* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download 5 µm
Survey
Document related concepts
Transcript
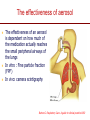
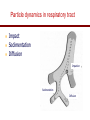
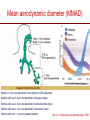
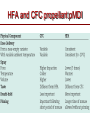
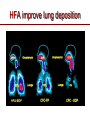
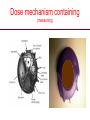
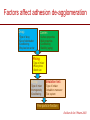
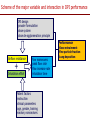
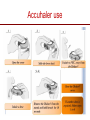
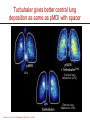
The important of inhalers device in asthma management? นายแพทย์ธีระศักดิ์ แก้วอมตวงศ์ หน่วยโรคระบบการหายใจและเวชบาบัดวิกฤต ภาควิชาอายุรศาสตร์ โรงพยาบาลรามาธิบดี Respiratory pharmacology Inhaled drug administrations are widely used in pulmonary medicine Asthma COPD Bronchiectasis Cystic fibrosis Drugs available for respiratory care Anti-inflammation: corticosteroids Anti-infective agents : antibiotics, antifungal Bronchodilators : β adrenergic agonist and muscarinic receptor antagonist Mucoregulator : Dornase alpha Medication for asthma and COPD Asthma COPD Anti-inflammatory drugs -Corticosteroids -Anti-leukotriene -Cromone -Theophylline Bronchodilators -Short and long acting β2-agonits -Short and long acting anticholinergic -Theophylline Bronchodilator -Short and long acting β2-agonits -Short acting anticholinergic Anti-inflammatory drugs -corticosteroid ICS/LABA combination ICS/LABA combination Anti-immunoglobulin E Mucolytic drugs Antibiotic s Vaccination Advantages of inhaled therapy Providing local effect of medications that optimizes the desired therapeutic effects Requiring the lower dose Preferred characteristics Fast onset of action Low systemic bioavailability Less side effects than orally or intravenously administered drugs American Association of Respiratory Care Aerosol Consensus Statement . Respir Care 1991 Pulmonary drug delivery Lewis RA, Fleming JS. Br J Dis Chest 1985; 79(4):361-367. Respiratory drugs Development of inhalers The effectiveness of aerosol The effectiveness of an aerosol is dependent on how much of the medication actually reaches the small peripheral airways of the lungs In vitro : Fine particle fraction (FPF) In vivo: camera scintigraphy Burton G. Respiratory Care. A guide to clinical practice 1992 Airway anatomy (tree) Wiebel Upper & lower respiratory tract Conducting & gas exchange Airway generation and flow relationship Lung deposition of drugs Factors affecting lung deposition Particle size Speed of inspiration (inspiratory flow) Integrity of airway Proper inhaled device technique Particle dynamics in respiratory tract Impact Sedimentation Diffusion Impaction Sedimentation Diffusion Physical mechanism of drug movement & deposition Speed of inspiration (Ideal speed or flow is 30-60 L/min) •High flow facilitate central impaction but low flow facilitate sedimentation of particle Diffusion (< 0.5 µm) high speed movement and short haul exhaled Sedimentation (0.5-5 µm) Impact (> 5 µm) at upper airway and high flow rate Fine-particle fraction (FPF) Fine-particle fraction (FPF) is percentage of the aerosol between 1–5 μm that deposits in the lung Mean aerodynamic diameter (MMAD) Deposit of particles by size Particles > 8 µm are deposited in the oropharynx (90% absorbed) Particles with size 5-8 µm are deposited in the large airways Particles with size 2-5 µm are deposited in tracheobronchial region Particles with size 1-2 µm are deposited in the alveolar region Particles with size < 1 µm are passed expiration Rau JL Jr. Respiratory care pharmacology. 