* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Valence Bond Theory
History of quantum field theory wikipedia , lookup
Coupled cluster wikipedia , lookup
Hartree–Fock method wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Hidden variable theory wikipedia , lookup
Hydrogen atom wikipedia , lookup
Atomic theory wikipedia , lookup
Tight binding wikipedia , lookup
Atomic orbital wikipedia , lookup
Chemical bond wikipedia , lookup
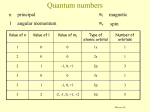
Valence Bond Theory When quantum mechanics is applied to a single atom the result is a 3-d probability distribution which indicates the likely location of an electron. … such as an s orbital, or a p orbital. When quantum mechanics is applied to a molecule (with several atoms) it becomes more complicated. Valence bond theory Valence bond theory says that electrons in a covalent bond occupy a region that is the overlap of individual atomic orbitals. For example, the covalent bond in molecular hydrogen can be thought of as result of the overlap of two hydrogen 1s orbitals. The two hydrogen atoms will approach until they are at the perfect distance from each other. At this point the system will have the least amount of energy and the highest amount of stability. - the atoms are more stable together than apart - too close … energy increases due to repulsion H H 1s1 1s1 - the orbitals are overlapped When an s and a p orbital overlap, the s and p lobe combine to form a larger lobe, while the opposite side of the p orbital is reduced in size. - the orbitals are still arranged in the x, y and z directions. ex NH3 N H H H 1s2 2s2 2px1 1s1 ex PCl3 P 1s2 2s2 2px2 2py2 2pz2 3s2 3px1 3py1 3pz1 Cl 1s2 2s2 2px2 2py2 2pz2 3s2 3px2 3py2 3pz1 Cl 1s2 2s2 2px2 2py2 2pz2 3s2 3px2 3py2 3pz1 Cl 1s2 2s2 2px2 2py2 2pz2 3s2 3px2 3py2 3pz1 2py1 2pz1 1s1 1s1 The problem with molecular orbital diagrams is they do not accurately represent the geometry of the molecule. According to the orientation of the atomic orbitals … each of the Cl’s in the PCl3 molecule should be arranged 90 o from each other. - one Cl along each of the x, y and z axis - this is not the case!