* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download increase
Coronary artery disease wikipedia , lookup
Mitral insufficiency wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
Myocardial infarction wikipedia , lookup
Jatene procedure wikipedia , lookup
Antihypertensive drug wikipedia , lookup
Dextro-Transposition of the great arteries wikipedia , lookup
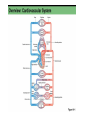
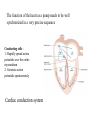
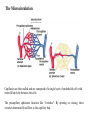
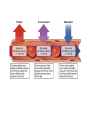
The Cardiovascular System Physiology Dr. Yasir M. Khaleel, M.Sc., PhD The College of Medicine, University of Mosul Components of the CVS • • • • Blood, Vessels, Heart, Associated control systems Circulatory System Overview Heart – "four chambered" - Right atrium & ventricle Pulmonary circuit - Left atrium & ventricle Systemic circuit - Blood Vessels – "closed circulation" Arteries –from heart Capillaries– cell exchange Veins – to heart Functions of the heart 1- Generating Blood Pressure Strength of contraction 2- Routing Blood Separate the pulmonary and systemic circulations 3- Ensuring one-way blood flow Valves prevent backflow of blood. 4- Regulating Blood supply Increase demand (e.g. exercise) results in increase heart rate and strength of contraction Hemodynamics 1- Velocity of Blood Flow: v = Q/A Where v: Velocity of blood flow (cm/sec), Q: Flow (mL/sec), A: Cross-sectional area (cm2) The smallest vessel represents the aorta, the medium-sized vessel represents all of the arteries, and the largest vessel represents all of the capillaries. The total blood flow at each level of blood vessels is the same and is equal to the cardiac output (constant) 2- Relationships between Blood flow, Pressure, and Resistance Q= ΔP/R Where, Q: Flow (mL/min), ΔP: Pressure difference or pressure gradient (mm Hg), R: Resistance (mm Hg/mL/min) The major mechanism for changing blood flow in the cardiovascular system is by changing the resistance of blood vessels, particularly the arterioles. R = ΔP/Q So, TPR= Pressure gradient (aorta – vena cava) / C.O 3- Resistance to Blood Flow R: Resistance; η: Viscosity of blood; L: Length of blood vessel; r4: Radius of blood vessel raised to the fourth power. When the radius of a blood vessel decreases, its resistance increases, not in a linear fashion but magnified by the fourth power relationship. For example, if the radius of a blood vessel decreases by one half, resistance does not simply increase twofold, it increases by 16-fold (24). 4- Series and Parallel Resistances Series resistance Arrangement of blood vessels within a given organ The total resistance of the system arranged in series is equal to the sum of the individual resistances Parallel resistance is illustrated by the distribution of blood flow among the various major arteries branching off the aorta The total resistance in a parallel arrangement is less than any of the individual resistances. Advantages of parallel resistances - The flow through each organ is a fraction of the total blood flow, and there is no loss of pressure in the major arteries and that mean pressure in each major artery will be approximately the same as mean pressure in the aorta. -Adding a resistance to the circuit causes total resistance to decrease, not to increase. -If the resistance of one of the individual vessels in a parallel arrangement increases, then total resistance increases. - Increase resistance in one individual vessel will reduce blood flow in that vessel and increase blood flow in other vessels in parallel 5- Laminar and Turbulent Blood Flow Laminar : the velocity of flow at the vessel wall is zero, and the velocity at the center of the stream is maximal Turbulent: the streams mix radially and axially. Because energy is wasted in propelling blood radially and axially, more energy (pressure) is required to drive turbulent blood flow than laminar blood flow. Turbulent flow is often accompanied by audible vibrations called murmurs. 