2002 MMAD and GSD Mass Median Aerodynamic Diameter (MMAD) is defined as the diameter at which 50% of the particles by mass are larger and 50% are smaller Geometric Standard Deviation (GSD) is a measure of the spread of an aerodynamic particle size distribution. Typically calculated as follows: GSD = (d84/d16)1/2 d84 and d16 represent the diameters at which 84% and 16% of the aerosol mass are contained, respectively, in diameters less than these diameters. Particle size distribution (Histogram) Particle size distribution MMAD =1 µm Histogram of particle size distribution Histogram of logarithmic particle size distribution MMAD (d50) MMAD =1 µm MMAD =5 µm means ? The calculated aerodynamic diameter that divides the particles of an aerosol in half, based on the weight of the particles. By weight, 50% of the particles will be larger than the MMAD and 50% of the particles will be smaller than the MMAD. MMAD of 5 μm =? 50 % of the total sample mass will be present in particles having diameters less than 5 μm, and that 50 % of the total sample mass will be present in particles having an diameter larger than 5 μm. Lung deposition and MMAD Leach C et al. Particle size of inhaled corticosteroids: Does it matter? J Allergy Clin Immunol 2009 Inhaler devices Metered-dose inhaler (MDIs) Conventional pressurized inhaler Activated by pressurized inhaler inspiration Dry-powder inhaler (DPI) Single dose Multi-dose Nebulizers Jet Ultrasonic pMDI and plum mechanism HFA and CFC propellant pMDI CFC pMDI HFA pMDI HFA improve lung deposition MDI with spacing device or VHC การใช้ยาสู ดร่ วมกับ Spacer ชนิดของ spacer แบ่งเป็ น Aerosol Cloud Enhancer (ACE) Volumetric spacer Aerochamber (VHC) vs Ventahaler Aerochamber plus Ventahaler 1) a 145-mL rigid cylinder made of polyester (Trudell Medical, London, ON) 2) Adapter that makes it compatible with most pMDIs 3) Is available with a mouthpiece or a mask 1) An elliptical-shaped device made of rigid, transparent plastic 2) Capacity of 750 Ml 3) Designed to fit GlaxoSmithKline Products Not fit all pMDIs. Spacer decrease orapharyngeal deposition Build in dose counter MDI and spacer use Types of dry powder inhaler (DPI) Single dose dry powder (SD-DPI) Handihaler Breezhaler Multi-dose dry powder (MD-DPI) Accuhaler Turbuhaler Basic design & functional elements (DPI) Powder formulation Dose mechanism containing (measuring) Powder de-agglomeration principle (Dispersing powder into inhaled air stream) Inhaler mouthpiece Powder formulation Active drug particles with 1-5 µm are extremely adhesive Drug stick together or surface of inhaler Excipients (micronized or agglomerate) Adhesive mixture (α lactose monohydrate) The detach of active drug from carriers Powder formulation 100 µm Adhesive mixture 100 µm Nuclear conglomerate The carrier molecules of excipient 1) Similar size to drugs (micronized) 2) Large size than drug (carrier) 500 µm Spherical pellet type Micronized drug and carrier particles Active drug 3-5 µM Large carrier lactose particle 500 µM Active drug 3-5 µM Micronized lactose molecule Adhesive and removal force balance The Fine Particle Fraction As a result of the balance between separate force (from de-agglomeration) and adhesive force (drug-carrier interaction) Basic design & functional elements (DPI) Powder formulation Dose mechanism containing (measuring) Powder de-agglomeration principle (Dispersing powder into inhaled air stream) Inhaler mouthpiece Dose measuring system De-agglomeration principles DPI Multi-dose dried powder Dose mechanism containing (measuring) Dose mechanism