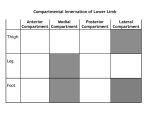
* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Solution Tutorial 4 - Aerospace Engineering, IIT Madras
R-value (insulation) wikipedia , lookup
Insulated glazing wikipedia , lookup
Heat capacity wikipedia , lookup
Heat transfer wikipedia , lookup
Thermoregulation wikipedia , lookup
Temperature wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
First law of thermodynamics wikipedia , lookup
Calorimetry wikipedia , lookup
Heat equation wikipedia , lookup
Dynamic insulation wikipedia , lookup
Van der Waals equation wikipedia , lookup
Thermal conduction wikipedia , lookup
Heat transfer physics wikipedia , lookup
Atmospheric convection wikipedia , lookup
Internal energy wikipedia , lookup
Second law of thermodynamics wikipedia , lookup
Thermodynamic system wikipedia , lookup
Equation of state wikipedia , lookup
History of thermodynamics wikipedia , lookup
TUTORIAL – 4 Solution (08/02/2017) Thermodynamics for Aerospace Engineers (AS1300) Temperature and Heat 1. The following table gives data, in kJ, for a system undergoing a thermodynamic cycle. Determine (a) the missing table entries and (b) whether the cycle is work producing or absorbing. Process ΔU Q W 1-2 ? 0 -610 2-3 670 ? 230 3-4 ? 0 920 4-1 -360 ? 0 In this question, the above blanks can be filled by using the first law of thermodynamics. The equation version of the first law of thermodynamics is as follows: 𝑄 = ∆𝑈 + 𝑊 [Eqn 1.1] where ΔU is the change in the internal energy of the system while changing its state. Here, W is the work done and Q is the heat given to the system. Always remember the sign conventions: The heat absorbed by the system: Positive sign (+ve) The heat released by the system: Negative sign (-ve) Work done by the system: Positive (+ve) Work done on the system: Negative (-ve) Using the above sign conventions and the Eqn 1.1 the blanks in the above table can be filled. For example in the 2nd row of the table Q=0, W=-610 kJ. The calculations is as follows: ∆𝑈 = 𝑄 − 𝑊 = 0 − (−610) = 𝟔𝟏𝟎 𝒌𝑱 (Row 2) 𝑄 = ∆𝑈 + 𝑊 = 670 + 230 = 𝟗𝟎𝟎 𝒌𝑱 (Row 3) ∆𝑈 = 𝑄 − 𝑊 = 0 − 920 = −𝟗𝟐𝟎 𝒌𝑱 (Row 4) 𝑄 = ∆𝑈 + 𝑊 = −360 + 0 = −𝟑𝟔𝟎 𝒌𝑱 (Row 5) So, finally the table becomes: Process ΔU Q W 1-2 610 0 -610 2-3 670 900 230 3-4 -920 0 920 4-1 -360 -360 0 As this is a cyclic process, the net internal energy change (ΔUnet) is 0 which can be shown by summing the ΔU values of all the steps in the cycle. So the net heat absorbed by the system is equal to the net work done by the system. The summation of work done in all the steps of the cyclic process gives 540 kJ which suggests that net work is done by the system. So, this cycle is producing work. (Answer) 2. One kg of Air expanded in piston-cylinder system from a specific volume v = 0.2 m3/kg and temperature of 580 K to a specific volume v = 0.8 m3/kg and a temperature of 290 K. The expansion process can be assumed to follow pv1.5=0.75 (with p in bar, and v in m3/kg). Determine the work and heat interaction. Assume specific heat at constant volume as 718 J/kg/K. In this, it has been asked to find the heat transferred to the system and the work done. The sign convention for heat and work done is provided in question no. 1. The state equation for the expansion process is given as follows: 𝑝𝑣 1.5 = 0.75 , where p is in bar, and v is in m3/kg [Eqn 2.1] If we convert bar to Pa (Pascal), the equation becomes, 𝑝𝑣 1.5 = 75000 , where p is in Pa [Eqn 2.2] Here p can be written in terms of v which will be useful for calculating workdone 𝑝= 75000 𝑣 1.5 , where p is in Pa [Eqn 2.3] So, work done can be calculated as follows: 0.8 𝑊 = (∫0.2 𝑝𝑑𝑣 ) ∗ 𝑚𝑎𝑖𝑟 , as the unit of ∫ 𝑝𝑑𝑣 is J/kg 0.8 75000 𝑊 = (∫0.2 𝑣 1.5 75000 0.8 𝑑𝑣) ∗ 𝑚𝑎𝑖𝑟 = (− 0.5𝑣0.5 = −150000 ∗ ( 1 √0.8 − 1 0.2 [Eqn 2.4] )∗1 ) ∗ 1 = 167720 𝐽 = 𝟏𝟔𝟕. 