* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Introduction to Geo-medicine
Mesothelioma wikipedia , lookup
Amanita phalloides wikipedia , lookup
Wittenoom, Western Australia wikipedia , lookup
Asbestos and the law wikipedia , lookup
Nitrogen dioxide poisoning wikipedia , lookup
Spodden Valley asbestos controversy wikipedia , lookup
Furthering Asbestos Claims Transparency (FACT) Act of 2015 wikipedia , lookup
Asbestos and the law (United States) wikipedia , lookup
Introduction to Geo-medicine
Module Name:
Introduction to Geo-medicine
Module Code: MEG 2643
Lecture Notes
• Lecturer: Ms Mundalamo
Department of Mining and Environmental Geology
School of Environmental Geology
University of Venda
Outline
•
•
•
•
•
•
•
•
•
•
•
•
•
•
•
What is Geomedicine? Definition and Concept
Periodic table of elements – Revision
Principal chemical constituents of the human body
Essential elements in life systems
Control on elemental intake
Toxic elements
Sources of toxic elements
Bioavailability of toxic elements to bioreceptors
Pathways of toxic elements in soils , plants and animal systems
Dose-response curves
The changing nature of disease
Health effects of excess/deficiency of selenium, lead. mercury, zinc, cadmium and arsenic
Investigation of casual links between specific diseases and the geological environment
Geogenic sources of atmospheric particulates
Dust emission in mining and mineral processing and the fate of inhaled particles
– Chronic Bronchitis
– Allergic lung disease
– Coal workers pneumoconiosis
– Asbestosis
– Cancer and mutagens
– Silicosis
Outline
• Geological aspects of waste disposal including radioactive
waste
• Ionization and radon gas
• Balneology of peat deposits
• Health spas and mineral waters
• Geochemical maps in medical geology
• Environmental data sources
• Medical data sources
• Data analysis using Pearson’s correlation
• Use of remote sensing and GIS in human health studies
• Analytical techniques in environmental Geochemistry
• Geochemical Data bases
Projects
• Berylliosis
Group A
• Talcosis
Group B
• Siderosis
Group C
• Geophagia
Group D
• Podoconiosis and
Kaposis’s sarcoma
Group E
References
• Author Komatina, Miomir M.
Medical geology : effects of geological environments on human
health Publisher Amsterdam, The Netherlands : Elsevier, 2004.
• Medical Geology : A regional Synthesis
• Closing the Gap
What is Geomedicine?
• - Geomedicine, often referred to as Medical Geology, is
upcoming discipline that links earth sciences and
medicine.
• - It is the science that investigates the effects of the
presence or absence of trace elements in organisms.
• - The discipline tries to establish casual links between
specific diseases in human, animals and plants and
geological processes and human activities in space
and time.
• - The study of geomedicine includes industrially
derived exposure to known toxic elements originating
from mining and mineral processing.
What is Geomedicine?
• - Geomedicine is a multidisciplinary subject, which requires
cooperation among geologists, clinicians, epidemiologists,
biologists, veterinarians etc.
• - The role of geologist is to help isolate geological aspects that
might influence the incidence of diseases.
• - Medical Geology has acquired its present status due partly to
inter–disciplinary approaches in solving scientific problems and
partly to advances in analytical methodology, enabling the
determination of nutritional trace elements at lower detection
levels (ppm/ppb)
• - Trace elements are important not only for their deficiency, but
also for their excess. Besides such elements such as As, F, Se, Pb, Zn,
Fe, Mo can cause severe consequences in circumstances of natural
chronic poisoning.
What is Geomedicine?
• - Both, Acute and Chronic toxicity have to be
considered.
• - Deficiencies, excess or imbalances in the supply of
inorganic elopements from dietary sources can have
an important influence on human and animal health.
• - Such situations arise mainly from anomalies in the
inorganic elements composition of food chains, which
are frequently attributed to the composition of the
geochemical environment as modified by the
influence of soil composition and bacterial variables.
Why this new discipline?
• Create awareness of, and concern for the environment during
the exploitation of natural resources
• Develop concepts and skills for assessing and resolving
environmental issues, questions and problems related to
mining and mineral processing
• Introduce a firm background in professional expertise and
environmental principles in research and development
activities in society, leading to improved methodologies and
techniques for mitigation
• Promote participation in practical environmental activities
with a view to enhancing environmental quality in the lives of
local communities.
Why this new discipline?
•
Improve communication amongst the various disciplines concerned
with diseases caused by geological factors which influence the well
being of man and minerals
•
Developed information material for the use of schools, publics and
private organisation interested in these fields to show the impact of
geologic factors on well being of man and animals.
•
Encourage geological surveys, universities, and geological societies to
take a more active role in providing useful information on geologic
conditions in Geoscience and health problems.
•
Encourage the development of local working groups of multidisciplinary Geomedicine, Geoscientists, Geo environment and health
expect
•
Encourage research in the area of producing more effective
methodologies for the study of geological factors in environment
medicine
Essential elements in life systems
Major elements
• Are those which are needed most in life system.
• More than 99% of human body is made of the
six major elements: O, C, H, N, Ca, and P.
• Human body is about 60% water. The rest:
• protein, fat, carbohydrates containing: O, C, H,
N, Ca, and P
• Teeth and bone: Ca, O, P, H
Major elements
• Dissolve salts: K, Na, Mg, Ca, Cl, Br, I, C, O, P
• Major elements of the human body are
obviously critical to life and health.
• Thus most part of the Geomedicine is the
study of the effects of the presence or
absence of the trace elements
Trace elements
• Trace elements are those found in concentrations
of about 10 to 100 ppm or less.
• The essential trace elements are required in small
amounts for proper functioning of various enzyme
system
• These elements can have quite different effects,
depending on the concentration in which it occurs
or consumed.
• That is effects such as acute or chronic toxicity.
