* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Anterior Intercostal Artery Perforator Flap Autologous Augmentation
Survey
Document related concepts
Transcript
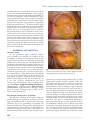
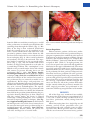
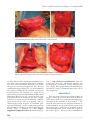
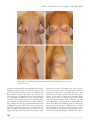
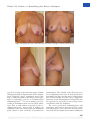
COSMETIC Anterior Intercostal Artery Perforator Flap Autologous Augmentation in Bariatric Mastopexy Paolo Persichetti, M.D., Ph.D. Stefania Tenna, M.D., Ph.D. Beniamino Brunetti, M.D. Achille Aveta, M.D. Francesco Segreto, M.D. Giovanni Francesco Marangi, M.D. Rome, Italy Background: Breast reshaping after massive weight loss represents a challenging procedure because of severe hypoplasia and tissue ptosis. Standard mastopexy techniques are often inadequate to restore a pleasant profile and volume. The authors present their experience with the anterior intercostal artery perforator flap in breast autologous augmentation and remodeling. Methods: Fifteen bariatric patients (30 breasts) affected by severe breast ptosis and tissue laxity in the upper abdominal wall underwent superior pedicle mastopexy with anterior intercostal artery perforator flap autologous augmentation. The flap was harvested including soft tissues above and below the inframammary fold, extending cranially 5 to 6 cm above the fold and inferiorly over the costal cage and hypochondrium. The flap was completely islanded on intercostal perforators originating from the fifth to seventh intercostal spaces. It was cranially advanced and sutured to the pectoralis major fascia. The medial and lateral borders were sutured together to shape an “autologous implant.” Results: All of the flaps were transferred successfully. The donor site was always closed primarily and upper abdominal laxity corrected. All of the breasts presented soft at palpation, with no clinical signs of flap necrosis early or late postoperatively. At 1-year follow-up, the breasts maintained good shape and projection. Conclusions: The anterior intercostal artery perforator flap proved to be a reliable option in bariatric mastopexy. The technique can be performed easily and allows the harvesting of a large amount of tissue with a wide range of motion, providing adequate breast volume and projection without the need for implant placement. (Plast. Reconstr. Surg. 130: 917, 2012.) CLINICAL QUESTION/LEVEL OF EVIDENCE: Therapeutic, IV. T he number of bariatric surgery procedures has increased constantly over the past decade.1 Although they may solve the excess weight and many of its associated comorbidities, the patient is left with excess skin and soft tissue, often leading to referral to the plastic surgeon. The breast profile may be severely distorted as a consequence of massive weight loss, with upper pole deficiency, high-grade ptosis, and lowered inframammary fold. In these patients, mastopexy is challenging, traditional techniques are often inadequate because of poor quality of tissues, and restoration of breast volume is a main issue for a pleasant morphology. The thin overstretched dermis and the poor subcutaneous tissue make implant placement alone unadvisable. Colwell and From the Plastic and Reconstructive Surgery Unit, Campus Bio-Medico di Roma University. Received for publication January 25, 2012; accepted April 19, 2012. Copyright ©2012 by the American Society of Plastic Surgeons DOI: 10.1097/PRS.0b013e318262f38a colleagues2 already described the use of cadaveric dermis (AlloDerm; LifeCell Corp., Branchburg, N.J.) to supplement suspension of the breast and the implant in an anatomical position, but the ideal volume restoration in this scenario is repre- Disclosure: The authors have no financial interest or commercial association with the subject matter or products mentioned in this article. No funding was received for this work. Supplemental digital content is available for this article. Direct URL citations appear in the text; simply type the URL address into any Web browser to access this content. Clickable links to the material are provided in the HTML text of this article on the Journal’s Web site (www.PRSJournal.com). www.PRSJournal.com 917 Plastic and Reconstructive Surgery • October 2012 sented by the use of autologous tissues. Some flaps such as the rotation-advancement of the superomedial pedicle described by Losken and Holtz3 or the use of composite tissues nourished by intercostal perforators have been proposed. Starting from Kerrigan and Daniel,4 several studies investigated the anatomy of perforators originating from