The structure of the energy bands and optical absorption in osmium
... The optical conductivity of osmium, a = nkw/2~,calculated from the values of the optical constants for two crystallographic directions Elc and Ellc, are shown in Fig. 1,a. In the energy range 2-5 eV, both curves, a, and a l l ,have a very intense interband absorption band with two maxima, C and D. T ...
... The optical conductivity of osmium, a = nkw/2~,calculated from the values of the optical constants for two crystallographic directions Elc and Ellc, are shown in Fig. 1,a. In the energy range 2-5 eV, both curves, a, and a l l ,have a very intense interband absorption band with two maxima, C and D. T ...
Key Concept 1: An atom is the smallest unit of an element that
... Key Concept 7: Electrons are located outside of the nucleus and arranged by energy levels in the electron cloud. There are a certain number of electrons that each energy level can hold. ...
... Key Concept 7: Electrons are located outside of the nucleus and arranged by energy levels in the electron cloud. There are a certain number of electrons that each energy level can hold. ...
Quantum Manipulation of Ultracold Atoms—V. Vuletic
... complicated level scheme has not been possible. In Doppler cooling, the preferred absorption of photons from a beam counterpropagating relative to the atom’s motion leads to slowing and cooling of the atom. Since the momentum “kick” associated with each photon absorption event is much smaller than t ...
... complicated level scheme has not been possible. In Doppler cooling, the preferred absorption of photons from a beam counterpropagating relative to the atom’s motion leads to slowing and cooling of the atom. Since the momentum “kick” associated with each photon absorption event is much smaller than t ...
Topic 15
... So, if we attempt to reduce uncertainty in position by decreasing λ, we INCREASE the uncertainty in the momentum of the particle!!!!!! Product of the uncertainties in position and momentum given by: ...
... So, if we attempt to reduce uncertainty in position by decreasing λ, we INCREASE the uncertainty in the momentum of the particle!!!!!! Product of the uncertainties in position and momentum given by: ...
THE CHEMICAL BASIS OF LIFE
... 11. Calcium (Ca) has an atomic number of 20; chlorine (Cl) has an atomic number of 17. a. The number of electrons in the outer shell of calcium is ______________. b. The number of electrons in the outer shell of chlorine is ______________. c. In a chemical reaction between these two atoms, _________ ...
... 11. Calcium (Ca) has an atomic number of 20; chlorine (Cl) has an atomic number of 17. a. The number of electrons in the outer shell of calcium is ______________. b. The number of electrons in the outer shell of chlorine is ______________. c. In a chemical reaction between these two atoms, _________ ...
A Spectral Analysis of Laser Induced Fluorescence of Iodine
... from the lowest vibrational state of the excited level. To understand why, note that at room temperature the electrons populate mostly the lowest vibrational levels of the ground state, indexed by the quantum number v 00 . Under incident visible light, the molecule will absorb photons and undergo vi ...
... from the lowest vibrational state of the excited level. To understand why, note that at room temperature the electrons populate mostly the lowest vibrational levels of the ground state, indexed by the quantum number v 00 . Under incident visible light, the molecule will absorb photons and undergo vi ...
Basic Fluorescence Principles I
... fully. He discovered that after many infusions the wood lost its power to give color to the water and concluded that there was some “essential salt” in the wood responsible for the effect. He also discovered that addition of acid abolished the color and that addition of alkali brought it back. Hence ...
... fully. He discovered that after many infusions the wood lost its power to give color to the water and concluded that there was some “essential salt” in the wood responsible for the effect. He also discovered that addition of acid abolished the color and that addition of alkali brought it back. Hence ...
File
... of energy levels for electrons. Eg. Elements with 3 energy levels (more than 10 electrons) are found in period (row) 3. Groups – elements have similar properties as those found above and below them. Sometimes called “families” eg. Group 1 is the Alkali Metals (look on page 25 for the name of the r ...