Toxic elements
• Elements which have effects at very low concentration
• These elements often replace trace elements in the
body, but get stuck where they are and shut down the
essential function
• Example:
–
–
–
–
–
Cd, Hg: replace Zn, C u
Sr, Ba: replace Ca
As, Sb: replace P
Se, Te replace S
W replace Cr, Mo
Non essential elements
• These elements can be added to the body to
produce a beneficial effect
• The warning is that small amount is beneficial,
but too much is dangerous and can lead to
toxicity
Heavy metals/Trace elements
• Harmful elements known to have adverse
physiological significance at relatively low
levels include heavy metals such as
- As, Cd, Cr, Pd, Zn and Hg and
- also include the radioactive elements such
as U,Ra, Rn, Th, Es
• All trace elements are toxic if ingested or
inhaled at sufficiently high levels for long
enough periods of time
TOXICITY
• Toxicity is the degree at which chemical
substances have ability to induce biochemical
harmful effects to bio-receptors (living
organisms- plants, human, animals)
• Three categories of toxicity
– Acute toxicity
– Accelerated toxicity
– Chronic toxicity
• Acute toxicity
-is the effect of toxic when one
massive dose of toxic element is
taken from a single exposure or from
multiple exposure in a short period
of time
-Mainly occurs at less than 24 hours
-But still can be up to 14 days from
the days of taking the substances
• Accelerated toxicity
-it is the intermediate stage of toxic
effect after single or multiple
exposure in an intermediate
period of time
- Mainly occurs after exposed to
acute toxicity when no treatment
had encountered
• Chronic toxicity
– effects of exposure to small doses
over a long period of time
- Progressive effect of toxic elements
- Mainly effect becomes permanent
and can not be cured
Sources of toxic elements
• Natural and
• Anthropogenic Sources
Natural Sources
Accumulation of heavy metals due to natural sources is
through:
•
•
•
•
•
•
•
•
•
Chemical and physical weathering of rocks
Decomposition of plants, animals detritus
Precipitation
Volcanic activity
Wind erosion
Forest fire smoke
Oceanic spray
Floods
Earth quakes
Anthropogenic activities
Accumulation of heavy metals due to anthropogenic activities include
activities such as:
• metalliferous mining and smelting
• Agricultural and Horticultural materials
• Sewage sludges
• Fossils fuel combustion
• metallurgical industries, manufacture, use and disposal of metals
commodities
• electronics
• waste deposal
• metal, smelting ,
• corrosion of metals in use,
• forestry,
• agriculture,
• sports and leisure activities.
Natural sources:
• The movement of chemical compounds
among various path through the lithosphere,
hydrosphere, Atmosphere and biosphere
• These constitute the Geochemical circle
Geochemical
Circle
• The most common natural origin of trace
elements is the Geochemical origin- chemical
constituents from geological environment.
• Trace elements occurs as trace constituents of
primary minerals in igneous rocks –
crystallized magma
• They become incorporated into these minerals
by isomorphously substitution which is
governed by ionic charge, ionic radius and
electronegativity of the major elements and
trace element .
Trace constituent of rock forming minerals:
Mineral
Trace constituents
• Olivine
• Hornblende
• Biotite
Ni, Co, Mn, Li, Zn, Cu, Mo
Ni, Co, Mn, Li, V Zn Cu Ga
Rb, Ba, Ni, Co, Zn, Cu,
easily weathered
•
•
•
•
Mn, Cr, Ga
Rb, Ba, Sr, Cu, Ga
F, Rb, Ba, Sr, Cu, V
Zn Co, Ni, Cr, V
moderately stable
Garnet
Orthoclase
Muscovite
Magnetite
• Quarts
susceptibility to weathering
-
Very resistant
• Weathering of rocks will contribute on soils
contamination through processes such as
hydrolysis,
hydration,
dissolution,
oxidation and reduction,
ion exchange and
carbonation
• The trace element concentrations in sedimentary
rocks are depended upon the mineralogy and
adsorptive properties of the sedimentary
materials.
Mean heavy metal contents of major rock types:
Bioavailability of toxic elements
• This explains the FORM in which
elements are available for bioreceptors.
• The form in which the element might
have negative or positive impacts on
the environment
• The property of a substance that makes
its chemical uptake biota
• This can be defined as
•
•
•
•
•
Element Speciation
Distribution of an element among defined chemical
species in a system
Speciation – species distribution
Chemical species - chemical elements : specific form
of an element defined by its isotopic composition
Speciation analysis – analytical activities in analytical
chemistry of identifying and or measuring the
quantities of one or more individual chemical species
in a sample
This can be denoted as oxidation state and or
complex molecular structures.
• Which then influences the bioavailabity of
toxic elements
OXIDATION STATE
examples
As
As (III) and As (IV) are toxic
Cr
Cr (III) -- essential
Cr (VI) – highly toxic and cancer promoting
• Cu
• Ionic Cu (II)is toxic in aquatic systems
• It is the normal oxidation state for soluble
aqueous complexes
• It is stable at aqueous complexes
C(III)
Unstable in aqueous solution
Cu+ is classified as soft acid and forms
complexes with typical soft base metals
•
•
•
•
•
Fe
Fe (II) – less toxic
Absorption capacity is lower compared
to Fe(III)
Only Fe (II) is effective against
deficiency
Important for supplementation
But Fe (III) toxic
Hg
• Inorganic mercury salts are less
dangerous than methylated forms
• These are more toxic than inorganic
mercury
Inorganic Compounds & Complexes
• These are readily consumed in either liquid or
gas, especially water
• The distribution of an element among
different compounds affects its transport and
bioavailability by determining properties as
charge, solubility and diffusion coefficient.
