* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download outline3514
Survey
Document related concepts
Transcript
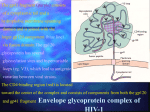
I. Introduction: over 20 years ago … A. B. C. D. E. II. Kaposi’s sarcoma and Pneumocystis carinii pneumonia – June 1981 MMWR At-risk groups in the U.S. – early 1980’s AIDS = acquired immunodeficiency syndrome; acronym from 1982 Retrovirus isolated in 1983 HIV’s introduction in the U.S. back-dated to 1978 or earlier Introduction: over 10 years ago … A. Death toll in the U.S. passed 100,000 but still considered a “niche” disease B. WHO predicts up to 40 million HIV infections by the year 2000 III. Worldwide epidemiology of HIV/AIDS A. B. C. D. E. F. IV. “Statistics are people with the blood and tears dried off …” Number of infections currently 40 million and over 60 million to date Number of fatalities to date 21.8 million By regions: sub-Saharan Africa, Asia, Latin America, North America Other countries: Russia (Ukraine), India, China Causes of death vary widely (infections in Africa vs. liver/blood/kidney in US) Pathogenesis of HIV infection A. Zoonosis by Pan troglodytes troglodytes chimpanzee in Africa 1. Mother virus of HIV resides in chimps, makes the “jump” to humans 2. Infections remain rural but spread to urban areas 3. Epidemics: horizontal (sex or needles) and vertical (mother-to-child) B. Risk of transmission routes 1. Behaviors at risk (sexual contact, needle sharing, mother-to-child, blood) 2. Probability of infection by type of exposure 3. Viral load of infected source and genital disease significantly alter risks C. HIV infection of host cells: binding by HIV at 2 sites on host cell, followed by viral enzyme activity (reverse transcriptase, integrase, and protease) D. Immunologic processing and dissemination of HIV into lymph nodes after infection E. Host’s immune response to HIV infection 1. CD4 T helper cells stimulate production of IL-2 and cytokines; establishes CD4 helper cells specific for future anti-HIV activity plus CD8 (killer) cells HIV infects the very cells that orchestrate an immune response against it HIV replicates in CD4 cells (free virus) and in lymph tissue Balance between production of HIV and destruction of infected cells F. Viral load averages 10 billion viruses produced daily 1. Measurements by polymerase chain reaction or branched DNA 2. Viral load must be measured in log units 3. Viral load reflects the speed of CD4 cell destruction and progression to AIDS G. AIDS is the culmination of a continuous, progressive pathogenic process 1. Primary infection, chronic/asymptomatic phase, symptomatic phase with opportunistic infections and neoplasms 2. Average time to severe immunosuppression after infection is 10-11 years H. CDC definitions: 1987 (25 indicator conditions), 1993 (CD4 count < 200, HIV ) 2. 3. 4. V. Introduction to antiretroviral therapy A. Suppress viral replication as low as possible using combinations of drugs B. Nucleoside reverse transcriptase inhibitors (NRTIs) mimic nucleoside components of viral DNA, are inserted into viral DNA chain and prevent reverse transcription C. Non-nucleoside reverse transcriptase inhibitors (NNRTIs) alter the structure of virus- associated reverse transcriptase and render the expanding viral DNA non-functional D. Protease inhibitors (PIs) block the catalytic site of HIV-associated protease and render new viral progeny non-infectious VI. Nucleoside reverse transcriptase inhibitors (NRTIs) Zidovudine, AZT; RetrovirTM Didanosine, ddI; VidexTM Zalcitabine, ddC; HividTM Stavudine, d4T; ZeritTM Lamivudine, 3TC; EpivirTM Zidovudine + lamivudine fixed combination; CombivirTM Abacavir, ABC; ZiagenTM Zidovudine + lamivudine + abacavir fixed combination; TrizivirTM Tenovofir disoproxil fumarate; VireadTM Any NRTI is insufficiently strong when used singly; evolutionary pressure selects for viral mutants that resist the drug (survival of the fittest) K. NRTI pairs without overlapping toxicity: d4T/3TC, AZT/3TC, d4T/ddI, AZT/ddI A. B. C. D. E. F. G. H. I. J. VII. Non-nucleoside reverse transcriptase inhibitors (NNRTIs) A. B. C. D. Nevirapine; ViramuneTM Delavirdine; RescriptorTM Efavirenz; SustivaTM NNRTI group considerably stronger than NRTI but resistance occurs quickly if viral suppression is incomplete VIII. Protease inhibitors (PIs) A. B. C. D. E. F. G. H. IX. Saquinavir hard-gel; InviraseTM Saquinavir soft-gel; FortovaseTM Ritonavir capsules, liquid, soft-gel; NorvirTM Indinavir; CrixivanTM Nelvinavir; ViraceptTM Amprenavir; AgeneraseTM Lopinavir + ritonavir fixed combination; KaletraTM Combination therapy with PIs is complex, due to short drug half-lives, difficult food requirements, timing of doses, and toxicity (GI and metabolic