* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Ch. 17 Reaction Energy (Thermochemistry )
Entropy in thermodynamics and information theory wikipedia , lookup
Second law of thermodynamics wikipedia , lookup
Internal energy wikipedia , lookup
Thermodynamic system wikipedia , lookup
Heat transfer physics wikipedia , lookup
Conservation of energy wikipedia , lookup
History of thermodynamics wikipedia , lookup
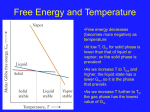
Ch. 17 Reaction Energy (Thermochemistry )and Reaction Kinetics 4.03 Analyze the relationship between entropy and disorder in the universe. A student should: Understand entropy as a measure of disorder. Recognize that the entropy of the universe is increasing. Explain that, along with a tendency for systems to proceed toward the lowest energy level, they also move in the direction of the greatest entropy. (Increasing Entropy: solid liquid gas; Ionic compounds ions in solution) Honors: Understand Gibbs free energy and how it is used to predict spontaneity. Calculations with G=H-TS lst Law of thermodynamics: Energy is Conserved Euniv = Esystem + Esurroundings = constant 2nd Law of Thermodynamics: Entropy of the universe is increasing Define the following terms: Thermochemistry ______________________________ Heat _____________________________ Temperature _______________________ Calorimeter _________________________ Specific heat_____________________________ Specific heat of Al ____________________________ As the temperature of 50.0 grams of Al changes from 20.0 C to 70.0 C, how much energy is required? ___________________ Specific heat of Pb is ___________________ As the temperature of 50.0 grams of Pb changes from 20.0 C to 70.0 C, how much energy is required? ___________________ HEAT of REACTION Write the thermochemical reaction for the synthesis of water (p.515) ____________________________________________________ What does a thermochemical equation contain that other equations do not contain? _________________________ How much heat is released or absorbed from the synthesis reaction of 6.50 moles of water? Define enthalpy? ______________________________________________________________________________ Rules for Thermodynamic equations: circle answer when applicable. 1. Coefficients represent ___________________ 2. Physical state is or is not important. 3. Energy change is directly or not directly related to the number of moles of reactant. 4. Value if H is or is not affected by temperature change of the reaction. Heat of Formation: p. 517 Define Molar Heat of Formation ____________________________________________ 1 Stability and Heat of formation p. 518 If a large amount of heat is released when a compound is formed from its elements, it has a high negative . heat of formation. These compounds are very _________________. Elements in their standard states are defined as having Hfo = 0 It is -393.5 kJ/mole for carbon dioxide (very stable molecule) Just be aware that there are Heats of Combustion (per mole of reactant) and Heats of Reactions per mole of reaction as written. These are calculated from the standard thermodynamic Hfo values. Problems: Determine the H and the type of reaction (endothermic or exothermic) for the following reactions: 1. C(s) + O2(g) CO2(g) + 393.15 kJ H = __________________________ 2. CaCO3 (s) + 176 kJ CaO(s) + CO2(g) H = __________________________ Rewrite the following equation as a thermodynamic reaction. Include ΔH and if the reaction is endo or exo. 3. 2 Mg (s) + O2(g) 2 MgO (s) ; Ho = -1200 kJ/mol 4. Write the reaction for CaCl2(s) illustrating the formation of each of the compounds from its elements. Write the ΔH as part of each equation and indicate the ΔH for the reverse reaction. ___________________________________________________________________________________________ Finish all of Problems 18, 19 and 20 from p. 548 using the Appendix Table A-14 on p.903. Driving Force of Reactions p.526 A Brief Review of Enthalpy: Most, but NOT all spontaneous rxns evolve energy For a rxn to be exothermic: Hrxn = Hf(prod) - Hf(reactants) must be less than 0 Hrxn is called Standard Enthalpy Change Thermodynamic standard states = 1 atm, 1 M for sol'ns, 25 oC (298 K) Hrxn = Hf(prod) - Hf(reactants) Standard enthalpy change is approximately equal to the Standard energy change Enthalpy is relatively independent of pressure and temperature. Standard Enthalpy Change is given by: Hrxn = ΔHfo(prod) - ΔHfo(reactants) Spontaneous- a process that takes place w/o input of energy from external source. Natural tendency --------> decrease in energy i.e. ΔH is Examples water flows downhill, a spring unwinds CH4(g) + 2O2(g) ------> C02(g) + 2H2O(g) ΔHrxn = - 802 kJ 4Fe(s) + 3O2(g) -----> 2Fe2O3(s) ΔHrxn = -1648kJ Zn(s) + 2HCl(aq) -----> H2(g) + ZnC12(aq) ΔHrxn = -150kJ These rxns do not occur spontaneously in the reverse direction. Practice: 1. How much energy is released from 1.00 moles of methane in reaction 1? _________________ 2. How much energy is released from 2.00 moles of iron in reaction 2? ____ 2 3. How much energy is released from 6.00 moles of Zinc in reaction 3? ____ Standard Entropy Change is given by: ΔSrxn = SΔSo (prod) - SΔSo (reactants) Practice: Define entropy _____________________________________ 1. Is entropy increasing or decreasing with the following? a. Ice is melting _____________ b. Copper (II) Chloride is dissolved into water. _________ c. Gas is forming in a chemical reaction. _________ d. A reaction occurs between Magnesium and hydrochloric acid. _____________ e. Crystals of potassium nitrate are forming _________ f. Liquid is formed from vapor ________ Calculate H, S, and G for each of the following. Need reference Tables ( in Holt only enthalpy p. 903) One example 1. Cl2 (g) + 2HBr (g) 2HCl (g) + Br2 (g) products [HCl + Br2] [Cl2 + HBr ] reactants H = [ 2(-92.307) + 0] - [0 + 2(-36.40)] = -80.914 kJ S = [ 2 (186.908) + 152.231] - [223.066 + 2(198.695)] = -1.174 J/K G = [ 2(-95.299) + 0] - [ 0 + 2(-53.430)] = -80.598K 2. 