;EXAMPLES 1 (Ni)
Ni salts of NIcl or Ni(SO4) are water soluble and of low
oral toxicity
Ni oxides or sulphur are highly insoluble in water, but
their bioavailability may be influenced by biological
legands. (A legands is a group of ions or molecule
coordinated to a central atom or a molecule in a
complex)
;Trinickel disulphide (Ni3S2) is an important carcinogen
in animals.
• Exposures are seldom to a single species of Ni.
For example, in various refining operations
workers may encounter N3S2, NiO, Ni, Ni-Cu
oxides, Ni-Fe oxides, NiSO4, Nicl2 and NiCO3
• Inmost natural waters (ph 5-9) the free
dominant Ni species are either the free Ni2+,
NiCO3, Ni(OH)2, NiSO4, Nicl2 and Ni (HN4)2.
• Under the reducing conditions the low
solubility of Ni sulphide may limit dissolved
nickel concouter
EXAMPLE 2 (Cu
In contrast to Ni, Cu can form stable compounds
in more than one oxidation state
Cu(ii) is the normal oxidation state for soluble
aqueous complexes,
• Although insoluble complexes of Cu(i) are also
stable.
• Cu(iii) complexes are relatively few in numbers
and are unstable in aqueous media.
• Cu(i) is classified as a soft acid and forms
complexes with typical soft bases- iotides
sulphur thiosulphates etc. It is only stable at
vary low equilibrium concentration.
• Stable insoluble Cu(i) compounds includes
CuCN, Cucl, Cu2S. The Cucl and Cu-cyanide are
even more stable in water than Cu(ii)
complexes
• The relative stabilities of Cu(i) and Cu(ii)
species in soluble values considerably
depending upon the nature of the ligands
present
EXAMPLE 3 (Zn)
• Zn has only one oxidation state, Zn(ii) that is
stable.
• In terms of its reactions with ligands, it is
intermediate between the hard and soft acids
• It has greater solubility and generally higher
concentrations than most metals.
• However the widespread use of Zn makes
contamination a real problem
• Concentrations are generally low for wider drainage;
0.00002-0.0018 mg/liter
• But concentration in rivers are relatively high up to
0.0156 mg/liter or more in cases of anthropogenic
pollution. For example, giltrale fraction of water
drainage
• In normal pH range waters;
- Free Zn-ion is the predominant form.
- Then CO32- and (OH)- are the dominant species
EXAMPLE 4 (Pb)
•
•
•
•
A member of group IV (C, Si, Ce, Sn)
Like Cu and Zn it is intermediate between
hard and soft acids.
It has more than one oxidation state.
Background concentration of Pb; 0.0000060.000050 mg/L, measured in remote
streams in the U.S.A.
• Concentrations of rivers affected by mining are
generally up to 0.065-1.11 mg/L
• It is the low oxidation state, Pb(ii), which is the most
stable under normal oxidising conditions.
• Below pH 7.1; Pb(ii) is present as free ion and for pH
>7.1 it exists as a carbonate (PbCO3) and hydroxide
• Adsorption plays a major role in controlling
concentration in solution.
Organic Complexes
Organometallic Compounds
• Organometallic compounds are compounds
containing a covalent bond between a carbon
atom and a metal.
• Toxicity often results from bioaccumulation of
fatty tissues and penetrations of membrane
barriers
EXAMPLE
• Ionic Hg(ii) is toxic to the mammalian kidney
and corrosive at sides of mucosal absorptions
• CH3Hg+ Crosses the blood-brain barrier,
acting as teratogen (any substance that interferes with
the normal embryonic development) and central
nervous system toxin.
• Metals that undergo biomethylation-Hg, Ce &
Sn
• Biomethylation of metals is generally
restricted to microorganisms in sediments and
soils with the exception of Co in Vitamin B12
• Note; Methylation of metals generally
increases their toxicity, Methylation of As & Se
contributes to their detoxification
• Manufactured organometallic also find their
way into the environment through deliberate
addition to treat seed grains eg (Phenyl) Sn as
a fungicide and CH3As(ONa)2 as a herbicides
or as (CH3)4 Pb and (C2H5)4 Pb during
gasoline.
• In the environment, binding to polyamons
such as humic and solic acids and to colloidal
particles contributes to the patterns of
transport or availability of elements e.g. Cd
,Cn & Pb
Pathways of toxic elements is soils
animals and plants
• For identification of environmental
impact, conceptual model of
pollution can be used as a flow chart
in order to follow the chain in
determination of the pathways of
toxic materials to bioreceptors
Conceptual model
• Elements emitted from sources are dispersed in
the environment (air, in water and soils) through
several transport agents
– Wind transportation- metal rich dust and particulates
– Rainwater and flood – flows into rivers and lakes
– Gravity – movement of particles down the sides of
heaps into rivers or onto soil
– Land , air or sea—anthropogenic transportation
– Placement – direct tipping of material
• During transportation, some heavy metals are
deposited in soils that are taken by biota
• and some are absorbed by humans and
follows the route of chemical absorption
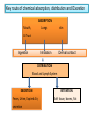
Key routs of chemical absorption, distribution and Excretion
ABSORPTION
Mouth,
Lungs
skin
Inhalation
Dermal contact
GI Tract
Ingestion
DISTRIBUTION
Blood and Lymph System
EXCRETION
Feces, Urine, Expired Air,
secretion
RETENTION
Soft tissue, bones, Fat
Absorption, where toxic materials might be ingested
through the mouth, or inhaled through the lungs or
through dermal contact with the skin
Distribution, this where these toxic chemicals will be
distributed through the blood and the lymph system
Excretion, toxic materials will then be excreted as feces,
urine, expired air
Retention, when the toxic materials are not excreted then
they are retained in the body such as in soft tissue, bones or
as fat
In plants absorption of heavy metals are taken up
by roots system from soil solution, which can be
by:
• Passive (non-metabolic) uptake, involving
diffusion of ions in the soil solution into the
root endodermis
• Active (metabolic) uptake which takes place
against the concentration gradient but
requires metabolic energy and can therefore
be inhibited by toxins
• Relative difference in the uptake of the ions is due
to several factors such as surface area of the root,
rate of evapotranspiration and roots exudates
• General order of transfer coefficient for most of
biologically controlled heavy metals were deduced
as indicated in tabled below, which is the metal
concentration in plant tissue above ground divided
by total metal concentration in the soil
Soil-plants transfer coefficients of heavy metals
Element
Soil-Plant Transfer coefficient
(SPTC)
As
0.01 – 0.1
Cr
0.01 -0,1
Cu
0.1 – 10
Pb
0.01 – 0.1
Zn
1 – 10
Dose Response Curve
– Dose-response curve is a simple X-Y graph relating
the magnitude of a stressor
(e.g. concentration of a pollutant, amount of a drug,
temperature, intensity of radiation)
to the response of the receptor (e.g. organism under
study)
- The response may be a physiological or
biochemical response, or even death (mortality)
• The measured dose (milligram, micrograms, or
grams per kilogram of body-weight) is
generally plotted on the X axis
• and the response is plotted on the Y axis
• the curve is typically sigmoidal , with the
steepest portion in the middle
• It usually illustrate the dose of the essential
elements
• Simply relates the exposure dose and the
measurable biological effect
• It should be realised that dose-response
relationships will generally depend on the
• exposure time; quantifying the response after
a different exposure time leads to a different
relationship and possibly different conclusions
on the effects of the stressor under
consideration.
