quarantining affected households. Two types of individual are needed because we want to assess the effect of closing schools: Type 1 = school attendees, and Type 2 = others The effect of intervention is assessed by their reduction of R. Reduce the number of types of infective that we need to keep track of by attributing infections as follows: Attribute to an infective A the individuals she infects in other households AND all infections that arise in those household outbreaks. Do not attribute infections to A any infectives she infects within her own household. 3 Figure 1: How we’re attributing infections. 4 (single type of infectiv = mean number of between household infections ν= mean number of within household infections By attributing infections in this way the household structure does not add new types of infective. Our two types are: Type 1 = school attendees, and 1 The mean matrix is 1 2 Type 2 = others 2 M 11 M 12 M 21 M 22 The effective reproduction number is R 5 1 M 11 M 22 (M 11 M 22 )2 4M 12M 21 2 For specifying details of the model it is useful to separate out the between and within household transmission components. Between-household transmission ij = mean number type-j individuals infected by an infective of type i. Within-household transmission vij = mean number type-j cases in a household outbreak arising when a randomly selected type-i individual is infected. Note that M11= 11ν11 + 12ν21 , M12= 11ν12 + 12ν22 Other terms similarly. 6 Therefore M 11 M 12 11 M 21 M 22 21 12 11 12 22 21 22 Between-household transmission fS = fraction of community-time spent at school i = proportion of individuals of Type i in the community reflects enhanced mixing at school 11 21 7 12 1 2 0 f S (1 f S ) 22 0 1 2 Within-household transmission Assume uniform mixing within households. This gives, for example, 12 n : ( ) 1 m h n1 ( )n 2 ( ) n1 ( ) n 2 ( ) 1 E(N 1 ) where = (n1,n2) is the type of household m = mean outbreak size in a household of type , with 1 primary infective h = proportion of households of type Use Reed-Frost transmission chain model 8 Aside Reed-Frost transmission chain model To illustrate, consider a household of size 4. θ = probability of avoiding infection by one infective The probability distribution for the chains of transmission is 9 Chain Size Probability 10 1 θ3 110 2 3θ2(1-θ).θ2 1110 3 3θ2(1-θ).2θ(1-θ). θ 112 4 3θ2(1-θ).(1-θ)2 1111 4 3θ2(1-θ).2θ(1-θ).(1-θ) 120 3 3θ (1-θ)2.θ2 121 4 3θ(1-θ)2.(1-θ2) 13 4 (1-θ)3 R-F assumption The probability of avoiding infection when exposed to two infectives is θ2 From this we find the probability distribution for the outbreak size to be Size Probability 1 θ3 2 3θ4(1-θ) 3 3θ3(1-θ)2.(1+2θ) 4 balance of probability What is ν? Similarly for other household sizes. (End of Aside) Now let’s assign parameter values. Begin with the demographic quantities which we determine from Australian census data. 10 Proportion of household type (h x 1000) for Australia Household size (n) Number of school children (n1) 0 2 3 4 5 236 2 317 18 3 110 42 14 0 4 51 43 61 5 0 5 13 15 22 24 1 0 6 0 1 = 0.18 Var(N1) = 0.75 4 2 = 0.82 6 h(0,1) = 0.0236 1 6 11 1 0 4 For example: 0 7 7 h(2,2) = 0.0061 0 E(N1) = 0.46 Var(N2) = 0.96 E(N2) = 2.12 Cov(N1,N2) = 0.05 Assigning other parameter values for a plausible scenario RH0 = 6 θ = 0.8 = 2.63 fS = 0.4 =3 θ = 0.2 = 1.38 (from school hours in Australia) (from forces of infection estimated for measles) Next we model the effect of the interventions. 12 The effect of promoting reduced exposure Exposure is reduced by • wearing a mask • making fewer close contacts • reducing hand-to-mouth contacts • washing hands more regularly This makes individuals less susceptible and less infectious Model this by: Between households: * = S I (= 2 for simplicity) Within households: log(θ*) = S I log(θ) (= 2 log(θ) for simplicity) 13 The effect of isolating each case at diagnosis This reduces the infectious period Mathematically this is the same as reduction infectivity That is, Between households: * = Within households: 14 log(θ*) = log(θ) or θ* = θ Figure 2a (low infection rate within households, θ = 0.8) Figure 