6- Compliance of Blood vessels The volume of blood the vessel can hold at a given pressure C=V/P C: Compliance (ml/mmHg), V: Volume (ml), P: Pressure (mmHg) The higher the compliance of a vessel, the more volume it can hold at a given pressure. Veins: highly compliant = hold large volume under low pressure (unstressed blood volume) Arteries: much less compliant = hold low volume under high pressure ( stressed blood volume) 7- Pressure in the CVS Blood flow requires a driving force or pressure difference. The pressure differences that exist between the heart and blood vessels are the driving force for blood flow. Systemic arterial pressure Systolic pressure is the highest pressure measured during systole. Diastolic pressure is the lowest pressure measured during diastole. Pulse pressure is the difference between systolic pressure and diastolic pressure. Mean arterial pressure is the average pressure in a complete cardiac cycle and is calculated as follows: MAP= Diastolic Pressure + 1/3 Pulse Pressure Blood Pressure Measurement Cardiac Electrophysiology The function of the heart as a pump needs to be well synchronized in a very precise sequence Conducting cells : 1- Rapidly spread action potentials over the entire myocardium 2- Generate action potentials spontaneously Cardiac conduction system Normal Sinus Rhythm It means: 1- The action potential must originate in the SA node 2- The SA nodal impulses must occur regularly at a rate of 60 to 100 impulses per minute > 100 + from sinus node= sinus tachycardia < 60 + from sinus node= sinus bradycardia 3- The activation of the myocardium must occur in the correct sequence and with the correct timing and delays. Action Potentials of Ventricles, Atria, and the Purkinje System 1- Long duration 250 msec Ventricles 150 msec Atria 300 msec Purkinje fibers Long AP = Long RP 2- Stable resting membrane potential 3- Plateau Sustained period of depolarization Phase 0: Upstroke. phase of rapid depolarization Phase 1: Initial repolarization Phase 2: Plateau. a long period (150 to 200 msec) of relatively stable, depolarized membrane potential Phase 3: Repolarization Phase 4: Resting membrane potential Inward and outward currents are equal Ventricular action potential Action Potentials in the SA Node Phase 0: Upstroke T-type Calcium channel Phase 3: Repolarization Phase 4: Spontaneous depolarization or pacemaker potential Funny or leaky Na channels = inward Na+ current called If If is turned on by repolarization from the preceding AP, thus ensuring that each AP in the SA node will be followed by another AP (1) The SA node exhibits automaticity; that is, it can spontaneously generate action potentials without neural input. (2) It has an unstable resting membrane potential (3) It has no sustained plateau. Sympathetic NS Activation of β1 receptors in the SA node produces an increase in If, which increases the rate of phase 4 depolarization (Positive chronotropic effects ) Parasympathetic NS activate muscarinic (M2) receptors in the SA node. Has two effects - Decrease in If decreases the rate of phase 4 - Increases the conductance of K+ resulting in outward K+ current that hyperpolarize the maximum diastolic potential so that more inward current is required to reach threshold potential The pacemaker with the fastest rate of phase 4 depolarization controls the heart rate. Normally, the SA node has the fastest rate of phase 4 depolarization, and therefore, it sets the heart rate When the SA node drives the heart rate, the latent pacemakers are suppressed, a phenomenon called overdrive suppression. That is, latent pacemakers as long as their firing rate is driven by the SA node, their own capacities to spontaneously depolarize are suppressed. Mechanism of Propagation of Cardiac Action Potential The cardiac muscle work as syncytium - Atrial syncytium - Ventricular syncytium This achieved by Intercalated discs which connects cardiac muscle cells together to work as one unit (syncytium) Intercalated discs - Desmosomes make strong mechanical attachments between the cells and transmit the force of contraction. - Gap junctions cause every cell in the heart to be electrically coupled to its neighboring cells and that is what causes the heart to behave like a single motor unit. CONDUCTION VELOCITY Differences in conduction velocity among the cardiac tissues have implications for their physiologic functions ELECTROCARDIOGRAM (ECG) The entire myocardium is not depolarized at once: - The atria depolarize before the ventricles; -The ventricles depolarize in a specific sequence; -The atria repolarize while the ventricles are depolarizing; - The ventricles repolarize in a specific sequence. As a result of the sequence and the timing of the spread of depolarization and repolarization in the myocardium, potential differences are established between different portions of the heart, which can be detected by electrodes placed on the body surface. The electrocardiogram (ECG or EKG) is a measurement of