• 90 natural occurring elements
• 73 are trace elements in the body
• 18 of 73 are essential trace elements
Abundance by mass % of major and minor
elements in the human body
Element
Mass%
element
Mass%
Oxygen
65
Calcium
1.4
Carbon
18
Phosphorus 1
Hydrogen
10
Magnesium
0.50
Nitrogen
3
Potasium
0.34
Sulfur
0.26
Sodium
0.14
Chlorine
0.14
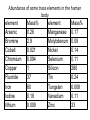
Abundance of some trace elements in the human
body
element
Mass%
element
Mass%
Arsenic
0.26
Manganese 0.17
Bromine
2.9
Molybdenum 0.08
Cobalt
0.021
Nickel
0.14
Chromium
0.094
Selenium
0.11
Copper
1
Silicon
260
Fluoride
37
Tin
0.24
Iron
60
Tungsten
0.008
Iodine
0.19
Vanadium
0.11
lithium
0.009
Zinc
33
• If an essential trace element is completely
withdrawn from the diet ,
• signs and symptoms of deficiency should
occur ,
• such as growth retardation and loss of hair
• supplementation should alleviate the
symptoms and reverse the deficiency state
Changing nature of diseases
Toxicity
• This can be explained by toxicity
• Toxicity/Poisoning - chemical injury to body organs or
a chemically induced disturbance of the functions in
biological systems
• Such toxic effects may follow the exposure to
exogenous (environmental) substances
• Substances that normally are considered as harmless
may also, if the dose is big enough, cause deleterious
effects and thereby act as poisons
• Examples of this are sodium chloride, oxygen and
water.
• The science devoted to the study of the
structures, effects and fate of poisonous
substances is called toxicology
• Clinical toxicology deals with problems related
to poisonings in humans and their treatment
• Poisoning may be acute, accelerated or
chronic
Acute Toxicity
• In acute poisoning the body is exposed to the toxic substance
in a high dose, on one occasion and during a short period of
time
•
Symptoms of poisoning develop in close relation to the
exposure
• Examples of acute poisonings are massive drug overdoses,
poisonings after mushroom meals, alcohol poisonings, and
bites or stings
• Acute poisoning just means 1 big dose of a
poison that kills something or someone all at
once
• So, death by lethal injection is acute poisoning
• The circumstances around an acute poisoning
are often obvious and easy to detect
• Mainly occurs at less than 24hours
• But still can be up to 14 days from the days of
taking the substances
Chronic Toxicity
• In chronic poisoning the organism is on the contrary
exposed repeatedly to toxic agents during a long period,
• but every exposure means that just a low dose of the
poison is entering the body
• Normally no symptoms develop in relation to each
exposure, although that may happen (e.g. in chronic
exposure to solvents)
• Instead the patient gradually becomes ill after a period of
months or years
• In chronic poisoning the toxic substance may
accumulate in body tissues or cause a small
irreversible damage at each exposure
• After a long time, enough poison has been
accumulated in the body, or the damage has become
significant enough, to cause clinical symptoms
• Examples of typical chronic poisonings are long-term
exposure to heavy metals like lead, mercury or
cadmium, inhalation of organic solvents in the
occupational context, and exposure to pesticides.
• chronic poisoning is a buildup of toxic
chemicals over time
• the slow and insidious onset of a chronic
poisoning may cause considerable diagnostic
difficulties.
Accelerated toxicity
• Is intermediate between chronic and acute
toxicity
• Develops after 5 -10 years of heavy exposure
to toxic elements
• It is the late stage of acute toxicity and early
stage of chronic effect
• Symptoms of chronic diseases starts to show
off.
Sources , pathways and effects of
excess/deficiency
of :
Selenium, lead, mercury, zinc,
and cadmium
Selenium
• Selenium is a non metallic chemical element.
• In chemical activity and physical properties it
resembles sulfur and tellurium
• Selenium appears in a number of allotropic
forms: the most popular are a red amorphous
powder, a red crystalline material, and a gray
crystalline metal like form called metallic
selenium.
Se
• Selenium burns in air and is unaffected by water, but
dissolves in concentrated nitric acid and alkalis
• Low levels of selenium can end up in soils or water
through weathering of rocks
• It will then be taken up by plants or end up in air
when it is adsorbed on fine dust particles.
• Selenium is most likely to enter the air through coal
and oil combustion, as selenium dioxide.