... of energy levels for electrons. Eg. Elements with 3 energy levels (more than 10 electrons) are found in period (row) 3. Groups – elements have similar properties as those found above and below them. Sometimes called “families” eg. Group 1 is the Alkali Metals (look on page 25 for the name of the r ...
7 Problems Chapter 7: Coulomb Blockade and the Single Elec! tron
... 7.5. There is always a capacitance between conductors separated by an insulating region. For the case of conductors associated with di¤erent circuits (i.e., circuits that should operate independently from one another), this is called parasitic capacitance, and, generally, C / 1=d, where d is some me ...
... 7.5. There is always a capacitance between conductors separated by an insulating region. For the case of conductors associated with di¤erent circuits (i.e., circuits that should operate independently from one another), this is called parasitic capacitance, and, generally, C / 1=d, where d is some me ...
AP Chemistry Unit 3 Test Review Topics Covered: Gases Liquids
... (D) XeF6 (E) XeF8 7) A rigid metal tank contains oxygen gas. Which of the following applies to the gas in the tank when additional oxygen is added at constant temperature? (A) The volume of the gas increase. (B) The pressure of the gas decreases. (C) The average speed of the gas molecules remains th ...
... (D) XeF6 (E) XeF8 7) A rigid metal tank contains oxygen gas. Which of the following applies to the gas in the tank when additional oxygen is added at constant temperature? (A) The volume of the gas increase. (B) The pressure of the gas decreases. (C) The average speed of the gas molecules remains th ...
Laser Communication Systems.pdf
... Gas lasers consist of a gas filled tube placed in the laser cavity. A voltage (the external pump source) is applied to the tube to excite the atoms in the gas to a population inversion. The light emitted from this type of laser is normally continuous wave (CW). ...
... Gas lasers consist of a gas filled tube placed in the laser cavity. A voltage (the external pump source) is applied to the tube to excite the atoms in the gas to a population inversion. The light emitted from this type of laser is normally continuous wave (CW). ...
PPT
... 4: If the potential U(x) has a center of symmetry (such as the center of the well above), the eigenstates will be, alternately, even and odd functions about that center of symmetry. Lecture 12, p 3 ...
... 4: If the potential U(x) has a center of symmetry (such as the center of the well above), the eigenstates will be, alternately, even and odd functions about that center of symmetry. Lecture 12, p 3 ...
4. Appraising the Proximate Analysis System
... In addition to all the problems previously mentioned, Proximate analysis: – doesn’t account for fecal, urine and gaseous losses. – Ignores palatability, digestibility, toxicity etc Hence, most labs have replaced aspects of the Proximate analysis system with modern analytical ...
... In addition to all the problems previously mentioned, Proximate analysis: – doesn’t account for fecal, urine and gaseous losses. – Ignores palatability, digestibility, toxicity etc Hence, most labs have replaced aspects of the Proximate analysis system with modern analytical ...
Chapter 2.4 Periodic properties of the elements
... First Ionization Energy The energy needed to completely remove one electron from a ground state gaseous atom is called the ionization energy. Energy must be added to the atom to remove an electron in order to overcome the force of attraction exerted on the electron by the nucleus. Since multi-elect ...
... First Ionization Energy The energy needed to completely remove one electron from a ground state gaseous atom is called the ionization energy. Energy must be added to the atom to remove an electron in order to overcome the force of attraction exerted on the electron by the nucleus. Since multi-elect ...
PHYSICAL SETTING CHEMISTRY
... may require the use of the 2011 Edition Reference Tables for Physical Setting/Chemistry. Base your answers to questions 66 through 68 on the information below. John Dalton, an early scientist, sketched the structure of compounds using his own symbols for the elements known at the time. Dalton’s symb ...
... may require the use of the 2011 Edition Reference Tables for Physical Setting/Chemistry. Base your answers to questions 66 through 68 on the information below. John Dalton, an early scientist, sketched the structure of compounds using his own symbols for the elements known at the time. Dalton’s symb ...