tiny potential differences on the surface of the body that reflect the electrical activity of the heart. P wave duration is (80-110 msec) PR interval (120-200 msec) It reflects delay in the AV node. QRS duration (60-100 msec). QT interval duration (330-450 msec) It represents the whole ventricular depolarization and ventricular repolarization. T wave represents repolarization of the ventricles ECG Lab CONTRACTILITY Inotropism The intrinsic ability of myocardial cells to develop force at a given muscle cell length Agents that produce an increase in contractility are said to have positive inotropic effects. Positive inotropic agents increase both the rate of tension development and the peak tension. Agents that produce a decrease in contractility are said to have negative inotropic effects. Negative inotropic agents decrease both the rate of tension development and the peak tension Excitation Contraction Coupling Mechanisms for Changing Contractility 1- Intracellular Ca2+ concentration - Action P. , inward Ca2+ current during plateau, - Trigger more calcium release from SR (Ca2+-induced Ca2+ release) -Ca2+ enhance the interaction of actin and myosin -Contraction The magnitude of the tension developed by myocardial cells is proportional to the intracellular Ca2+ concentration. The amount of Ca2+ released from the SR depends on two factors: 1- Size of the inward Ca2+ current during the plateau 2-Amount of Ca2+ previously stored in the SR for release. Therefore, the larger the inward Ca2+ current and the larger the intracellular stores, the greater the increase in intracellular Ca2+ concentration and the greater the contractility. 2- Autonomic Nervous System Sympathetic nervous system: Stimulation of the sympathetic NS has a positive inotropic effect mediated via activation of β1 receptors. This positive inotropic effect has three important features: 1-increased peak tension, 2- increased rate of tension development, and 3- faster rate of relaxation. Faster relaxation means that the contraction is shorter, allowing more time for refilling Parasympathetic NS: Stimulation of the parasympathetic nervous system has a negative inotropic effect on the atria. This effect is mediated via the action of Ach on muscarinic receptors; 3- Effect of Heart Rate When the heart rate increases, contractility increases; when the heart rate decreases, contractility decreases. (1) When heart rate increases, there are more action potentials per unit time and an increase in the total amount of trigger Ca2+ that enters the cell during the plateau phases. (2) Because there is greater influx of Ca2+ into the cell during the action potentials, the sarcoplasmic reticulum accumulates more Ca2+ for subsequent release (i.e., increased stored Ca2+). Tension rises stepwise, like a staircase ; With each beat, more Ca2+ is accumulated by the sarcoplasmic reticulum, until a maximum storage level is achieved (Positive staircase effect) Positive staircase effect 4- Cardiac muscle fiber length (Length-Tension Relationship ) a- (Lmax). At this length, there is maximal overlap of thick and thin filaments; at either shorter or longer cell lengths, the tension developed will be less than maximal (2.2 μm) b- Increasing muscle length increases the Ca2+-sensitivity of troponin C c- Increasing muscle length increases Ca2+ release from the sarcoplasmic reticulum. Frank-Starling relationship Under normal physiological conditions, the volume of blood ejected by the ventricle (strength of ventricular myocardial contraction )during systole, depends on the volume of blood present in the ventricle at the end of diastole. Ventricular end-diastolic volume, or end-diastolic fiber length; (Preload) That is, preload is the resting length from which the muscle contracts. End diastolic pressure-volume relationship (EDPVR) Number 3 and 4 are the best to develop contraction The upper curve is the relationship between ventricular pressure developed during systole and enddiastolic volume (or end-diastolic fiber length). This pressure development is an active mechanism. On the ascending limb of the curve, pressure increases steeply as fiber length increases, reflecting greater degrees of overlap of actin and myosin. The curve eventually levels off when overlap is maximal. If enddiastolic volume were to increase further and the fibers were stretched to even longer lengths, overlap would decrease and the pressure would decrease (descending limb of the curve). The afterload is the force