containing (measuring) DPI Inhaler performance Inspiratory flow performance ‘Intrinsic resistance of device’ Patients inspiratory flow ability Humidity and moister exposure Inpiratory flow range of DPI Flow dependence DPI Turbuhaler Flow independent DPI Accuhaler Flow rate and FPF from inhalers Intrinsic resistance of DPI (kPa0.5/min/L) Inhalers and airflow resistance 120 Flow rate (L/min) 100 80 60 40 2.2 10-2 kPa1/2 L-1 min 2.7 10-2 kPa1/2 L-1 min 3.4 10-2 kPa1/2 L-1 min 5.1 10-2 kPa1/2 L-1 min Breezhaler Diskus Turbuhaler Handihaler 20 0 0 2 4 6 Inspiratory effort (kPa) 8 10 Singh D et al. ATS 2010 (poster) Factors affect adhesion de-agglomeration Drug: -Type of drug Carrier: -Surface properties -Bulk properties -Conditioning -Stability (aging) -Size of distribution -Conditioning -Play-load on carrier Mixing: -Type of mixer -Mixing time -Batch size Mixing: -Type of mixer -Homogeneity -Conditioning Inhalation test: -Type of inhaler -Inhalation manouvor -Test system Fine particle fraction De Boer Ah Int J Pharm 2003 Tubuhaler as flow dependent Necessary inspiratory flow rate (L/m) Drug deposition in lungs (%) Drug deposition in oropharyns 35 14.8 3.3 66.6 8.0 60 27.7 4.5 57.3 13.0 Dolovich M. AJRCCM 1988;137:A433. Scheme of the major variable and interaction in DPI performance DPI design -powder formulation -dose system -dose de-agglomeration principle Performance -Dose entrainment Airflow resistance + Inhalation effort Flow maneuvers -peak flow rate -flow increase rate -inhalation time Patient factors -instruction -clinical parameters -age, gender, training -smoker, nonsmokers -Fine particle fraction -Lung deposition Accuhaler use Turbuhaler use Recommended age for inhalation therapy SVN with mask SVN with mouthpiece pMDI with holding chamber/spacer and mask pMDI with holding chamber/spacer Dry-powder inhaler Metered-dose inhaler Breath-actuated MDI (e.g., Autohaler™) Breath-actuated nebulizers ≤3 years 3 years < 4 years 4 years ≥ 4 years ≥ 5 years ≥ 5 years ≥ 5 years Rau JL Jr. Respiratory care pharmacology. 2002 Turbuhaler® is fully effecitve at flow rate ≥ 30L/min at patients aged ≥ 6 years of age FEV1 (% of predicted) 1 mg terbutaline* 90 80 Mean PIF of Turbuhaler (L/min) 60 31 70 22 13 60 0 0 0.5 1 2 3 4 Time after medication (hours) 0.25 mg terbutaline via Turbuhaler® Pederson S, et al. Arch Dis Child 1990; 65: 308-310 5 30 min after administration of 1mg terbutaline via Nebuhaler treatment. Turbuhaler® is clinically effecitve at both standard & low inspiratory flow rate similar level of bronchodilation & FEV1 FEV1 (litres) 4.0 Standard inhalation conditions at peak inspiratory flow of 83.9L/min Low inspiratory flow rate (30L/min) through entire inhalation 3.5 3.0 0 0.25 0.5 1 Terbutaline (mg) Meijer RJ, et al. Thorax 1996; 51: 433-434 2 4 Higher proportion fine particle dose and lung deposition leads to better efficacy % of metered 40 dose (L) 0.25 mg terbutaline pMDI 30 3.0 Turbuhaler 20 10 0 2.5 Fine particle dose Lung deposition Borgström L, et al. Am J Respir Crit Care Med 1996; 153: 1636-1640 FEV1 0 Turbuhaler gives better central lung deposition as same as pMDI with spacer ® 38% 12% (Central lung deposition 11%) 26% Thorsson L, et al. Int J Pharmaceut 1998; 168: 119-127 (Central lung deposition 11%) Lung deposition of budesonide is greater than that of fluticasone via Diskus or pMDI 1000 Lung deposition 800 budesonide turbuhaler 36% 600 400 200 0 Thorsson L, et al. Br J Clin Pharmacol 2001; 52: 529-538 fluticasone pMDI 20% fluticasone Diskus 12% BUD/FOR turbuhaler delivers higher % of fine particle dose on both BUD & FORM Fine particle dose (% of labeled dose) MMAD (µm) BUD/FOR Turbuhaler Budesonide 63 2.2 Formoterol 55 2.4 SAL/FLU Disku Fluticasone 22 4.4 Salmeterol 22 4.4 MMAD = mass median aerodynamic diameter Granlund KM, et al. Eur Respir J 2000; 16 (suppl. 