Se
• This substance will be converted into selenium acid
in water or sweat
• Selenium substances in air are usually broken down
to selenium and water fairly quickly, so that they are
not dangerous to the health of organisms
• Selenium can accumulate in the body tissues of
organisms and can then be passed up through the
food chain
Se
• Usually this bio magnification of selenium starts
when animals eat a lot of plants that have been
absorbing large amounts of selenium, prior to
digestion
• Due to irrigation run-off concentrations of selenium
tend to be very high in aquatic organisms in many
areas
• When animals absorb or accumulate extremely high
concentrations of selenium it can cause reproductive
failure and birth defects
Se
• - Selenium exposure takes place either through food or water,
or when we come in contact with soil or air that contains high
concentrations of selenium.
• -This is not very surprising, because selenium occurs naturally
in the environment extensively and it is very widespread.
• -The exposure to selenium mainly takes place through food,
because selenium is naturally present in grains, cereals and
meat.
• - Selenium uptake through food may be higher than usual in
many cases, because in the past many selenium-rich fertilizers
have been applied on farmland.
Se
• People that live near hazardous waste-sites will
experience a higher exposure through soil and air
• Selenium from hazardous waste-sites and from
farmland will end up in groundwater or surface water
through irrigation
• This phenomenon causes selenium to end up in local
drinking water, so that exposure to selenium through
water will be temporarily increased.
Se
• People that work in metal industries, selenium-recovery
industries and paint industries also tend to experience a
higher selenium exposure, mainly through breathing
• Selenium is released to air through coal combustion
• People that eat a lot of grains that grow near industrial sites
may experience a higher exposure to selenium through food
• Exposure to selenium through drinking water may be
increased when selenium from hazardous waste disposals
ends up in water wells.
Se
• Exposure to selenium through air can cause dizziness, fatigue
and irritations of the mucous membranes
• When the exposure is extremely high, collection of fluid in the
lungs and bronchitis may occur.
• Selenium uptake through food is usually high enough to meet
human needs; shortages rarely occur. When shortages occur
people may experience heart and muscle problems.
• When selenium uptake is too high health effects will be likely
to come about.
Se
• The seriousness of these effects depends upon the
concentrations of selenium in the food and how often this
food is eaten.
• The health effects of various forms of selenium can vary from
brittle hair and deformed nails, to rashes, heat, swelling of the
skin and severe pains
• When selenium ends up in the eyes people experience
burning, irritation and tearing.
• Selenium poisoning may become so severe in some cases that
it can even cause death.
Se
• Overexposure of selenium fumes may produce
accumulation of fluid in the lungs, garlic breath,
bronchitis, pneumonitis, bronchial asthma, nausea,
fever, headache, sore throat, shortness of breath,
conjunctivitis, vomiting, abdominal pain, diarrhea
and enlarged liver
• Selenium is an eye and upper respiratory irritant and
a sensitizer. Overexposure may result in red staining
of the nails, teeth and hair
Se
• Selenium deficiency causes rare endemic
diseases where there is low level of Se in soil
• Keshan cardiomyopathy and Kashin – Beck
diseases, which are deforming arthritis (China)
• Mild deficiency of Se causes heart and cancer
(Europe)
Lead
• -Lead is a soft, malleable poor metal, also considered
to be one of the heavy metals
• -Pb is mainly light blue in colour which is soft and
resistance to corrosion
• -It was mainly used in petrol (gasoline), but is being
phased out on environmental grounds
• -Used in manufacturing car battries, electrodes, in
the glass of computer and television screens, bullets,
Pb
• Native lead is rare to find in nature, but
naturally is found in zinc, silver and copper
ores and it is extracted together with these
metals
• Metallic lead does occur in nature, but it is
rare.
• Lead is usually found in ore with zinc, silver
and (most abundantly) copper, and is
extracted together with these metals.
• The main lead mineral is galena (PbS), which
contains 86.6% lead.
Pb
• Other common varieties are cerussite (PbCO3) and
anglesite (PbSO4).
• Besides occurring naturally more concentrations
found in the environment are a result of
anthropogenic activities
• Example is Pb from petrol - Chlorines bromines and
oxides pollute the environment- through the
exhausts of cars
• Larger particles pollute soils or surface waters, the
smaller particles will travel long distances through
air and remain in the atmosphere
Pb
• Its main pathways are through uptake of food, (70%),
water (20%) and air (10%).
• Example of Foods staff include fruit, vegetables,
meats, grains, seafood, soft drinks and wine may
contain significant amounts of lead
• Cigarette smoke also contains small amounts of lead
• Lead can enter (drinking) water through corrosion of
pipes especially when water is highly acidic
Pb
• Effects of Pb include:
• Brain damage, Miscarriages, anaemia, blood
pressure, Kidney damage, Infertility of men, Lowering
IQ of children, hyperactivity and damage to the
nervous system and the brains of unborn children.
• Other anthropogenic activities include not fuel
combustion, industrial processes and solid waste
combustion
Lead can end up in water and soils through corrosion
of leaded pipelines in a water transporting system
and through corrosion of leaded paints.
Pb
• It cannot be broken down; it can only converted to other
forms.
• Lead accumulates in the bodies of water organisms and soil
organisms. --Fish can take place even when only very small
concentrations of lead are present.
• Body functions of phytoplankton (O) can be disturbed when
lead interferes.
• -Soil functions are disturbed by lead intervention, especially
near highways and farmlands, where extreme concentrations
may be present.