against which the ventricles eject blood. The mechanical "load" on the ventricle during ejection. For the left ventricle is aortic pressure. The velocity of shortening of cardiac muscle is maximal when afterload is zero, and velocity of shortening decreases as afterload increases. Stroke volume is the volume of blood ejected by the ventricle on each beat. SV= End diastolic volume – End systolic volume (about 70 mL) Ejection fraction is the fraction of the end-diastolic volume ejected in each stroke volume, which is a measure of ventricular efficiency. Cardiac output is the total volume ejected by the ventricle per unit time. Cardiac output = Stroke volume x Heart rate SAMPLE PROBLEM: A man has an end-diastolic volume of 140 mL, an end-systolic volume of 70 mL, and a heart rate of 75 beats/min. What are his stroke volume, his cardiac output, and his ejection fraction? Stroke volume= 140 mL – 70 mL = 70 mL Cardiac output= 70 mL x 75 bpm = 5250 mL/min Ejection fraction= SV/EDV = 70 mL/140 mL = 0.50 = 50% Positive inotropic agents like digoxin produce increases in stroke volume and cardiac output for a given end-diastolic volume. The result is that a larger fraction of the end-diastolic volume is ejected per beat and there is an increase in ejection fraction. Negative inotropic agents produce decreases in stroke volume and cardiac output for a given enddiastolic volume. The result is that a smaller fraction of the enddiastolic volume is ejected per beat and there is a decrease in ejection fraction. Ventricular Pressure-Volume Loop Isovolumetric contraction (1 → 2) (all valves are closed) Ventricular ejection (2 → 3) Isovolumetric relaxation (3 → 4) (all valves are closed) Ventricular filling (4 → 1) Width of the loop is the difference between EDV and ESV = SV A- Increase preload (EDV) …..Increased SV B- Increase afterload …………Decreased SV C- Increase contractility……….Increased SV SV= End diastolic volume – End systolic volume (about 70 mL) So, SV determined by (Preload, Afterload, Contractility) Cardiac Cycle The mechanical and electrical events that occur during a single cardiac cycle; A-ATRIAL SYSTOLE atrial systole is atrial contraction. It is preceded by the P wave on the ECG B-ISOVOLUMETRIC VENTRICULAR CONTRACTION -It begins during the QRS complex - Mitral & Tricuspid valve close (S1) C-RAPID VENTRICULAR EJECTION - Begins by opening of aortic and pul valves D-REDUCED VENTRICULAR EJECTION E-ISOVOLUMETRIC VENTRICULAR RELAXATION -After full repolarization , ventricles start to relax (diastole) - Aortic and pul valves close (S2) F-RAPID VENTRICULAR FILLING -Mitral and tricuspid valves open (lowest LVP) - Blood flows from atria to ventricles passively Rapid ventricular filling may cause S3 G-REDUCED VENTRICULAR FILLING Reduced ventricular filling or diastasis is the longest phase of the cardiac cycle and includes the final portion of ventricular filling A-ATRIAL SYSTOLE Active ventricular filling (S4) may be heard if ventricular compliance is decreased Relationships between Cardiac Output and Venous Return It should be clear now that one of the most important factors determining cardiac output is left ventricular end-diastolic volume (Preload). In turn, left ventricular end-diastolic volume depends on venous return, Venous return determines right atrial pressure. So, there is no only a relationship between cardiac output and end-diastolic volume but also a relationship between cardiac output and right atrial pressure Cardiac Function curve As venous return increases, RAP increases, and end-diastolic volume and end-diastolic fiber length increase. Increase in enddiastolic fiber length produce increases in cardiac output. Thus, in the steady state, the volume of blood the left ventricle ejects as cardiac output equals or matches the volume it receives in venous return. When right atrial pressure reaches a value of approximately 4 mm Hg, cardiac output can no longer keep up with venous return, and the cardiac function curve levels off. This maximum level of cardiac output is approximately 9 L/min. Vascular Function Curve The relationship between venous return and right atrial pressure. There is inverse relationship between venous return and right atrial pressure. Increase right atrial pressure will lead to decrease venous return Because increased RAP will lead to reduction in pressure gradient between the veins and the RA, so reduce blood flow to the RA (decrease venous return). Vascular Function Curve Continue…….. If RAP