2b (high infection rate within households, θ = 0.2) Effective reproduction number for different levels of control : reducing susceptibility by a factor reducing infectivity by a factor reducing infectious period by a proportion Compare graphs corresponding to the following controls: Lower exposure outside the household only Lower exposure within and outside of the household Isolating cases when diagnosed Lower exposure and isolation 15 (all of the above) Figure 2a (θ = 0.8) Reduced suscy & infty: community and household Reduced suscy & infty: community only Isolating cases All controls 16 Figure 2b (θ = 0.2) Reduced suscy & infty: community and household Reduced suscy & infty: community only Isolating cases All controls 17 The effect of closing schools Recall that 11 21 12 1 2 0 f S (1 f S ) 22 0 1 2 Relatively more transmission occurs at school when > 1 We assess the effect of closing schools by comparing R for the case of fS = 0 relative to fS = 0.4 18 Figure 2c (low infection rate within households, θ = 0.8) Figure 2d (high infection rate within households, θ = 0.2) The effect of closing schools Effective reproduction number for different levels of control : reducing susceptibility by a factor reducing infectivity by a factor reducing infectious period by a proportion Compare graphs corresponding to the following controls: Lower exposure outside the household only Lower exposure within and outside of the household Isolating cases when diagnosed 19 Lower exposure and isolation Figure 2c (θ = 0.8) Reduced suscy & infty: community and household Reduced suscy & infty: community only Isolating cases All controls 20 Figure 2d (θ=0.2) Reduced suscy & infty: community and household Reduced suscy & infty: community only Isolating cases All controls 21 The effect of quarantining affected households Quarantining is a more stringent control • It isolates potentially infected individuals, without waiting for their diagnosis • Usually some quarantined individuals are not infected When the time from infection until isolation is larger than twice the latent period, then quarantining households can prevent secondary household cases from infecting individuals from other households. Primary case latent Primary case infected 22 If primary case isolated here Earliest time a secondary case can be infected If primary case isolated here The effect of quarantining traced contacts Household members are considered to be traced contacts. As well, a fraction of contacts outside the community is identified and quarantined. Calculations require assumptions about the duration from infection until (i) infectious, (ii) diagnosed and (iii) recovered. Calculating the exact reduction in R is much more difficult. We make a simplifying assumption. For traced primary cases who were infected assume that they were infected as soon as their “source” became infectious. This leads to a conservative estimate of the reduction in R achieved. 23 Choice of parameters (motivated by SARS) • The incubation period is 6.5 days • The latent period is 6.5 days. • The infectious period is 9 days. Figure 3a (low infection rate within households, θ = 0.8) Figure 3b (high infection rate within households, θ = 0.2) Compare graphs corresponding to the following control measures: • Isolating cases only (for reference) • Quarantining entire household at first diagnosis • Quarantining household and 50% of other contacts 24 Black arrows indicate where R = 1 Figure 3a (θ=0.8) Quarantining households Isolating cases only Quarantining households and 50% of traced contacts 25 Figure 3b (θ=0.2) Isolating cases only Quarantining households Quarantining households and 50% of traced contacts 26 Future work: Application of these ideas to help preparedness for pandemic influenza • Contribution of antivirals towards containment of an outbreak initiated by one imported case • How much are antivirals likely to delay the start of a major outbreak? • Use of antivirals in maintaining the health care service / other essential services. • How many courses of antivirals are needed? • How do we estimate the efficacy of antivirals? The End 27