intercostal arteries.5,6 Pedicled and free flaps nourished by these vessels have been described for hand,5 breast, and trunk reconstruction.7 In particular, the use of intercostal artery perforator flaps nourished by lateral branches of the intercostal system (lateral intercostal artery perforator flap) has been previously reported in postbariatric patients. The authors report their experience with the anterior intercostal artery perforator flap, based on anterior intercostal perforator branches originating from the fifth to seventh intercostal spaces, in the field of postbariatric autologous augmentation mastopexy. MATERIALS AND METHODS Cadaveric Study In our preclinical phase, cadaveric studies were performed to evaluate the viability of the technique, focusing on localization of the perforators and range of motion of the flap. A large skin paddle above and below the inframammary fold was dissected to identify the number and course of the main perforators emerging from the pectoralis major, serratus anterior, and rectus abdominis muscle fascia. On average, two or three musculocutaneous perforators for each intercostal space were found (Fig. 1). Flap dimensions ranged from 6 to 12 cm in width and 6 to 20 cm in length, depending on upper abdomen laxity. Range of motion of the flap was also assessed and was 3 to 5 cm. The greatest advancement has been achieved by sacrificing the most caudal perforators. [See Figure, Supplemental Digital Content 1, which shows a cadaveric study, http://links.lww. com/PRS/A539. (Left) Mobility of the flap. (Right) Increased range of motion after sacrifice of the most caudal perforators.] Flap Design and Operative Technique Preoperative marking of inverted-T superior pedicle mastopexy8 is planned. A pinch test of the upper abdominal wall, in supine and orthostatic positions, has to be performed to quantify the amount of skin laxity. The position and number of cutaneous intercostal perforators is identified by a hand-held Doppler probe above and below the inframammary fold, and then the anterior inter- 918 Fig. 1. Cadaveric study. (Above) Two or three perforators for each intercostal space have been isolated. (Below) Perforators emerging from the pectoralis major muscle fascia. costal artery perforator flap is drawn (Fig. 2). Flap dimensions usually extend cranially until the lower margin of the areola (at least 5 to 6 cm above the fold), and inferiorly 5 to 10 cm below the inframammary fold, according to upper abdominal skin laxity that needs to be corrected and always checking manually for feasibility of primary closure. Flap harvesting is performed in supine position using 2.5⫻ loupe magnification: the superior portion of the flap is first dissected craniocaudally along and pectoralis major fascia; then, inferiorly, the flap is undermined following a plane superficial to the serratus anterior and rectus abdominis muscles fasciae. Major intercostal perforators emerging through the pectoralis major, serratus anterior, and rectus abdominis muscles are isolated. The anterior intercostal artery perforator flap is islanded on perforator vessels Volume 130, Number 4 • Remodeling after Bariatric Mastopexy Fig. 2. Preoperative markings after Doppler mapping. from the fifth to seventh intercostal spaces, and all of the perforators are initially spared without skeletonizing them through the muscle (Fig. 3). Mobility of the flap is then evaluated. Perforators from the seventh space can be sacrificed to enhance cranial advancement and avoid their compression by primary closure of the donor site. Minor lateral vessels may also be cut to obtain a better breast reshaping (Fig. 4). After careful evaluation of vascularity, the flap is decorticated. The superior border is anchored to the pectoralis major fascia (Fig. 5, left), and the medial and lateral borders are flipped and sutured to each other in a round shape fashion. The “autoimplant” is stabilized to the pectoralis major fascia with absorbable sutures to prevent shearing forces on the perforators (Fig. 5, right). (See Figure, Supplemental Digital Content 2, which shows the surgical technique, http://links.lww.com/PRS/A540. Medial and lateral borders of the anterior intercostal artery perforator flap are sutured to model an autoprosthesis.) Breast shape and projection are checked and abdominal subcutaneous tissue is undermined to achieve primary closure of the donor site, as in a reverse abdominoplasty. The superficialis fascia must be fixed to rib periosteum with nonabsorbable stitches to redefine the inframammary fold in the position planned during the