becomes negative What will happen? knee (flat portion) of the vascular function curve occurs at negative values of RAP. At such negative values, the veins collapse, impeding blood flow back to the heart. Although the pressure gradient has increased venous return levels off because the veins have collapsed Slope of the Vascular Function Curve Decreased resistance of the arterioles (decreased TPR) makes it easier for blood to flow from the arterial to the venous side of the circulation and back to the heart. So , for a given RAP, venous return is increased and there will be clockwise rotation of the vascular function curve. The reverse is true, Increase TPR for a given RAP….decrease venous return and there will be counter clockwise rotation of the vascular function curve. So, the slope of the vascular function curve is determined by total peripheral resistance Mean circulatory pressure (Mean Systemic Pressure) Is the pressure that would be measured throughout the cardiovascular system if the heart is stopped, Under these conditions, pressure would be the same throughout the vasculature and, by definition, would be equal to the mean systemic pressure. When pressures are equal throughout the vasculature, there is no blood flow, and therefore, venous return is zero (since there is no pressure gradient or driving force). In other word it is the value for RAP at which venous return is zero and it is the point at which the vascular function curve intersects the X-axis (i.e., where venous return is zero and right atrial pressure is at its highest value) Factors determine the mean systemic pressure (1) The blood volume, (2) The distribution of blood between the unstressed volume and the stressed volume. The unstressed volume (the volume of blood in the veins) is the volume of blood in the vasculature that produces no pressure. The stressed volume (the volume of blood in the arteries) is the volume that produces pressure by stretching the elastic fibers in the blood vessel walls. 1-Effect of changing blood volume on MSP: When the blood volume ranges from 0 to 4 L, all of the blood will be in the unstressed volume, producing no pressure, and the MSP will be zero. While, if the total blood volume is 5 L, 4 L is in the unstressed volume, producing no pressure, and 1 L is in the stressed volume, producing a pressure of approximately 7 mm Hg. So, when blood volume increases (>4 L) unstressed volume will be unaffected (it is already full), but stressed volume will increase. When stressed volume increases, MSP increases, and the vascular function curve and its intersection point with the X-axis shift to the right. When blood volume decreases then stressed volume decreases, MSP decreases and the vascular function curve and its intersection point with the X-axis shift to the left. 2-Redistribution of blood between the unstressed volume and the stressed volume also produces changes in MSP. For example, if the compliance of the veins decreases, then veins can hold less blood and blood shifts from the unstressed volume to the stressed volume. Although total blood volume is unchanged, the shift of blood increases the MSP and shifts the vascular function curve to the right. (Similar changes of increased blood volume) Conversely, if the compliance of the veins increases, the veins can hold more blood. Hence, the unstressed volume will increase, the stressed volume and mean systemic pressure will decrease, and the vascular function curve shifts to the left (Similar changes of decreased blood volume) Changes in the cardiac output Changes in cardiac output can be produced by any of the following mechanisms: (1) Positive or negative inotropic effects that alter the cardiac function curve (i.e. Contractility) (2) Changes in blood volume or venous compliance that alter the vascular function curve by changing mean systemic pressure (3) Changes in TPR that alter both the cardiac and vascular function curves. (1) Inotropic Effects A-Positive inotropic agents (e.g. adrenalin, digoxin), produce an increase in contractility, an increase in SV, and an increase in cardiac output for any level of right atrial pressure. Thus, the cardiac function curve shifts upward, but the vascular function curve is unaffected. Also note that increased contractility will result in decrease RAP B- Negative inotropic agents……The reverse effect. (2) Effects of Changes in Blood Volume A- Increased blood volume , results in increase MSP, vascular function curve shift to right. Cardiac