* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Review 1
Survey
Document related concepts
Transcript
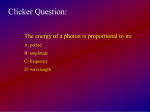
Light & Matter 1. Dual Nature: Wave & Particle 2. Light travels at a speed of c=300,000 km/s 3. Wave: Light is made of different colors. Each color has its own wavelength λ. 4. Light is electromagnetic radiation: from radio waves to Gamma rays. 5. Wavelength and Frequency are related: frequency ν = c/λ 6. A dense hot object emits light according to its temperature T.: The Black Body Radiation 7. Wien’s Law: λmax = 0.0029/T ⇒ The wavelength of maximum emission of a black-body is inversely proportional to its temperature in Kelvins. 8. Stefan-Boltzmann’s Law: F = σ T 4 ⇒ The flux from a black body is proportional to the the fourth power of the object’s temperature. 9. Particle nature: Each photon has an unique energy E. 10. Planck’s laws: E = h ν or E = h c/λ ....relate energy of a photon to its wavelength. 11. Each element, e.g. H, He, C,..., has its own unique set of spectral lines. 12. Kirchhoff’s Laws: : 1. A hot opaque body produces continuous spectrum. : 2. A hot transparent gas produces an emission line spectrum. : 3. A cooler, transparent gas in front of a source of continuous spectrum produces an absorption line spectrum. 13. An atom is made of a small dense nucleus (containing protons and neutrons) surrounded by electrons. 14. Spectral lines are produced when an electron jumps from one energy level to another within an atom. 15. In the Bohr model of an atom, electrons have discrete orbits. This model, together with the particle nature of light, helped explain the emission and absorption lines. 16. Doppler Effect: ∆λ/λ = v/c ⇒ If an object is moving towards the observer, its spectrum appears blueshifted, i.e. the wavelengths of its spectral lines appear smaller. If an object is moving away from the observer, its spectrum appears redshifted, i.e. the wavelengths of its spectral lines appear larger. The Sun Let us remind ourselves: 1. Photosphere produces the continuum spectrum peaking in yellow. This tells us that its temperature is 5800 K 2. Upper levels of Photosphere are cooler, making an absorption line spectrum. 3. Dark edges in Solar granules imply lower temperature. 4. The Chromosphere has an emission line spectrum. Its pink color is produced by the emission of Hα line. 5. The Corona is very hot: over two million K. It also has an emission line spectrum of highly ionized metals. Sun as a star: why does it shine? 6. Age crisis I: Known sources of energy, e.g. chemical, do not seem to be good enough to keep the Sun shining for billions of years. 7. Understanding stellar structure: Gas laws: Pressure= constant × density × Temperature 8. Laws of Gravity: Gravitational bining is stronger for smaller radii, for the same mass (i.e. if we compress a star it is held together better by its gravity). 9. Hydrostatic Equlibrium: when pressure and gravity are in exact balance. Then a star neither expands nor contracts. 10. Kelvin-Helmholtz Mechanism: use of gravitational energy to power the sun. Age Crisis II: This energy, again, is not good enough. 11. The Sun continues to shine because of the thermonuclear reactions in its core. 12. In Hydrogen burning: 4 H → He + Energy. ⇒ The small amount of mass lost when Hydrogen is converted to Helium, is converted to energy. 13. Proton-Proton Chain tells us how 4 Hydrogen nuclei make one Helium nucleus. 14. The high temperature found in the Sun’s core enables thermonuclear reactions as it helps overcome the strong repulsive electric force between protons.