31): 455s Higher % of fine particle dose with BUD/FOR turbuhaler even at low inspiratory flow 35 (% of label claim) Fine particle dose 30 25 20 15 10 5 0 BUD/FOR Turbuhaler, budesonide Inspiratory flow at 40 L/min. Asking L, et al. J Aerosol M 2001; 14: 502 SAL/FLU Diskus, fluticasone Inspiratory flow at 49 L/min. % fine particle mass at low flow rates in young asthmatic children is also higher with turbuhaler 35 30 25 20 15 10 5 0 BUD/FOR Turbuhaler 80/4.5 µg (LABA component) Gustafsson PM, et al. Am J Respir Crit Care Med 2003; 167: A117 SAL/FLU Diskus 50/100 µg (LABA component) Fine particle size of BUD via Turbuhaler is consistent at all strengths; & higher than FP via Diskus budesonide fluticasone 60 (% of labelled dose) Fine particle dose 50 40 30 20 10 0 80/100 160/250 320/500 Nominal dose of budesonide / fluticasone (µg) Lipniunas et al , 2002 Lipniunas P, et al. Eur Respir J 2002; 20 (suppl. 38): 541s Turbuhaler® Spiral channels • The air enters through air inlets and passes through desiccant store to keep humidity out Dose counter Air inlets Desiccant store Air inlets Turning grip Diskus® • The device should be discarded after removal from the moisture-protective foil overwrap pouch • Diskus® itself does NOT contain desiccant Aluminum blisters may fail to protect against humidity in DiskusTM 25 Serevent® DiskusTM, 50 µg/dose 25°C/60%RH* Fine particle dose % of label claim 20 40°C/75%RH* 15 10 5 0 0 1 2 3 4 5 6 7 Months storage Asking L, et al. J Aerosol Med 1999; 12 (No 3): 204 *RH – relative humidity in vivo lung deposition of budesonide via Turbuhaler is higher even when the inhaler is stored under hot & humid condition (40°/75%) Relative lung deposition 1.0 0.5 0 Budesonide viaTurbuhaler Borgström L, et al. Am J Respir Crit Care Med 2003; 167(suppl. 7): A896 Fluticasone via Diskus Fine particle dose via Turbuhaler at 40°/75% over 3 months is higher Proportion of initial value (%) 120 Fine particle dose Turbuhaler® 100 Diskus™ 80 Delivered dose Turbuhaler® 60 Diskus™ 40 20 0 Initial value Borgström and Lipniunas, 2003 3 months Lipniunas P, et al. Eur Respir J 2003; 22 (suppl. 45): 237s Drug deposition from various inhalers Rau JL Jr. Respiratory care pharmacology. 2002 Hand function in elderly and device Age related physical change Potential effects of inhalation technique in elderly Advantages and disadvantages Advantages Disadvantages pMDI -Quick to use -compact and portable -multi-dose -Difficult inhalation technique -propellant required -High oropharyngeal deposition pMDI +Space (VHC) -Practical advantages as p MDI -Easier to use effectively than p MDI -Reduced oro-pharyngeal deposition -More bulky than p MDI -Propellant required -Susceptible to effect of static charge DPI -Practical advantages similar to p MDI (Multidose/multiple single dose) -No propellant needed -Inspiratory flow-actuated -Easy to use than p MDI -Usually more costly than p MDI -Some may be moisture sensitive -Inspiratory flow-driven (potential problem of low inspiratory force) Treatment include medication Physical communication Symptoms and side effect Disease history HRQL and functionality Treatment history Expectation Direct consumer advising Satisfaction with medication Other influence on expectation Other influence on satisfaction