output increased, RAP increased. Decreases in venous compliance , produce similar changes. B- Decreased blood volume , results in decreased MSP, vascular function curve shift to left. Cardiac output decrease, RAP decrease. Increase venous compliance, produce similar changes (3) Effects of Changes in TPR A- Increase in TPR, -By restricting the flow of blood out of the arteries, produces an increase in arterial blood pressure (Increase afterload , so decrease C.O) - At the same time it decreases the venous return - The curves intersect at a new steady state point at which both cardiac output and venous return are decreased In the figure, right atrial pressure is shown as unchanged. Actually, the effect of increased TPR on right atrial pressure is not easily predicted because TPR has different effects via the cardiac and vascular function curves. -An increase in TPR decreases cardiac output, which increases right atrial pressure (less blood is pumped out of the heart). -An increase in TPR decreases venous return, which decreases right atrial pressure (less flow back to the heart). Depending on the relative magnitude of effects on the cardiac and vascular function curves, right atrial pressure can be -slightly increased, -slightly decreased, -or unchanged. B- Decrease in TPR -Decrease arterial BP, decrease afterload, so C.O will be increased and cardiac function curve shifts upward. - Decrease in TPR produces a clockwise rotation of the vascular function curve, which means that more venous return for a given right atrial pressure. -The curves intersect at a new steady state point at which both cardiac output and venous return are increased The effect of decreased TPR on RAP is not easily predicted because a change in TPR has different effects via the cardiac and vascular function curves. -A decrease in TPR increases cardiac output, which decreases RAP (more blood is pumped out of the heart). -A decrease in TPR increases venous return, which increases RAP (increased flow back to the heart). - Depending on the relative magnitude of the effects, right atrial pressure can be slightly increased, slightly decreased, or unchanged. Regulation of Arterial Pressure Mean arterial pressure (Pa) is the driving force for blood flow, and it must be maintained at a high, constant level of approximately 100 mm Hg. Because of the parallel arrangement of arteries out of the aorta, the pressure in the major artery serving each organ is equal to Pa. (The blood flow to each organ is then independently regulated by changing the resistance of its arterioles through local control mechanisms.) Pa = CO X TPR Pa can be changed by •Altering the cardiac output (or any of its parameters), •Altering the TPR (or any of its parameters), or • Altering both cardiac output and TPR. Cardiac output and TPR are not independent variables. In other words, changes in TPR can alter cardiac output, and changes in cardiac output can alter TPR. Therefore, it cannot be stated that if TPR doubles, Pa also doubles. (In fact, when TPR doubles, cardiac output simultaneously is almost halved, and Pa will increase only modestly.) Likewise, it cannot be stated that if cardiac output is halved, Pa also will be halved. (Rather, if cardiac output is halved, there is a compensatory increase in TPR, and Pa will decrease but it will not be halved.) There are mechanisms closely monitor Pa and compare it with the set-point value of approximately 100 mm Hg. If Pa increases above the set point or decreases below the set point, the cardiovascular system makes adjustments in cardiac output, in TPR, or in both, attempting to return Pa to the set-point value. 1- The baroreceptor reflex (neurally mediated) attempts to restore Pa to its set-point value in a matter of seconds. 2- Renin-angiotensin-aldosterone system, which regulates Pa more slowly, primarily by its effect on blood volume. The baroreceptor reflex mechanisms are fast, neurally mediated reflexes that attempt to keep arterial pressure constant via changes in the output of the sympathetic and parasympathetic nervous systems to the heart and blood vessels Reflex arc -Sensors for blood pressure; -Afferent neurons, (IX, X ) -Brain stem centers, which process the information and coordinate an appropriate response NTS (medulla) cardiovascular centers (which are tonically active) -Efferent neurons, (sympathetic and parasympathetic) The parasympathetic outflow is the effect of the vagus nerve on the SA node to decrease the heart