𝟕𝟐𝟎 𝒌 √0.2 The change in internal energy of the system is as follows: ∆𝑈 = 𝑚𝑝 ∗ 𝐶𝑣 ∗ (𝑇2 − 𝑇1 ) = 1 ∗ 718 ∗ (290 − 580) = −208220 𝐽 = −𝟐𝟎𝟖. 𝟐𝟐𝟎𝒌𝑱 So the heat interaction of the system calculated using the first law of thermodynamics as follows: 𝑄 = ∆𝑈 + 𝑊 = −208.220 + 167.720 = −𝟒𝟎. 𝟓 𝒌𝑱 (Answer) [Eqn 2.5] As the sign of the heat interaction is negative which suggest heat is released or given out by the system. 3. Consider the insulated vessel, with compartment A (initially evacuated) of volume 0.5 m3 and separated by a thin membrane from compartment B of volume 0.25 m 3 which contains 2 kg of a pure substance in compartment B is stirred by a fan until the membrane ruptures. The membrane is designed to rupture at a pressure of 2MPa. Determine (a) the temperature in B when the membrane ruptures (b) the work done by the fan (c) the final equilibrium temperature and pressure when the substance fills the entire vessel. Given 𝑉𝐵 = 0.25 m3, 𝑉𝐴 = 0.5 m3, mass of the substance in compartment B, 𝑚 = 2 kg, 𝐶𝑣 = 720 J/kg/K, 𝑃𝑣 = 286 𝑇 and initial pressure 𝑃𝐵1 = 700 kPa. The membrane ruptures when pressure in compartment B becomes 𝑃𝐵2 = 2 MPa. Before we solve the problem, we must clarify some conventions. Usually ideal gas law is written as 𝑃𝑉 = 𝑛𝑅𝑢 𝑇, where 𝑃 is pressure, 𝑉 is the volume, 𝑛 is the total number of moles, 𝑅𝑢 is the universal gas constant and 𝑇 is the temperature. Now, we can rewrite this equation as 𝑚 𝑃𝑉 = 𝑀 𝑅𝑢 𝑇, where 𝑚 is the mass of the substance and 𝑀 is the molecular mass of the substance. Define specific gas constant as 𝑅 = 𝑅𝑢 ⁄𝑀 and specific volume as 𝑣 = 𝑉 ⁄𝑚. Rewriting the ideal gas law in terms of 𝑅 and 𝑣 yields 𝑃𝑣 = 𝑅𝑇 This is the form of ideal gas law that you will be dealing with in many future courses. Note that 𝑣 = 1⁄𝜌, where 𝜌 is the density. So, this equation can also take the form 𝑃 = 𝜌𝑅𝑇. In this question, the specific gas constant 𝑅 = 286. (a) The fan does work on the gas and the pressure of the gas in B increases while the volume remains constant. We need to find the temperature of the gas when the final pressure is 𝑃𝐵2 = 2 MPa. Using the ideal gas law, evaluate the final temperature 𝑇𝐵2 = 𝑃𝐵2 𝑉𝐵 ⁄𝑚𝑅 = 𝟖𝟕𝟒 K. (Answer) (b) The work done by fan is used entirely to build the internal energy of the gas since the walls are insulated (∴ 𝑄 = 0) and volume of gas is constant (∴ 𝑊𝑔 = 0). Using the available data, evaluate the initial temperature in compartment B, 𝑇𝐵1 = 305.97 K. Apply first law, ∆𝑈 = 𝑄 − 𝑊 = −𝑊𝑔 − 𝑊𝑓 = −𝑊𝑓 Therefore, 𝑊𝑓 = −∆𝑈 = −𝑚𝐶𝑣 (𝑇𝐵2 − 𝑇𝐵1 ) = −818 kJ. The work done by the fan = −𝑊𝑓 = 𝟖𝟏𝟖 kJ. (Answer) (c) When the membrane breaks, the gas expands and fills the total volume 𝑉 = 𝑉𝐴 + 𝑉𝐵 = 0.75 m3. Consider A and B together as the system. Total volume of this system is constant (∴ 𝑊 = 0) and the walls of the system are insualted (∴ 𝑄 = 0). Therefore, the internal energy of the system remains constant during the process. Thus, the final temperature of the system, 𝑇𝑓 = 𝑇𝐵2 = 874 K. Using the ideal gas law, evaluate the final pressure, 𝑃𝑓 𝑃𝑓 = 𝑚𝑅𝑇𝑓 ⁄𝑉 = 𝟔𝟔𝟔𝟓𝟕𝟎. 𝟔𝟔 Pa. (Answer) 4. 