2KBr (s) + H2SO4 (l) K2SO4 (s) + 2HBr (g) optional if time allows Gibbs-Helmholtz Equation ( Gibbs Free Energy or Free Energy): must be – to be spontaneous ΔGrxn = ΔHrxn - TΔSrxn Tendency in nature -----> higher entropy Processes that are both energy favored and entropy favored will be spontaneous. ΔH less than 0 and ΔS greater than 0 Criteria for spontaniety: if ΔGrxn is less than 0, then rxn is spontaneous i.e. ________if ΔGrxn greater than 0, then rxn is not spontaneous i.e. ________ if ΔGrxn = 0, then rxn is at equilibrium, no net change occurs. Practice: 1. Is entropy increasing or decreasing with the following? a. Ice is melting _____________ b. Copper (II) Chloride is dissolved into water. _________ c. Gas is forming in a chemical reaction. _________ d. A reaction occurs between Magnesium and hydrochloric acid. _____________ e. Crystals of potassium nitrate are forming _________ 2. Compute ΔGrxn and decide if the following is a spontaneous process? Yes or No ΔH =125 kJ/mole and ΔS =0.350 kJ/mole at T = 293 K ____ ΔH =-85 kJ/mole and ΔS =0.125 kJ/mole at T = 127 C _______ Standard Enthalpy Change is given by: ΔHrxn = SΔHf(prod) - SΔHf(reactants) 3 What is the molar heat of formation? ________________________________________ Skip the rest of this unit 17-1 Section 17-2 Driving Force of Reactions What is the relationship between enthalpy and reaction tendency? _________________________________________________________________________________ What is the relationship between entropy and reaction tendency? _________________________________________________________________________________ What is entropy? ____________________________________________________ What is free energy? ____________________________ Write the reaction for free energy ________________________________________ If free energy has a positive value, is the reaction spontaneous? If free energy has a negative value, is the reaction spontaneous? If a reaction occurs at 25 C, and releases 160 kJ/mol of energy and has an entropy value of positive 50 J/mol K, what is the free energy of the reaction? Is it spontaneous? Section 17-3 Reaction Process What is a reaction mechanism? ____________________________________________________ What is collision theory? ___________________________________________ What is activation energy? _____________________________________________ Do sample problem 17-5 p.536, label well, then do the practice problem on p. 537 Section 17-4 Reaction Rate Define reaction rate _________________________________________________ Define chemical kinetics ________________________________ Rate Influencing factors List: Draw Figure 17-15 with reaction with no catalyst and the reaction with one catalyst, label all parts well. Skip Rate Laws 4 Fill in the answers to the following questions about the energy profile diagram. See p. 534 The following questions refer to the diagram below. (a) Which letter represents the potential energy of the products? ________ (b) Which letter represents the potential energy of the reactants? ________ (c) Which letter represents the potential energy of the activated complex? ________ (d) Which letter represents the activation energy for the reverse reaction? ________ (e) Which letter represents the activation energy for the forward reaction? ________ (f) If the potential energy of the reactants is 8.3 kJ and the potential energy of the products is 10.7 kJ, what is the heat of the forward reaction (Hfwd)? (g) Is the reverse reaction endothermic? ________ (h) If a catalyst was added to the reaction, which lettered quantities would change? (List all that apply.) (i) If a catalyst was added to the reaction, what would happen to the heat of reaction? ___________________________________________________________________ 5 Fill in the answers to the following questions about the energy profile diagram. (a) What is the amount of the potential energy of the products? ________ (b) What is the amount of the potential energy of the reactants? ________ (c) Which letter represents the potential energy of the activated complex? ________ (d) Which letter represents the activation energy for the reverse reaction? ________ (e) Which letter represents the activation energy for the forward reaction? ________ (f) If the potential energy of the reactants is 100.0 kJ and the potential energy of the products is 300 kJ, what is the heat of the forward reaction (Hfwd)?________ (g) Is the reverse reaction exothermic? ________ (h) If a catalyst was added to the reaction, which lettered quantities would change? (List all that apply.) ___________________________________________________________________ (i) If a catalyst was added to the reaction, what would happen to the heat of reaction? ___________________________________________________________________ 6