rate. The sympathetic outflow has four components: effect on the SA node to increase heart rate, an effect on cardiac muscle to increase contractility and stroke volume, an effect on the arterioles to produce vasoconstriction and increase TPR, and an effect on veins to produce venoconstriction and decrease unstressed volume. Response of baroreceptor reflex to increased arterial pressure Response of the Baroreceptor Reflex to Hemorrhage Renin-angiotensin -aldosterone system The renin-angiotensin -aldosterone system regulates Pa primarily by regulating blood volume. This system is much slower than the baroreceptor reflex because it is hormonally, rather than neurally, mediated. This system is activated in response to a decrease in the Pa, which in turn, produces a series of responses that attempt to restore arterial pressure to normal. Angiotensin II is an octapeptide with biologic actions in the: 1- Adrenal cortex : stimulates synthesis and secretion of aldosterone which acts on the principal cells of the renal distal tubule and collecting duct to increase Na+ reabsorption and, thereby, to increase ECF volume and blood volume. Actions of aldosterone require gene transcription and new protein synthesis in the kidney. These processes require hours to days to occur and account for the slow response time of the renin-angiotensin II-aldosterone system. 2- Vascular smooth muscle, direct action on the arterioles to cause vasoconstriction which increase TPR 3- Kidneys, direct action, stimulates Na+-H+ exchange in the renal proximal tubule and increases the reabsorption of Na+ and HCO3 4- Brain acts on the hypothalamus to increase thirst and water intake. It also stimulates secretion of ADH, which increases water reabsorption in collecting ducts. By increasing total body water, these effects complement the increases in Na+ reabsorption (caused by aldosterone and Na+-H+ exchange), thereby increasing ECF volume, blood volume, and blood pressure. Other regulatory mechanisms for blood pressure 1- Peripheral chemoreceptors for O2 are located in the carotid bodies near the bifurcation of the common carotid arteries and in the aortic bodies along the aortic arch. They respond to decreased arterial Po2 especially when the PCO2 is increased or the pH is decreased. When stimulated-----------activate sympathetic vasoconstrictor centers 2- Central (cerebral) Chemoreceptors: the brain is intolerant of decreases in blood flow, chemoreceptors are located in the medulla and they are most sensitive to CO2 and pH and less sensitive to O2. Changes in PCO2 or pH stimulate the medullary chemoreceptors, which then direct changes in outflow of the medullary cardiovascular centers. Cushing reaction 3- Antidiuretic hormone (ADH) Its secretion from the posterior pituitary is increased by two types of stimuli: by increases in serum osmolarity and by decreases in blood pressure. There are two types of receptors for ADH: -V1 receptors, which are present in vascular smooth muscle, and - V2 receptors, which are present in principal cells of the renal collecting ducts. When activated, the V1 receptors cause vasoconstriction of arterioles and increased TPR. The V2 receptors are involved in water reabsorption in the collecting ducts and the maintenance of body fluid osmolarity. 4- Cardiopulmonary (Low-Pressure) Baroreceptors They are located in the veins, atria, and pulmonary arteries. They sense changes in blood volume, or the "fullness" of the vascular system. They are located on the venous side of the circulation where most of the blood volume is present. When there is an increase in blood volume, the resulting increase in venous and atrial pressure is detected by the cardiopulmonary baroreceptors. The function of the cardiopulmonary baroreceptors is then coordinated to return blood volume to normal, primarily by increasing the excretion of Na+ and water. The responses to an increase in blood volume include the following: 1- Increased secretion of ANP which is secreted by atrial cells in response to increase atrial pressure. Results in vasodilation and decreased TPR. In the kidneys, this vasodilation leads to increased Na+ and water excretion 2- Decreased secretion of ADH. Pressure receptors in the atria also project to the hypothalamus, where the cell bodies of neurons that secrete ADH are located. In response to increased atrial pressure, ADH secretion is inhibited and, as a consequence, there is decreased water reabsorption in collecting ducts, resulting in increased water excretion 3- Renal vasodilation. There is inhibition of sympathetic vasoconstriction in renal arterioles, leading to renal vasodilation and increased Na+ and water excretion, complementing the action of ANP on the kidneys. 