* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Valence Bond Theory
Survey
Document related concepts
Transcript
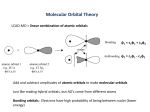
How does bonding take place with orbitals described by Quantum Theory? Learning Goals Students will be able to: 1) understand how bonding occurs with respect to the Quantum Mechanical Model 2) understand the concept of Valence Bond Theory Success Criteria Student will show their understanding by: 1) explaining how half-filled orbitals from two different atoms overlap to form covalent bonds 2) explaining how hybrid orbitals (sp, sp2, sp3 ,sp3d, sp3d2) are needed to explain bonding with atoms such as carbon. 3) explaining how double and triple bonds are formed. Pre-requisite Knowledge Student will already know: 1) shapes of orbitals (Quantum Mechanical Model) 2) Energy Diagrams Valence Bond Theory Linus Pauling was a long-time friend of Gilbert Lewis. He dedicated a famous textbook, The Nature of the Chemical Bond, to Lewis. Pauling is one of only four two-time winners of a Nobel Prize—his in two different fields, Chemistry (1954) and Peace (1962). The orange is part of a personal experiment in which he believed that high doses of vitamin C could rid the body of free radicals (ions in the body) that would damage tissues and lead to aging. His quest for longer life was fairly successful he lived to 93. Quantum Mechanical Model We have seen how Quantum Mechanical Theory has changed how we think of electron orbitals. The Bohr-Rutherford model that we learned in Grade 9 and 10 has been replaced. Orbitals are now regions where the electron has a probability of occurring – more like a cloud where the electron can exist. Now we have to move from physics to chemistry! s, p, d, and f orbitals Simpler Bohr-Rutherford Diagram Bonding & the Quantum Mechanical Model We need to talk about how elements actually bond. The Bohr-Rutherford model helped us explain bonding and the combining capacities of elements (for example Ca wants to give away 2 electrons and Cl wants to gain 1 electron) Now we are interested in a more accurate picture of bonding How are electrons shared between elements (and these odd shaped orbitals)? How can we use this knowledge to infer the shapes of molecules According to valence bond theory, a covalent bond is formed when two orbitals overlap (share the same space) to produce a new combined orbital containing two electrons of opposite spin. This arrangement results in a decrease in the energy of atoms forming the bonds. Great Videos: http://www.youtube.com/watch?v=vVEqx9gSl3Y Valence Bond Theory I: Intro to Valence Bond Theory – Ben’s Chem Videos (start here) http://www.youtube.com/watch?v=mTHW9W-2-N8 sp3, sp2, and sp Hybridization – Ben’s Chem Videos Figure 1: The formation of a single covalent bond in a hydrogen molecule by the overlap of two 1s orbitals of individual atoms. The two shared electrons spend most of their time between the two hydrogen nuclei. This represents a new, lower-energy state of the two atoms. Notice that the new combined orbital contains a pair of electrons of opposite spin, just like a filled orbital. Any two half-filled orbitals can overlap in the same way. The total number of electrons in the bonding orbital must be two. Figure 2: Consider hydrogen fluoride, a hydrogen atom has only one occupied orbital, the 1s orbital. The hydrogen 1s orbital is believed to overlap with the halffilled 2p orbital of the fluorine atom to form a covalent bond. This approach can also be used for larger molecules. An oxygen atom has two half-filled p orbitals. It is reasonable to propose that the 1s orbitals of the two hydrogen atoms overlap with the two half-filled 2p orbitals of the oxygen atom to produce a stable, lower-energy state. Figure 3: The two covalent bonds are created by two sets of combined s-p orbitals. The measured angle be the H-O-H bond is about 105° - this is explained by additional manipulation of atomic orbitals and a consideration of the repulsions between pairs of electrons. Overall, when atoms bond, they arrange themselves in space to achieve the maximum overlap of their halffilled orbitals. Maximum overlap produces a bonding orbital of lowest energy. Problems with Lewis Bonding Theory Problem 1 Could not explain the four equal bonds represented by the four pairs of electrons in a carbon compound like methane, CH4(g) Problem 2 Could not explain the existence of double and triple bonds. Solution to Problem 1 Explaining equal bonds in methane Pauling and others created the idea of electron promotion from an s to an empty p orbital Experimental evidence indicated that the