preoperative drawing. Mastopexy is then completed with the patient sitting: the breast is sutured around the “autologous implant” as in a standard inverted-T superior pedicle technique. (See Figure, Supplemental Digital Content 3, which shows an intraoperative view of the breast sutured over the anterior intercostal artery perforator flap in a standard inverted-T technique, http://links.lww. com/PRS/A541.) Suction drains are placed for each side and kept for at least 48 hours. Fig. 3. Anterior intercostal perforators. Adequate mobility of the flap is achieved without skeletonizing perforators through the muscle. Patient Population Fifteen bariatric patients (30 breasts) underwent anterior intercostal artery perforator flap autologous augmentation combined with inverted T-mastopexy at the Department of Plastic, Reconstructive, and Aesthetic Surgery of “Campus BioMedico di Rome” University from March of 2010 to April of 2011 (Table 1). In eight patients, the severe breast ptosis was associated with significant skin laxity in the upper abdominal wall. The mean age of the patients was 40.3 years and the average body mass index was 32.7. Sleeve gastrectomy had been performed in four patients, biliopancreatic diversion had been performed in nine patients, transoral gastroplasty had been performed in one patient, and gastric bypass had been performed in one patient. The average weight loss was 45 kg, and the average suprasternal notch–to–nipple distance was 28.9 cm. All of the patients had stable weight control within the previous 6 months. No other surgical procedure was performed at the same time. Follow-up ranged from 6 to 12 months. RESULTS All of the flaps were transferred successfully. The mean size of the flaps was approximately 8.8 ⫻ 12.9 cm. Usually the flap was harvested on at least three perforators, mainly arising from the fifth space. Mean harvesting time for a single flap was 40 minutes, and all of the procedures were performed bilaterally with a double team to reduce operative time. Mean operative time was 3 hours, and the use of anterior intercostal artery perfora- 919 Plastic and Reconstructive Surgery • October 2012 Fig. 4. (Left) Anterior intercostal artery perforator flap completely islanded and decorticated. (Right) Increased mobility of the flap after sacrifice of the minor caudal perforators. Fig. 5. (Left) “Key stitches” to the pectoralis major fascia. (Right) An “autoprosthesis” is shaped and stabilized to the pectoralis major fascia to prevent shearing forces on the perforators. tor flaps did not add a significant amount of extra time to the classic mastopexy procedure. All donor sites were closed primarily. Patients were all discharged on the first postoperative day. Physical examination was performed 1, 3, 6, and 12 months after surgery. Within the first month, one patient needed vertical scar revision of a single breast and another presented inframammary fold caudal dislocation that had to be corrected with resuspension under local anesthesia. No other complications were registered. In all follow-up examinations, breasts were soft at palpation, with no clinical signs of flap necrosis. At 12-month postoperative follow-up, a stable result with good nipple position, breast shape, and projection was registered (Figs. 6 through 8). [See Figure, Supplemental Digital Content 4, which shows pa- 920 tient 7, http://links.lww.com/PRS/A542. (Left) Intraoperative view: the “autologous implant” has been shaped and sutured. (Center) Preoperative view. (Right) Photograph obtained 6 months postoperatively.] Upper abdominal skin laxity was always improved. DISCUSSION The use of the intercostal vascular pedicle to supply a skin flap was first described by Esser in 1931.9 Since then, several anatomical studies have investigated the anatomy of these vessels.5,6 The anterior intercostal arteries originate from the internal mammary artery from the first to the sixth intercostal spaces and from its terminal branch, the musculophrenic artery, from the seventh to the ninth spaces. These arteries communicate Volume 130, Number 4 • Remodeling after Bariatric Mastopexy Table 1. Patients Who Underwent Anterior Intercostal Artery Perforator Flap Autologous Augmentation Combined with Inverted-T Mastopexy Patient Age (yr) Bariatric Procedure Suprasternal Notch–to–Nipple Distance (cm) 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 55 50 43 33 28 42 32 51 37 48 35 40 46 34 31 Biliopancreatic diversion Gastric bypass Sleeve gastrectomy Biliopancreatic diversion Transoral gastroplasty Biliopancreatic diversion Sleeve gastrectomy