abnormalities) Recommended regimens for highly active antiretroviral therapy (HAART) – released by Department of Health and Human Services (DHHS) 2/01 A. B. C. D. E. F. G. H. Indinavir, nelfinavir, or KaletraTM plus one pair of compatible NRTIs Efavirenz plus AZT/3TC (or other compatible NRTI pair) Saquinavir (HGC or SGC) plus low-dose ritonavir plus compatible NRTI pair Indinavir or amprenavir plus low-dose ritonavir plus compatible NRTI pair Abacavir plus AZT plus 3TC (TrizivirTM) if viral load < 100,000 Amprenavir, saquinavir-SGC, or ritonavir plus compatible NRTI pair Nevirapine or delavirdine plus compatible NRTI pair Choices A. thru D. preferred over E. thru G. due to potency and/or convenience X. Initiating treatment in HIV seropositive patients, DHHS 2001 Symptomatic patient (AIDS or severe symptoms of fever, weight loss) – TREAT Asymptomatic patient with AIDS (CD4 < 200) – TREAT Asymptomatic patient, 200 CD4 350 – OFFER TREATMENT Asymptomatic patient, CD4 > 350 and elevated viral load (branched DNA > 30,000 or polymerase chain reaction > 55,000) – OFFER TREATMENT due to viral load E. Asymptomatic patient, CD4 > 350 and lower viral load (branched DNA < 30,000 or polymerase chain reaction < 55, 000) – FOLLOW PATIENT F. Based on risk of progression, benefits/risks of therapy, and patient willingness A. B. C. D. XI. Initiating treatment in HIV seropositive patients, International AIDS Society USA, 2002 version A. CD4 < 350, regardless of viral load – RECOMMEND TREATMENT B. CD4 350-500, regardless of viral load – CONSIDER TREATMENT C. CD4 < 500 – DEFER TREATMENT unless viral load very high XII. Starting HAART in the asymptomatic patient A. Benefits: regimens are more durable, short-term side effects are more tolerable, fewer viral mutations have occurred, regimens most successful at higher CD4 counts B. Risks: unknown long-term side effects, reduced quality of life, high cost, limited options if treatment fails (only 3 unique combos available without cross-resistance) XIII. Mutations and HIV resistance A. Mutations occur in HIV genes coding for reverse transcriptase or protease enzymes B. Resistance results from frequent mutations, error-prone reverse transcriptase, prolific levels of viral replication, and evolutionary pressure selecting for resistant viruses C. About 50% of HAART regimens are unsuccessful in “real life” due to inability to achieve suppression of viral replication caused by previous sequential monotherapy, low CD4 count at baseline, high viral load at baseline, poor adherence to treatment D. Resistance testing: genotypic or phenotypic methods XIV. Impact of HAART on AIDS healthcare A. Immune reconstitution causing increases in CD4 numbers (both memory and naïve cells) and improvements in cell-mediated immunity B. Opportunistic infections have dropped over 50%, with 75% reductions in Pneumocystis, Mycobacterium avium complex, and cytomegalovirus retinitis C. Drug-related toxicities due to protease inhibitors include fat redistribution, lipid abnormalities, insulin resistance, diabetes, and possible increase in MI risk D. Drug-related toxicities due to NRTIs include focal fat loss from extremities and face XV. New drug strategies in the future A. B. C. D. E. F. Once-daily regimens (depend on efavirenz or ritonavir-boosting of PIs) Protease inhibitors: atazanavir (ZrivadaTM) and tipranavir NNRTI: TMC 125, both very potent and effective against NNRTI-resistant HIV Fusion inhibitors (T-20, T-1249) inhibit fusion of HIV with host cell membrane CCR5 binding inhibitors (SCH-C, SCH-D) prevent HIV binding at chemokine site Integrase inhibitors (L-7088906 and L-731988) inhibit HIV integrase enzyme XVI. Immunologic strategies A. Interleukin-2 to improve CD4 counts B. C. D. E. F. Switching from PI-based regimens to NNRTI-based or triple NRTI regimens Structured treatment interruptions to improve host’s anti-HIV immunity Structured intermittent therapy (on/off cycles) to cost and improve quality of life Pulsed therapy = stopping therapy until reaching a predetermined CD4 or viral load Immunologic vaccines to stimulate either anti-HIV CD4 cells or killer (CD8) cells XVII. Worldwide strategies A. B. C. D. E. F. Education, STD prevention, condom distribution Vaccines Reducing HIV transmission at birth Increasing availability of HAART in developing countries Reducing mortality from TB and common bacterial infections Global AIDS fund XVIII. Cytomegalovirus retinitis update A. Valganciclovir (ValcyteTM) = IV ganciclovir, same toxicities, oral doses bid/qd B. Discontinuing anti-CMV retinitis therapy with HAART-induced immune recovery C. Immune recovery uveitis (iritis, vitritis, CME, ERM, etc.) XIX. In-office precautions: handwashing, gloves, instrument disinfection XX. Conclusions