Frans R., Boksenbojm E., Tamassia L.,(2014) Quantum SpinOff
... (phosphorous). P has ……… electrons in their outer shell. …… of these electrons will form a bond with their neighboring atoms (Si or Ge). But the fifth one has no other electron to form together a bonding state. We lowered in fact the conduction band by introducing these unbound electrons. Electrons ...
... (phosphorous). P has ……… electrons in their outer shell. …… of these electrons will form a bond with their neighboring atoms (Si or Ge). But the fifth one has no other electron to form together a bonding state. We lowered in fact the conduction band by introducing these unbound electrons. Electrons ...
RSC ChemComm Template (PC) - Royal Society of Chemistry
... Transient absorption measurements were employed to corroborate the fluorescence assignment and also to shed light onto the fate of the C59N singlet excited state. The C60 and C59N references (i.e., 4 and 5) each have very distinct singlet absorption features, which are summarized in Figure S2. Excit ...
... Transient absorption measurements were employed to corroborate the fluorescence assignment and also to shed light onto the fate of the C59N singlet excited state. The C60 and C59N references (i.e., 4 and 5) each have very distinct singlet absorption features, which are summarized in Figure S2. Excit ...
Chapter 15 PowerPoint
... equivalent amount of positive charge, he concluded that atom was a mass of (+) charge, taking up almost total volume of atom, with tiny, near massless, electrons embedded in it Like raisins in a bun – Thomson Raisin Bun Model ...
... equivalent amount of positive charge, he concluded that atom was a mass of (+) charge, taking up almost total volume of atom, with tiny, near massless, electrons embedded in it Like raisins in a bun – Thomson Raisin Bun Model ...
Which notation represents an atom of sodium
... 16) On the potential energy diagram provided draw an arrow to represent the activation energy of the forward reaction. ...
... 16) On the potential energy diagram provided draw an arrow to represent the activation energy of the forward reaction. ...
Document
... solute(mol/kg), i is the Van 't Hoff factor (the number of dissolved particles the solute will create when dissolved), and Kb is the ebullioscopic constant unique to each solvent. Freezing-point depression is the difference between the freezing points of a pure solvent and a solution mixed with a so ...
... solute(mol/kg), i is the Van 't Hoff factor (the number of dissolved particles the solute will create when dissolved), and Kb is the ebullioscopic constant unique to each solvent. Freezing-point depression is the difference between the freezing points of a pure solvent and a solution mixed with a so ...
From waves to bullets: testing Feynman`s idea on the two slit
... consider our detectors as effectively one-dimensional objects, rather than physically two-dimensional. The continuous light pattern on the screen is then transformed into a discrete set of counts that can be easily visualized in an histogram, where each bar corresponds to a single detector and the h ...
... consider our detectors as effectively one-dimensional objects, rather than physically two-dimensional. The continuous light pattern on the screen is then transformed into a discrete set of counts that can be easily visualized in an histogram, where each bar corresponds to a single detector and the h ...
Nugget
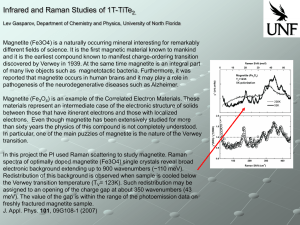
... Magnetite (Fe3O4) is an example of the Correlated Electron Materials. These materials represent an intermediate case of the electronic structure of solids between those that have itinerant electrons and those with localized electrons. Even though magnetite has been extensively studied for more than ...
... Magnetite (Fe3O4) is an example of the Correlated Electron Materials. These materials represent an intermediate case of the electronic structure of solids between those that have itinerant electrons and those with localized electrons. Even though magnetite has been extensively studied for more than ...
X-ray fluorescence

X-ray fluorescence (XRF) is the emission of characteristic ""secondary"" (or fluorescent) X-rays from a material that has been excited by bombarding with high-energy X-rays or gamma rays. The phenomenon is widely used for elemental analysis and chemical analysis, particularly in the investigation of metals, glass, ceramics and building materials, and for research in geochemistry, forensic science and archaeology.