Sleeve gastrectomy Biliopancreatic diversion Biliopancreatic diversion Sleeve gastrectomy Biliopancreatic diversion Biliopancreatic diversion Biliopancreatic diversion Biliopancreatic diversion 34.5 Left, 30; right, 28.5 Left, 26; right, 27 Left, 28; right, 27.5 31 Left, 27; right, 27.5 28 Left, 26.5; right, 27.5 29 Left, 30.5; right, 30 30 Left, 30; right, 31.5 Left, 32; right, 31 28.5 Left, 27; right, 25.5 with the posterior intercostal arteries at approximately the medial third of the ribs, constituting a vascular arcade from which multiple perforators arise.7 The course of intercostal vessels can be divided horizontally into four segments: vertebral, intercostal, intermuscular, and rectal.4 Flaps are named by the segment from which their vascular supply arises. The pedicle of the dorsal intercostal artery perforator flap derives from the vertebral segment. The dorsal intercostal artery perforator flap has been used mainly as a pedicled flap for the closure of midline back defects.10 –12 The lateral intercostal perforator flap is based on vessels deriving from the costal segment. It was first described by Badran et al. in 198413 and has been used for correction of facial contour14 and for breast reconstruction.15–18 The use of this flap for autologous breast augmentation was first reported by Hurwitz and Agha-Mohammadi, who performed a “spiral flap.”19 Kwei et al.20 described its anatomical basis and, in addition to Hamdi and colleagues,21 reported their experience with the lateral intercostal artery perforator flap. In 2011, Akyurek published an article about the use of a pectoralis muscle sling to prevent the descent of the flap when used for autologous breast augmentation in bariatric patients.22 The lateral intercostal artery perforator flap has the advantages of improving breast fullness and correcting the axillary roll at the same time, but a long scar in the lateral chest wall results from the surgical procedure. Moreover, this flap has limited mobility because of its small arc of rotation and is nourished by few perforator vessels. In contrast, in the anterior chest, several perforating branches can be found from the fifth to eighth intercostal seg- Upper Abdominal Laxity Flap Dimension (cm) No. of Perforators Supplying the Flap ⫹⫹ 10 ⫻ 18 8⫻7 10 ⫻ 15 10 ⫻ 16 9 ⫻ 18 6 ⫻ 10 8⫻8 8 ⫻ 15 9 ⫻ 16 8 ⫻ 15 10 ⫻ 8 10 ⫻ 9 10 ⫻ 14 8 ⫻ 10 10 ⫻ 15 4 3 5 4 4 3 3 4 4 4 3 3 5 3 4 ⫹⫹ ⫹ ⫹ ⫹⫹ ⫹ ⫹⫹ ⫹ ments; these vessels extend horizontally along the rib cage and anastomose vertically with each other. Moreover, the vascular system deriving from these arteries shows anastomoses with the system belonging to the superior epigastric artery.5 The anterior intercostal artery perforator flap is supplied by vessels originating from the muscular or rectal segment of intercostal arteries7 and presents several advantages compared with the lateral intercostal artery perforator flap, such as the larger amount of harvestable tissue because of the greater number of perforators and superior mobility. The use of the anterior intercostal artery perforator flap has been described for coverage of epigastric abdominal wounds,23 for hand reconstruction,5 for correction of sequelae from augmentation mammaplasty24 and for reconstruction of sternal defects.7 Anterior intercostal perforators are also the vessels that nourish the inferior dermolipoglandular flap used in mastopexy procedures and first presented by Dr. Ribeiro at the Congress of the Brazilian Society of Plastic Surgery in 1971. This flap is vascularized by arteries that arise from the fourth to sixth intercostal spaces. In Dr. Ribeiro’s technique,25,26 it is harvested from the lower pole of the breast, then advanced cranially as a pedicled flap and sutured to the pectoralis major fascia to provide breast firmness and projection. Modifications of the classic Ribeiro technique to address massive weight loss patients have already been described in the literature. Daniel suggested the passage of the flap under a loop of pectoral muscle to improve longterm projection,27 whereas Ritz et al. reported their experience in passing the flap only under the pectoralis fascia.28 However, these procedures im- 921 Plastic and Reconstructive Surgery • October 2012 Fig. 6. Photographs of patient 3 obtained (left) preoperatively and (right) 6 months postoperatively. Notice the improvement in abdominal skin laxity, with transition from convexity to concavity (lateral view). ply increased morbidity and could harm oncologic diagnosis and treatment. Perforator flaps in the past decade have enhanced tissue recruitment and