* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Journal Club - WordPress.com
Survey
Document related concepts
Transcript
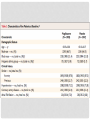
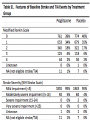
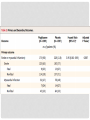
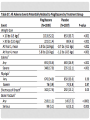
JOURNAL CLUB ANGELA AZIZ DONNELLY APRIL 5, 2016 PIOGLITAZONE AFTER ISCHEMIC STROKE OR TRANSIENT ISCHEMIC ATTACK NEJM FEB 17, 2016 IRIS Insulin Resistance Intervention after Stroke Hypothesis: Pioglitazone would reduce the rates of stroke and myocardial infarction after ischemic stroke or TIA in select patients BACKGROUND • Patients affected by stroke and TIA are at risk for another event • Current treatment for secondary stroke prevention is fairly limited • Explores insulin resistance as a possible new treatment target BACKGROUND • Many proposed mechanisms for why the presence of insulin resistance increases risk for vascular disease • There are various strategies to improve insulin sensitivity – including lifestyle modification, TZDs, and other medications (eg. metformin) RESEARCH METHODOLOGY • International, randomized, double-blind, placebocontrolled clinical trial • Eligible Patients: Men and Women at least 40 years of age who had a qualifying ischemic stroke or TIA during the previous 6 months AND met criteria for insulin resistance RESEARCH METHODOLOGY • Exclusion criteria • Patients with diabetes • Patients with class 3 or 4 heart failure, or patients with class 2 heart failure with reduced EF • Pregnancy • Moderate/severe pitting edema • Carotid revascularization within 14 days • Use of an estrogen-containing contraceptive or oral glucocorticoid • Temporary Exclusion • Abnormal liver function • Severe anemia RESEARCH METHODOLOGY • Patients then randomly assigned in a 1:1 ratio to receive either pioglitazone or matching placebo • Pioglitazone was titrated from an initial dose of 15 mg to a final dose of 45 mg if no side effects encountered • If patient’s had side effects, they were treated according to algorithms • Adherence was monitored by asking about drug use and performing pill counts • Patient’s individual primary care doctors had direct responsibility for providing best current medical care for risk factor modification TRIAL OUTCOMES Primary outcome • First fatal or nonfatal stroke or fatal or nonfatal MI Secondary outcomes • Recurrent stroke • Acute MI or unstable angina • The composite of stroke, MI, or HF resulting in hospitalization or death • All-cause mortality • Progression to overt diabetes • Cognitive decline from baseline STATISTICAL ANALYSIS • Analyses performed on an intention-to-treat basis • The primary outcome and all secondary outcomes (except cognitive function) were analyzed by means of the time-to-first-event method FINDINGS CONCLUSION Good study design Multi-center Double-blind Randomized controlled trial Large sample size 5 years Groups similar at baseline with similar control of comorbidities between groups Included the patients that would benefit the most from additional secondary stroke prevention (Low NIHSS/mRS) CONCLUSION • Strict inclusion/exclusion criteria – eg. No diabetes, no heart failure • Majority of population was white, male • Used HOMA-IR for measuring insulin resistance – how would we do this in clinical practice • No comparison to lifestyle modifications DISCUSSION/IMPLICATIONS • Can we apply this study to our current patient population? • Given risks versus benefits, would you use this in clinical practice? • Impressive NNT of 35. How does this compare to our current standard of care, eg. treatment with high intensity statin