Mercury (Hg)
•
•
•
•
•
•
•
•
•
It is heavy , silver metal
Liquid at room temp. but solidifies at - 40°C
Boils at 355 °C
High specific gravity – 13.6 g.cm-3
Alloy with other metals
Part finder of cinnabar
Main ore of Hg
Rhombohedral crystal structure
t can be extracted by heating cinnibar in a current of
air
Hg
• It occurs in two oxidation state Hg (I) and Hg (II)
• It exists in the form of inorganic and organic mercury
• Methylation of inorganic mercury into organic
mercury occurs in the microorganisms under
anaerobic conditions e.g in underwater sediments
• Organic mercury is highly poisonous and it is easily
absorbed by the gastric and intestinal organs
• It is then carried by blood into the brain , liver and
kidney
Natural sources Hg
• Extremely rare native element on Earth crust
• Also found in cinnibar and also in other minerals
such as corderite and livingstonite
• Mercury ore generally occurs in belts where rocks of
high density are forced to the crust of the earth
• Often in hot spring or other volcanic regions
• Natural source such as volcanic eruption is
responsible of almost half of the atmospheric Hg
emissions
Release in the environment
anthropogenic sources
• The rest of % is mainly released by human –
generated activities which include:
• Emission of stationary coal combustion and oil
combustion
• Gold production
• Cement production
• Waste disposal- municipal and hazardous waste and
sewage sluge
• From steel production
• Mercury production for batteries
Applications
• Cosmetics – manufacturing of mascara
• Production of chlorine and caustic soda – Cl is
produced from NaCl using electrolytes to
separate Na from Cl
• Caustic soda is produced by dissolving salt in
water and brine is produced with byproduct
being NaOH
• Dentistry – main ingredients in dental
amalgams
Hg in Gold mining
• For century Hg has been used for
amalgamation of gold
• It is estimated that about 1.32 kg of Hg is lost
for every 1 kg of Au produced
• About 40% of this loss occur during the initial
concentration and amalgamation stage of Au
• The lost is released directly into the soil,
streams and rivers, initially as inorganic Hg,
which later converts into organic Hg
Hg in Gold mining
• This is taken into the food chain, mainly by fish and
other aquatic life
• The remaining 60% Hg is released directly into the air
when the Hg is released directly into the air when Hg
– Au amalgam is heated during the purification
process and is often inhaled
• Hg is a very volatile element, thus dangerous levels
are readily obtained in air
• Safety standards require that Hg vapour should not
exceed 0.1 mg/m3 in air
Hg in Gold mining
• Hg once taken in, is accumulated into the human body and
attacks the central nervous system, resulting in:
• numbness and unsteadiness in the legs and hands,
• awkward movements,
• tiredness,
• ringing in the ears,
• narrowing of the field of vision,
• los of hearing,
• slurred speech,
• loss of sense of smell and taste and
• forgetness
Hg in Gold mining
• Hg poisoning may lead to disease known as minamata
• Minamata disease was first detected in 1956 in Minamata Bay
–Japan
• It was caused by eating large quantities of fish or shell fish
contaminated by industrial discharges of Hg compounds in
Minamata Basin
• 200mg/L of Hg in blood and 50mg/g in hair are the
provisionally established standards and any higher
concentration is considered to be at risk of poisoning
• The allowable level of methyl Hg in fish is 0.3ppm
Zinc
• Zinc, is a bluish-white, lustrous metal. It is less dense than iron
and has a hexagonal crystal structure.
• The metal is hard and brittle at most temperatures. Zinc is a
fair conductor of electricity.
• It has relatively low melting (420 °C) and boiling points
(900 °C).
• Metals long known to form binary alloys with zinc are
Aluminium, antimony, bismuth, gold, iron, lead, mercury,
silver, tin, magnesium, cobalt, nickel, tellurium and sodium.
• Zinc makes up about 75 ppm (0.007%) of the Earth crust,
making it the 24th most abundant element there.
Zn
• Soil contains an average of 64 ppm, but actual soils
have 5–770 ppm zinc.
• Seawater has only 30 ppb zinc and the atmosphere
contains 0.1 to 4 µg/m3.
• The element is normally found in association with
other base metals such as copper and lead in ores.
• Zinc is a chalcophile ("sulfur loving"), meaning the
element has a low affinity for oxygen and prefers to
bond with sulfur in highly insoluble sulfides.
Zn
• Other minerals from which zinc is extracted include
smithsonite (zinc carbonate), hemimorphite (zinc silicate),
wurtzite (zinc sulfide), and sometimes hydrozincite (basic zinc
carbonate).
• With the exception of wurtzite, all these other minerals were
formed as a result of weathering processes.
• Exploitable reserves of zinc exceed 100 million tonnes, with
large deposits in Australia, Canada and the United States.
• Zinc deficiency occurs where insufficient zinc is available for
metabolic needs.
Zn
• It is usually nutritional, but can be associated with
malabsorption, acrodermatitis enterropathica,
chronic liver disease, chronic renal disease, diabetes,
malignancy, and other chronic illnesses.
• Diets rich in foods that contain a lot of phytic acid,
such as cereals and legumes, decrease zinc
absorption into the body.
• Zinc supplements are important and help prevent
disease and reduce mortality, especially among
children with low-birth weight or stunted growth.
Zn
• Symptoms of chronic zinc deficiency are stunted
growth and retarded sexual development.
• Plants that grow in soils that are zinc-deficient are
more susceptible to disease.
• Even though zinc is an essential requirement for a
healthy body, excess zinc can be harmful.
• Excessive absorption of zinc suppresses copper and
iron absorption.
Zn
• The free zinc ion in solution is highly toxic to plants,
invertebrates, and even vertebrate fish.
• Stomach acid with HCl, will cause metallic zinc to
dissolves readily to give corrosive zinc chloride.
• Levels of zinc in excess of 500 ppm in soil interferes
with the ability of plants to absorb other essential
metals, such as iron and manganese.
• -Chronic ingestion of over 1 kg of zinc results in
death due to gastrointestinal bacterial and fungal
infections.
Zn
• Ingestion of about 12 grams of zinc, only shows lethargy and
ataxia (gross lack of coordination of muscle movements)
• Pennies and other small coins are sometimes ingested by
dogs, resulting in the need for medical treatment to remove
the foreign body.
