* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Atoms, Elements, and Minerals Chapter 2
Survey
Document related concepts
Transcript
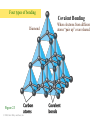
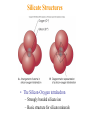
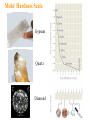
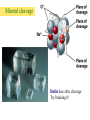
Atoms, Elements, and Minerals Chapter 2 Lauterbrunnen valley Switzerland Bryce Canyon Utah Rocks in the Earth's crust and mantle are made up of mineral assemblages with chemical compounds, elements, molecular bonds which are formed from ordered atomic structures. Where would you rather rock climb ? Why ? Atoms and Elements An element is a substance that can not be broken down into others by ordinary chemical reactions An atom is a chemical unit that cannot be broken down by chemical means composed of: • Protons (positively charged) • Neutrons (zero net charge) • Electrons (negatively charged) A molecule is made of 2 or more atoms bonded together. Atomic Structure • Protons and neutrons form the nucleus – Represents tiny fraction of the volume at the center of an atom, but nearly all of the mass • Electrons orbit the nucleus in discrete shells or energy levels – Shells represent nearly all of the volume of an atom, but only a tiny fraction of the mass – Numbers of electrons and protons are equal in a neutral atom – Chemical reactions involve outer shell (valence) electrons Chemical Bonding Chemical bonding is controlled by outermost shell (valence) electrons. Vacancies allow reactions. • Ionic bonding – Involves transfer of valence electrons from one atom to another • Covalent bonding – Involves sharing of valence electrons among adjacent atoms • Metallic bonding – Electrons flow freely throughout metals; results in high electrical conductivity Ionic bonding of NaCl (sodium chloride) Four types of bonding Ionic bonding Halite Figure 2.3 © 2008, John Wiley and Sons, Inc. When one atom transfers an electron to another. Four types of bonding Ionic bonding Halite Figure 2.3 © 2008, John Wiley and Sons, Inc. When one atom transfers an electron to another. Halite © 2008, John Wiley and Sons, Inc. Four types of bonding Covalent Bonding Diamond Figure 2.3 © 2008, John Wiley and Sons, Inc. When electrons from different atoms “pair up” or are shared. Diamonds: made of carbon atoms connected by covalent bonds © 2008, John Wiley and Sons, Inc. Four types of bonding Gold (Au) Metallic Bonding In metals, atoms are so tightly packed that electrons can be shared among several atoms. Here each atom is in contact with 12 other gold atoms. Outermost electrons are loosely held and drift easily – allowing for high heat and electrical conductivity. Gold This nugget was embedded in a rock, but weathering and erosion removed most of the rock. (Discovery in southern California. Four types of bonding Graphite Van der Waals Bonds A weak attraction that occurs between neutral molecules that have asymetrica charge distribution. (The positive end of one molecule is attracted to the negative end of another) Graphite © 2008, John Wiley and Sons, Inc. Covalent Bonds in Diamonds and Graphite Graphite Diamonds -Diamond and graphite are both made of carbon (C), but one is the hardest substance on Earth and the other very soft. Composition of Earth’s Crust • Common elements – Most common elements in Earth's crust • O, Si, Al, Fe, Ca, Na, K, Mg • Common mineral types – Most minerals are silicates (contain Si and O) Silicate Structures • The Silicon-Oxygen tetrahedron – Strongly bonded silicate ion – Basic structure for silicate minerals Silicate links Silicate minerals consist of (SiO4)4anionic groupings linked together. © 2008, John Wiley and Sons, Inc. Atomic Structures Activity - Organize into groups (10 or less) - Write everyone's name on one piece of paper - Examine your bag of molecules - When you are told “GO”: – Assemble each molecule below – The first group to CORRECTLY assemble All 3 molecules wins! - Turn in the group sheets (Hint: remember that molecules are 3-dimensional) The molecules are........ 1. Water 2. Table salt 3. Silica tetrahedra Examples of Minerals Halite (NaCl) -Table salt Gold (Au) Olivine (MgSiO4) -Minerals have a chemical formula. What is a Mineral ? A mineral is: -naturally formed, -inorganic substance, -solid, -crystalline, -chemically distinct Where do Minerals Form ? • • • • Geosphere (most minerals) Hydrosphere (e.g., halite) Biosphere (e.g., calcite) Atmosphere (water ice, snow) foraminifera © 2008, John Wiley and Sons, Inc. The Mineral Ice Ice occurs in nature and has a specific chemical formula (H2O). © 2008, John Wiley and Sons, Inc. Is water a mineral? Is this crocodile bone a mineral? How about this crocodile skull which has been fossilized? © 2008, John Wiley and Sons, Inc. Would you call coal a mineral? Why or why not? Steel in a processing plant. Is steel a mineral? © 2008, John Wiley and Sons, Inc. How about quartz? © 2008, John Wiley and Sons, Inc. Mineral Properties Physical and chemical properties of minerals are closely linked to their atomic structures and compositions • Hardness - Scratch-resistance • Cleavage – Breakage planes • Streak – Color left behind when mineral is scraped on unglazed porcelain • Luster – Manner in which light reflects off surface of a mineral • Color – Visible hue of a mineral ? Mystery Mineral ? Geologists have several “low tech” methods to identify unknown minerals in the field... © 2008, John Wiley and Sons, Inc. Mohs' Hardness Scale Gypsum Quartz Diamond Crystal Faces and Angles Crystal faces can have different surface area but will retain angle between surfaces. © 2008, the John Wiley and Sons, Inc. Crystal Faces and Angles Asbestos fibers have distinct growth habits. Crystals grow in long, cotton-like threads. Can be woven into fireproof fabric, also difficult to Mineral cleavage is different from crystal growth ! Cleavage: - Planar surfaces left from a freshly broken surface - Rocks break along defects or planes of weakness Mineral cleavage Halite has cubic cleavage Try breaking it! Color Streak Hematite is a grayish silver mineral but produces a red streak (high iron content) © 2008, John Wiley and Sons, Inc. Mineral Luster Luster is how light reflects off a surface Vitreous Quartz (SiO2) Resinous Sphalerite (ZnS) Pearly Talc (MgSi4O10(OH2)) What type of luster do you see in the mystery mineral ? Our mystery mineral has a metallic luster. Color Only can be Deceiving Corundum (Al2O3) has different colors: Ruby: red from substitution of Cr3 for Al3 High iron and titanium content © 2008, John Wiley and Sons,Sapphire: Inc. Magnetite Characteristics