4-Increased heart rate. Information from the low-pressure atrial receptors travels in the vagus nerve to the nucleus tractus solitarius. Increase in pressure at the venous low-pressure receptors produces an increase in heart rate (Bainbridge reflex). The low-pressure atrial receptors, sensing that blood volume is too high, direct an increase in heart rate and, thus, an increase in cardiac output; the increase in cardiac output leads to increased renal perfusion and increased Na+ and water excretion. The Microcirculation Capillaries are thin walled and are composed of a single layer of endothelial cells with water-filled clefts between the cells. The precapillary sphincters function like "switches": By opening or closing, these switches determine blood flow to the capillary bed. Exchange of substances across the capillary wall The exchange of solutes and gases across the capillary wall occurs by simple diffusion. Some solutes can diffuse through the endothelial cells, and others must diffuse between the cells. Generally, the route for diffusion depends on whether the solute or gas is lipid soluble. -Lipid soluble: through the endothelial cells (more surface area) - Water soluble : aqueous clefts between endothelial cells (less surface area) - Proteins: large molecules, retained in the capillaries and contribute to the colloidal osmotic pressure ( Oncotic Pressure) Fluid Exchange across Capillaries The Starling equation states that fluid movement (Jv) across a capillary wall is determined by the net pressure across the wall, which is the sum of hydrostatic pressure and oncotic pressures. The direction of fluid movement can be either into or out of the capillary. When net fluid movement is out of the capillary into the interstitial fluid, it is called filtration; when net fluid movement is from the interstitium into the capillary, it is called absorption. The magnitude of fluid movement is determined by the hydraulic conductance, Kf (water permeability), of the capillary wall. The hydraulic conductance determines how much fluid movement will be produced for a given pressure difference. A- Hydrostatic P= +30 -1 =+29 Oncotic = -26 +3= -23 Net pressure= +6 mmHg B- Hydrostatic P= +25-1 =+24 Oncotic = -32 +3= -29 Net pressure= -5 mmHg SAMPLE PROBLEM. In a skeletal muscle capillary, the following Starling pressures were measured: Pc= 30 mmHg, Pi = 1 mmHg, πc = 26 mmHg, πi = 3mmHg Assuming that Kf is 0.5 mL/min.mm Hg, what is the direction and magnitude of fluid movement across this capillary? Net pressure = +30 - 1 -26 + 3 mm Hg = +6 mm Hg Jv (fluid movement) = Net pressure * Kf (+6mmHg * 0.5 ml/min.mmHg = 3ml/min) So, the direction is filtration, and the magnitude of fluid movement is 3 ml/min. Lymph Lymphatic capillaries lie in the interstitial fluid, close to the vascular capillaries. Responsible for returning interstitial fluid and proteins to the vascular compartment 1- One-way flap valves 2- Lymphatic vessels have a smooth muscle wall, which has intrinsic contractile ability 3- Lymph flow back to the thoracic duct is promoted by contraction of the smooth muscle in the lymph vessels and by compression of the lymph vessels by activity of the surrounding skeletal muscle. Edema (swelling or increase in the interstitial fluid volume) it occurs when the volume of interstitial fluid (due to filtration out of the capillaries) exceeds the ability of the lymphatics to return it to the circulation. Thus, edema can form when there is increased filtration or when lymphatic drainage is impaired. Causes of Edema: ↑ Pc (capillary hydrostatic pressure) Examples: Arteriolar dilation, venous constriction, increased venous pressure, heart failure, extracellular fluid volume expansion. ↓ πc (capillary oncotic pressure) Examples: Decreased plasma protein concentration, severe liver failure (failure to synthesize protein), protein malnutrition, nephrotic syndrome (loss of protein in urine). ↑ Kf (hydraulic conductance); increase capillary permeability. Examples: Burns, inflammation (release of histamine; cytokines) Impaired lymphatic drainage Examples: Standing for long time (lack of skeletal muscle compression of lymphatics), removal or irradiation of lymph nodes, parasitic infestation of the lymphatics.