• The zinc content of some coins can also cause zinc toxicity,
which is commonly fatal in dogs, where it causes a severe
anemia, also liver or kidney damage; vomiting and diarrhea
are possible symptoms.
• Storing fruit juices in galvanized cans has resulted in mass
poisonings of zinc
Cd
• Cd is a Lustrous, silver –white , ductile, very
malleable metal
• Its surface has a bluish tinge and the metal is
soft enough to be cut with a knife, but it
tarnishes in air
• It is soluble in acids but not in alkalis
• It is similar in many respects to zinc but it
forms more complex compounds
Cd in the environment
Natural Sources
• Cd can mainly be found in the earth’s crust
• It always occurs in combination with zinc
• Naturally a very large amount of cadmium is
released into the environment, about 25 000
tons a year
• About half of this cadmium is released into
the air through forest fires and volcanoes
• The rest of the cadmium is released through
human activities, such as manufacturing
Natural Sources
• Mostly Cd ore is not mined for the metal,
because more than enough is produced as a
byprolduct of the smelting of zinc from its ore,
sphelarite (ZnS), in which CdS is a significant
impurity, making up as much as 3%
• Thus the main mining areas are those associated
with zinc
• World production is around 14.000 tons pre year,
the main producing country is Canada, with the
USA, Australia, Mexico, Japan and Peru also being
the major suppliers
Anthropogenic
• Cd also available in the industries as an
inevitable by-product of zinc, lead, and copper
extraction
• After being applied it enters the environment
mainly through the ground and pesticides
• Also produced during metalliferous mining
and smelting
Pathways
• Cd waste streams from the industries mainly end up in soils
• The causes of these waste streams are for instance zinc
production, phosphate ore implication and bio industrial
manure
• Cd waste stream may also enter the air through
(household) waste combustion and burning of fossils fuels
• Because of regulations only little cadmium now enters the
water through disposal of waste water from households or
industries
• Another important source of cadmium emission is the
production of artificial phosphate fertilizers
Pathways
• Part of the Cd ends up in the soil after the fertilizers is
applied on farmland and the restg of the cadmium
ends up in surface waters when waste from fertilizers
productions is dumped by production companies
• Cd can be transported over great distances when it is
absorbed by sludge. This Cd rich sludge can pollute
surface water as well as soils
• When Cd is present in soils it can be extremely
dangerous, as the uptake through food will increase
• Soils that are acidified enhance the Cd uptake by plants
• This is a potential danger to the animals that are
dependent upon the plants for survival
Pathways
• Animal eating or drinking cadmium sometimes
get high blood pressure, liver diseased and
nerve or brain damage
• The susceptibility to Cd can vary greatly
between aquatic organisms
• Salt water organisms are known to be more
resistant to Cd poisoning than fresh water
organisms
Pathways
• Cd can accumulate in their bodies, especially when they eat
multiple plants
• Cows may have large amounts of Cd in their kidneys due to this
earthworms and other essential soil organisms are extremely
susceptive to Cd poisoning
• They can die at very low concentrations and this has consequences
for he soil structure
• When Cd concentrations in soils are high they can influence soil
processes of microorganisms and threat the whole soil ecosystem
• In aquatuic ecosystem Cd can bio accumulate in muscles, shrimps,
lobsters and fish
• Salt water organisms are known to be resistant to Cd poisoning
than fresh water organisms
Health effect of Cd
• Human uptake of Cd takes place mainly through food
chain
• Foodstuffs that are rich in Cd can greatly increased the
Cd concentration in human bodies
• Examples are liver, mushroom, shellfish, and dried
seaweed
• An exposure significantly higher Cd levels occurs when
people smoke
• Tobacco smoke transport Cd into the lungs
• Blood will transport it through the rest of the body
where it can increase effects by potential Cd that is
already present from Cd-rich food
Health Effects of Cd
• This may even cause death
• Cd is firstg transported to the liver thropugh the blood
• It is then bounded to proteins to form complexes that
are transported to the kidneys
• Cd accumulates in kidneys, where it damages filtering
mechanisms
• This causes the excretion of essential proteins and
sugar from the body and further kidney damage
• It takes a very long time before cadmium that has
accumulates in kidneys is excreted from a human body
Health effects of Cd
• Other exposures can occur with people who
live near harzadeous waste sites or factories
that releases Cd into the air and people that
work in the metal refinery industry
• When people breathe in cadmium it can
severely damage the lungs
Health effects of Cd
•
•
•
•
•
•
•
•
Other health effects that can be caused by Cd are
Diarrhea, stomach pains and severe vomiting
Bone fracture
Reproductive failure and possibly even infertility
Damage to the central nevours system
Damage to the immune system
Psychological disorders
Possibly DNA damage or cancer development
Dust emission in mining and mineral
processing and the fate of inhaled
particles
Silicosis
Asbestosis
Coal workers pneumoconiosis
Silicosis
• Silica is like asbestos, can result in the development of fibrotic
lundg disese and results into a disease known as Silicosis
• Natural Sources: Rock , sand and soil
• The most common of crystaline silica is known as quarts
• Inhailation of air born dust, that contain crystalline silica can
occur in a wide variety of settings:
- mining, quarrying and stone cutting, founry operations, paintblasting and sand blasting, glass manufacturing and etching
and some types construction workings
Three types of Silicosis
• Chronic, acute and accelerated silicosis
• Chronic silicosis
• A progressive lung disease, characterized by
the development of fibrotic tissue in response
of inhalation of quarts particles in the size of
0.5 – 0.7 micrometer
• Heart and respiratory failure is the
consequences of these type of silicosis
Acute silicosis