5 kg of steam is contained within a piston-cylinder assembly. The steam undergoes an expansion process from state 1, where the specific internal energy is 2709.9 kJ/kg to state 2, where the specific internal energy is 2659.6 kJ/kg. During this process 80 kJ of heat is transferred to the steam and a paddle wheel transfers 18.5 kJ of work. Determine the work done by the steam on the piston. Given mass of steam, 𝑚 = 5 kg, initial specific internal energy 𝑢1 = 2709.9 kJ/kg, final specific internal energy 𝑢2 = 2659.6 kJ/kg, heat transferred, 𝑄 = 80 kJ and paddle work, 𝑊𝑝 = −18.5 kJ. Let work done by steam be 𝑊𝑠 . Applying first law, ∆𝑈 = 𝑄 − 𝑊 = 𝑄 − (𝑊𝑝 + 𝑊𝑠 ) ==> 𝑚(𝑢2 − 𝑢1 ) = 𝑄 − (𝑊𝑝 + 𝑊𝑠 ) Substitute the values and solve for 𝑊𝑠 to obtain 𝑊𝑠 = 𝟑𝟓𝟎 kJ. (Answer) 5. 1 kg of air, initially at 5 bar, 350 K and 3 kg of CO2 initially at 2 bar, 450 K are confined to opposite sides of a rigid, well insulated container as shown in the figure. The partition which is initially held in place by a pin is thermally conducting and free to move. The pin is now removed and the gases are allowed to come to equilibrium. Determine the final temperature and the final pressure. Assume air and CO2 to be pure substances governed by Pv=288.68T, Cv=733 J/kg/K and Pv=189T, Cv=750 J/kg/K respectively, where P is in Pa, v is in m3/kg and T is in K. Given: Mass of air, 𝑚𝑎 = 1 kg, initial pressure of air, 𝑃𝑎 = 5 bar, initial temperature of air, 𝑇𝑎 = 350 K, mass of CO2, 𝑚𝑐 = 3 kg, initial pressure of CO2, 𝑃𝑐 = 2 bar, and initial temperature of CO2, 𝑇𝑐 = 450 K. Also, it is given that for air, 𝑃𝑣 = 288.68 𝑇, 𝐶𝑣,𝑎 = 733 J/kg.K and for CO2, 𝑃𝑣 = 189 𝑇, 𝐶𝑣,𝑐 = 750 J/kg.K. Initial volume of air 𝑉𝑎,𝑖 = 288.68 𝑚𝑎 𝑇𝑎 ⁄𝑃𝑎 = 0.20207 m3. Initial volume of CO2, 𝑉𝑐,𝑖 = 189 𝑚𝑐 𝑇𝑐 ⁄𝑃𝑐 = 1.27575 m3. When the pin is removed, the partition moves in such a way that it reaches a final equilibrium state where the temperature 𝑇𝑓 (due to conducting partition) and pressure 𝑃𝑓 of both gases will be the same. This corresponds to thermal and mechanical equilibrium. Consider (air + CO2) as the combined system. The system is insulated and the total volume is a constant. Applying first law, ∆𝑈 = 𝑄 − 𝑊 = 0. Thus, ∆𝑈 = ∆𝑈𝑎 + ∆𝑈𝑐 = 0. It implies that 𝑚𝑎 𝐶𝑣,𝑎 (𝑇𝑓 − 𝑇𝑎 ) + 𝑚𝑐 𝐶𝑣,𝑐 (𝑇𝑓 − 𝑇𝑐 ) = 0. Solving the above equation yields 𝑻𝒇 = 𝟒𝟐𝟓. 𝟒 K. (Answer) The next condition to be satisfied is that the total volume of the system (air + CO2) is a constant i.e., 𝑉𝑎,𝑓 + 𝑉𝑐,𝑓 = 𝑉𝑎,𝑖 + 𝑉𝑐,𝑖 = 1.477826 m3. Substitute for 𝑉𝑎,𝑓 (= 288.68 𝑚𝑎 𝑇𝑓 ⁄𝑃𝑓 ) and 𝑉𝑐,𝑓 (= 189 𝑚𝑐 𝑇𝑓 ⁄𝑃𝑓 ) in terms of 𝑃𝑓 . Then, solve for 𝑃𝑓 to obtain 𝑷𝒇 = 𝟐𝟒𝟔𝟑𝟏𝟐 Pa. (Answer) 6. 4 Kg of air is contained in a vertical piston cylinder with the top open to atmosphere. The piston weighs 50 kg and has a face area of 0.01 m2. The air initially occupies a volume of 0.005 m3 . The air now undergoes a process wherein its volume deceases to 0.0025 m3 and 1.41 kJ of heat is lost to the surroundings. Determine the change in specific internal energy of the air. Take the atmospheric pressure to be 100 KPa. Given: Mass of air inside the cylinder, 𝑚𝑎 = 4 kg, mass of piston, 𝑚𝑝 = 50 kg, area of piston, 𝐴𝑝 = 0.01 m2, initial volume occupied by air, 𝑉1 = 0.005 m3, final volume, 𝑉2 = 0.0025 m3, atmospheric pressure, 𝑃𝑎𝑡𝑚 = 100 kPa and heat interaction, 𝑄 = −1.41 kJ. Pressure inside the cylinder, 𝑃 = 𝑃𝑎𝑡𝑚 + 𝑚𝑝 𝑔⁄𝐴𝑝 = 149050 Pa. Pressure at any instant inside the cylinder will be the same value 𝑃 = 149050 Pa, so that the system is always at equilibrium. Therefore work interaction for air inside cylinder, 𝑊 = 𝑃(𝑉2 − 𝑉1 ) = −372.625 J. From first law, change in internal energy of air, ∆𝑈 = 𝑄 − 𝑊 = −1037.375 J. Thus, change in specific internal energy ∆𝑢 = ∆𝑈⁄𝑚𝑎 = −𝟐𝟓𝟗. 𝟑𝟒 J/kg. (Answer)