flap mobility, and in this sense, our anterior intercostal artery perforator flap can be considered an evolution of Ribeiro’s flap without many tradeoffs. Vessel dissection in fact provides superior mobility compared with the classic Ribeiro flap, without the need to perform skeletonization through the muscle. Even if the anterior intercostal artery perforator flap may be nourished by the fifth to eighth intercostal space perforators, our experience proved that safely harvesting a large 922 amount of tissue by islanding the flap on only two or three perforators arising from the fifth or sixth intercostal space is possible. However, the fifth intercostal space perforators should be preferred, as the more caudal ones are more difficult to spare when the flap is advanced cranially and the inframammary fold redefined. In this case, distal flap vascularization is maintained by the vertical network of linking vessels connecting adjacent intercostal spaces.5 Furthermore, minor lateral branches may be cut either to increase the flap’s mobility or to improve its reshaping in a sort of round autologous implant. Finally, the advan- Volume 130, Number 4 • Remodeling after Bariatric Mastopexy Fig. 7. Photographs of patient 1 obtained (left) preoperatively and (right) 12 months postoperatively. Fig. 8. Lateral preoperative (right) and postoperative (left) views of patient 1. tage of correcting at the same time upper abdominal lipodystrophy and ptosis must also be emphasized. Currently, upper abdominal tissue laxity may be addressed with several surgical strategies, such as anchor-line, reverse, or circumferential abdominoplasties.29,30 In our technique, the harvesting of abdominal tissue has two main advantages: first, it adds further projection to the reconstructed breast; and second, it induces an abdominal tissue lift, solving superior abdominal ptosis, which is often refractory to standard ab- dominoplasty. The viability of the flap has never been compromised because of many perforators nourishing this flap, and no major complications have been recorded. In some cases, a temporary distortion of the inframammary fold profile may be experienced, especially in cases of large tissue recruitment from the abdomen. We never performed abdominoplasty and mastopexy with anterior intercostal artery perforator flap surgery at the same time because of the increased risk of complications and the additional 923 Plastic and Reconstructive Surgery • October 2012 tension that would be placed on the inframammary fold. In planning the two procedures, we would perform abdominoplasty first and then, after 6 months, when the result was stable and the tension had been redistributed, breast reshaping. In our experience, the anterior intercostal artery perforator flap proved to be a safe and reliable option in bariatric mastopexy: it requires easy dissection and allows the harvesting of a large amount of tissue with a wide arc of rotation, thus overcoming the need for implant placement. Possible contraindications to this technique are large scars in the area of flap harvesting, such as from open cholecystectomy, or prior inferior pedicle breast reduction. CONCLUSIONS Autologous augmentation with the anterior intercostal artery perforator flap represents a valid option for breast reshaping in the massive weight loss population. The procedure can be easily standardized and is easy to perform, increases breast volume and projection, and corrects upper abdominal skin laxity usually not responsive to classic abdominoplasty procedures. Inverted-T mastopexy with the anterior intercostal artery perforator flap proved to be a very good technique for restoring breast shape and projection in postbariatric patients. Stefania Tenna, M.D., Ph.D. Plastic and Reconstructive Surgery Unit Campus Bio-Medico di Roma University Via Alvaro del Portillo 200-00128 Rome, Italy [email protected] 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. REFERENCES 1. Matarasso A, Roslin MS, Kurian M; Plastic Surgery Educational Foundation Technology Assessment Committee. Bariatric surgery: An overview of obesity surgery. Plast Reconstr Surg. 2007;119:1357–1362. 2. Colwell AS, Driscoll D, Breuing KH. Mastopexy techniques after massive weight loss: An algorithmic approach and review of the literature. Ann Plast Surg. 2009;63:28–33. 3. Losken A, Holtz DJ. Versatility of the superomedial pedicle in managing the massive weight loss breast: The rotationadvancement technique. Plast Reconstr Surg. 2007;120:1060– 1068. 4. Kerrigan CL, Daniel RK. The intercostal flap: An anatomical and hemodynamic approach. Ann Plast Surg. 1979;2:411– 421. 