electron orbitals were equivalent in shape and energy The four bonds for carbon in molecules such as methane are explained by hybridization to four identical sp3 atomic orbitals Great Video: Molecular Shape and Orbital Hybridization https://www.youtube.com/watch?v=d1E1 8tBTlBg&feature=related (10:09) The two 2s electrons and the two 2p electrons form four sp3 hybrid orbitals with one bonding electron in each This explains the bonding capacity of four for carbon These orbitals are hybridized only when bonding occurs to form a molecule; they do not exist in an isolated atom. 16 Figure 4: (a) An s electron is promoted to an empty p orbital in a carbon atom. (b) The four orbitals are combined to produce four hybrid sp3 orbitals. (c) Each sp3 orbital is equivalent in energy and shape. Electron repulsion requires that the orbitals are as far apart as possible – pointing to the corners of a regular tetrahedron. Pauling suggested that there were a whole series of hybridizations that could occur Solution to Problem 2 - Explaining Double and Triple Covalent Bonds Experimental evidence determined that three substances can be formed when two carbons atoms bonded with hydrogen – C2H6(g), C2H4(g), and C2H2(g). Lewis suggested that between the carbon atoms there must a sharing of one, two, and three electron pairs in order to obtain a stable octet around the carbon atoms. How is it possible that electrons in what we would predict as being sp3 hybrid orbitals could overlap not once, but twice or three times with just one other atom? According to the valence bond theory, two kinds of orbital overlap are possible: (1) The end-to-end overlap of s orbitals, p orbitals, hybrid orbitals, or some pair of these orbitals. This type of overlap produces a sigma (σ) bond. Think of sigma bonds as the usual single covalent bonds that you are used to drawing in structural diagrams. Figure 5: Sigma bonds form with the overlap of (a) s orbitals (b) p orbitals and (c) hybrid orbitals. (2) Two orbitals can overlap side by side to form a pi (π) bond. Pi bonds are the second and third lines in the structural diagrams for double and triple covalent bonds. Figure 6: P orbitals form with the side-byside overlap of orbitals. Double Bonds We have already seen that the orbitals of a carbon atom can be hybridized to form four sp3 hybrid orbitals. To explain double bonds – The key new idea is a partial hybridization of the available orbitals leaving one or two p orbitals with single unpaired electrons. Double Bond Example – Ethene (C2H4(g)) Suppose that after promoting an electron in carbon’s 2s orbital to a 2p orbital, we form three sp2 hybrid orbitals leaving one p orbital with a single electron. Still have four orbitals to form bonds but three of these are hybrids and one is a “normal” p orbital. Figure 7: Instead of mixing all four orbitals, valence bond theory suggests that only three are mixed to form sp2 hybrid orbitals and a unhybridized p orbital for a carbon atom Figure 8: For this carbon atom, the sp2 hybrids are planar at 120° to each other and the p orbital is at right angles to the plane of the hybrid orbitals. In a molecule of ethene, the three hybrid orbitals are used to form sigma bonds between the carbon atoms and to the hydrogen atoms. Figure 9: (a) The sigma bond for a ethene molecule use the sp2 hybrid orbitals. The half-filled p orbitals on each carbon are believed to overlap sideways to form a pi bond. Figure 9: (b) The two half-filled p orbitals of the adjacent carbon atoms overlap sideways. Notice that the pi bond is a region of electron density appearing above and below the sigma bond directly joining the two carbon atoms. Figure 9: (b) The two half-filled p orbitals of the adjacent carbon atoms overlap sideways. A pi bond is a combined orbital containing a pair or electrons of opposite spin. Figure 9: (b) The two half-filled p orbitals of the adjacent carbon atoms overlap sideways. The additional shared pair of electrons in the pi bond provides greater attraction to the two carbon nuclei, which explains why the double covalent bond is shorter and stronger than a single bond. Figure 9: (c) The complete bonding orbitals for a ethene molecule. The Triple Bond Learning Checkpoint IN SUMMARY - we have a theory to explain how covalent bonding occurs but more intriguingly, we have a theory that can explain the SHAPES OF MOLECULES (the concept to be explored next class) REVIEW: Read p. 239 and explaining how a triple bond forms in ethyne (using the valence bond theory) Assigned Questions p. 232 Practice UC # 1, 2, 3, 4 p. 235 Practice UC # 8, 9, 10, 11 p. 239 Practice UC # 18, 19, 21 36