• In workers exposed to exceptionally high
concentrations of fine particles of silica,
usually quarts dust
• The lungs are often heavy and rigid as the air
spaces are filled a finely granular substances
Accelerated silicosis
• Is intermediate between chronic and acute silicosis
• Develops after 5 -10 years of heavy exposure to silica
dust grater than 1.0 micrometer of almost pure
quarts
• They show no clinical abnormalities other than
breathlessness
• Death by cardiopulmonary failure within ten years of
onset of symptoms is often the outcome of this form
of silicosis
Asbestos
• Asbestos is the name of a group of highly fibrous
minerals
• Separable, long and thin fibers
• Fibers are strong enough and flexible enough to
be woven
• Asbestos fibers are heat resistant, making them
useful for may industrial purposes
• Because of their durability, asbestos fibers that
get into lung tissue will remain for long periods of
time
Types of Asbestos
• Amphibolite – fibers stay in lthe lungs longer
than chrysolite – this tendency may account
for their increased toxicity
• Example; Brittle crystalline fibers amphibole
minerals (actinolite, tremolite, anthophylite,
crocidolite and amosite asbestos)
• Chryssolite – less toxic , e.g. serpentine
mineral with long and flexible fibers
Exposure to Asbestos
• We are all exposed to low levels of asbestos in
the air
• Much more concentrated levels of exposure
are known to cause health effects in humans
Anthropogenic exposure
• Anthropogenic exposures asbestos product
– manufacturing (insulation, roofing,
building
materials)
– Automotive rep air (breaks and clutches)
– Minig operations
– Railroads
– Sand or abrasive manufactures
– Shipyards/ships/
– Steel mills
Amphibole exposure
• Mining activities:
• Exposure to tremolite asbestos can occur in
workers involved in mining . Milling, and
handling of other ores and rocks containing
tremolite asbestos (such as vermuculite or
talc)
• Residence who live near milling, milling or
manufacturing sites
Amphibole Asbestos exposure
• Insulation and building materials – amphibole
asbestos can be found in a variety of building
materials such as insulation, ceiling boards, ,
floor tiles and cement pipes
• Occurs in some vermuciulite sources used as
home and building insilation
Amphibole asbestos exposure
• Consumer Products – Vermuculite was also
commonly sold in gardening and hardware
stores
• It was used as to improve soil quality or
fertilizers carrier
• Small amounts of amphibole asbestos have
been found in some talc containing crayons
• Manufactures of these crayons agreed to
eliminate talc from their products
Health effects
• Significant exposure to any type of asbestos will increase
the risk of lung cancer, mesothelioma and nonmaligmant
lung and pleural dissorders,
• This include asbestosis, pleural plaques, pleural thickening,
and pleural effusions (5 to 1,200 fiberyear/mL)
• Such exposure will result from 40 years of occupational
exposure to air concentrations of 0.125 to 30 fiber/mL
• diseases from asbestos exposure takes a long time to
develop
• Most cases of lung cancer or asbestosis in asbestos workers
occur 15 or more years after initial exposure to asbestos
Health Effects
• Tobacco smokers are at far greater risk-than
non-smokers who has been exposed to
asbestos
• Diagnosis of mesothelioma from exposure to
asbestos commonly has been 30 years or
more
Asbestos Facts
• When asbestos fibers are inhaled, most fibers are
expelled, but some can be lodged in the lungs and
remain there throughout life. Fibers can accumulate
and cause scaring and inflamation
• Inhaling longer, more durable asbestos fibers ( such as
tremolite and other amphiboles) contributes to the
severity of asbestos-related disorders
• Exposure to asbestos, including tremolite, camincrease
the likelihood of lung cancer, mesothelioma, ond
nonmalignant lung conditions such as asbestosis
• Fluorosis
• Acid Mine Drainage
Fluorosis
• A form of enamel hypomineralization leading to discoloration
due to excessive ingestion of fluoride during the development
of the teeth
• It is caused by over exposure to fluorides from all sources ;
• Fluoridated water, Fluoride toothpaste, Mouth rinses, tablets,
gels, sealants,
• Foodstuffs: tea, medicines, anaetsthetic, pesticides, ,
herbicides, polluted air/dust/fumes
• This condition can manifest in the form of skeletal fluorosis
and dental fluorosis
• The damage caused by fluorosis is permanent
• Prevention is crucial
Asbestos Facts
• Changes in the lining of the lungs (pleural)
such as thickening , plaques, calcification, and
fluid around the lungs (pleural effusion) may
be early signs of asbestos exposure
• These changes can affect breathing
• Pleural effusion can be an early warning sign
for mesothelioma (cancer of the lining of the
lungs)
Asbestos Facts
• Most cases of asbestos or lung cancer in workers
occurred 15 years or more after the person was first
exposed to asbestos
• Most cases of mesothelioma are diagnosed 30 years or
more after the first exposure to asbestos
• Mesothelioma has been diagnosed in asbesos workers
family members, and residents who live close to
asbestos mines
• Health effects from asbestos exposure may continue to
progress even after exposure is stopped
• Smoking or cigarette smoke, together with exposure to
asbestos, greatly increases the likelihood of lung cancer
Dental Fluorosis
•
•
•
•
It is sometimes called enamel fluorosis
It starts with small marks and sports on teeth
Mild transparency of the tooth enamel
If allowed to progress the teeth will become
pitted, cracked and brittle
• Dental fluorosis usually appears before
skeletal fluorosis
Skeletal fluorosis
• Fluoride attacks the bones of the body
• It causes brittle bones and joint pain
• Tricky to identify as other diseases may cause
the same symptoms
• In regions where it is endemic it is easily
detected
• Some tomes x-ray might be done to visualize
the bones
Importance of fluoride
• It is an important dietary
• Effective in cavity prevention
• The fact that fluorosis exists , is not a reason
to stop using fluoride in dental care
• But just to be careful
• Fluoride filters can be used to reduce the
content of fluoride in fluoridated water
Acid mine drainage
Geological aspects of waste disposal including
radioactive waste
Uranium
• Geogenic sources of uranium