5. Oki K, Murakami M, Tanuma K, Ogawa R, Ozawa H, Hyakusoku H. Anatomical study of pectoral intercostal perforators and clinical study of the pectoral intercostal perforator flap for hand reconstruction. Plast Reconstr Surg. 2009;123:1789– 1800. 6. Saint-Cyr M, Schaverien M, Arbique G, Hatef D, Brown SA, Rohrich RJ. Three- and four-dimensional computed tomographic angiography and venography for the investigation of 924 21. 22. 23. 24. 25. 26. 27. the vascular anatomy and perfusion of perforator flaps. Plast Reconstr Surg. 2008;121:772–780. Hamdi M, Van Landuyt K, de Frene B, Roche N, Blondeel P, Monstrey S. The versatility of the inter-costal artery perforator (ICAP) flaps. J Plast Reconstr Aesthet Surg. 2006;59:644–652. Pitanguy I. Principles of reduction mammaplasty. In: Georgiade NG, Georgiade GS, Riefkohi R, eds. Aesthetic Surgery of the Breast. Philadelphia: Saunders; 1990:191. Esser J. Biological or Artery Flaps of the Face. Monaco: Institut Esser de Chirurgie Structive; 1931. Minabe T, Harii K. Dorsal intercostal artery perforator flap: Anatomical study and clinical applications. Plast Reconstr Surg. 2007;120:681–689. de Weerd L, Weum S. The sensate medial dorsal intercostal artery perforator flap for closure of cervicothoracic midline defects after spinal surgery: An anatomic study and case reports. Ann Plast Surg. 2009;63:418–421. Isik D, Tekes L, Eseoglu M, Isik Y, Bilici S, Atik B. Closure of large myelomeningocele defects using dorsal intercostal artery perforator flap. Ann Plast Surg. 2011;67:159–163. Badran HA, El-Helaly MS, Safe I. The lateral intercostal neurovascular free flap. Plast Reconstr Surg. 1984;73:17–26. Badran HA, Youssef MK, Shaker AA. Management of facial contour deformities with deepithelialized lateral intercostal free flap. Ann Plast Surg. 1996;37:94–101; discussion 101–105. Hamdi M, Van Landuyt K, Monstrey S, Blondeel P. Pedicled perforator flaps in breast reconstruction: A new concept. Br J Plast Surg. 2004;57:531–539. Holmström H, Lossing C. Lateral thoracodorsal flap: An intercostal perforator flap for breast reconstruction. Semin Plast Surg. 2002;16:53–59. Hamdi M, Spano A, Van Landuyt K, D’Herde K, Blondeel P, Monstrey S. The lateral intercostal artery perforators: Anatomical study and clinical application in breast surgery. Plast Reconstr Surg. 2008;121:389–396. Rubin JP. Mastopexy after massive weight loss: Dermal suspension and total parenchymal reshaping. Aesthet Surg J. 2006;26:214–222. Hurwitz DJ, Agha-Mohammadi S. Postbariatric surgery breast reshaping: The spiral flap. Ann Plast Surg. 2006;56:481–486; discussion 486. Kwei S, Borud LJ, Lee BT. Mastopexy with autologous augmentation after massive weight loss: The intercostal artery perforator (ICAP) flap. Ann Plast Surg. 2006;57:361–365. Hamdi M, Van Landuyt K, Blondeel P, Hijjawi JB, Roche N, Monstrey S. Autologous breast augmentation with the lateral intercostal artery perforator flap in massive weight loss patients. J Plast Reconstr Aesthet Surg. 2009;62:65–70. Akyurek M. Vertical mastopexy and lateral intercostal artery perforator (LICAP) flap with pectoralis muscle sling for autologous tissue breast augmentation in the bariatric patient. Ann Plast Surg. 2011;66:29–35. Hallock GG. The island anterior intercostal artery perforator flap as another option for the difficult epigastric abdominal wound. Ann Plast Surg. 2009;63:414–417. Juri J, Valotta F, Létiz MS. Submammary flap for correction of severe sequelae from augmentation mammaplasty. Plast Reconstr Surg. 2004;114:567–574. Ribeiro L. A new technique for reduction mammaplasty. Plast Reconstr Surg. 1975;55:330–334. Ribeiro L, Accorsi A Jr, Buss A, Marcal-Pessoa M. Creation and evolution of 30 years of the inferior pedicle in reduction mammaplasties. Plast Reconstr Surg. 2002;110:960–970. Daniel M. Mammaplasty with pectoral muscle flap. Paper presented at: 64th Annual Scientific Meeting of the Ameri- Volume 130, Number 4 • Remodeling after Bariatric Mastopexy can Society of Plastic and Reconstructive Surgeons; October 7–11, 1995; Montreal, Quebec, Canada. 28. Ritz M, Silfen R, Southwick G. Fascial suspension mastopexy. Plast Reconstr Surg. 2006;117:86–94. 29. Agha-Mohammadi S, Hurwitz DJ. Management of upper abdominal laxity after massive weight loss: Reverse abdomino- plasty and inframammary fold reconstruction. Aesthetic Plast Surg. 2010;34:226–231. 30. Persichetti P, Simone P, Scuderi N. Anchor-line abdominoplasty: A comprehensive approach to abdominal wall reconstruction and body contouring. Plast Reconstr Surg. 2005;116: 289–294. 925