Chronological overview of the 2009/2010 H1N1 influenza
... The Centre for Infectious Disease Control (CIb) was set up to coordinate – at national level and wherever necessary – the prevention and control of outbreaks of infectious diseases. These outbreaks usually occur at local, regional or supraregional level and hardly ever on a national or international ...
... The Centre for Infectious Disease Control (CIb) was set up to coordinate – at national level and wherever necessary – the prevention and control of outbreaks of infectious diseases. These outbreaks usually occur at local, regional or supraregional level and hardly ever on a national or international ...
4.7 INFLUENZA 4.7.1 Virology 4.7.2 Clinical features 4.7.3
... community.24-32 The majority of effectiveness studies have been done with TIV due to their widespread use. Although the burden of influenza in young children is large, historically there has been limited data on the efficacy and effectiveness of influenza vaccines in this age group. However, there i ...
... community.24-32 The majority of effectiveness studies have been done with TIV due to their widespread use. Although the burden of influenza in young children is large, historically there has been limited data on the efficacy and effectiveness of influenza vaccines in this age group. However, there i ...
Prevention and Control of Influenza Recommendations of the Advisory Committee
... reassortment between human A (H3N2) and A (H1N1) viruses have been detected recently in many countries. Both influenza A and B viruses are further separated into groups on the basis of antigenic characteristics. New influenza virus variants result from frequent antigenic change (i.e., antigenic drif ...
... reassortment between human A (H3N2) and A (H1N1) viruses have been detected recently in many countries. Both influenza A and B viruses are further separated into groups on the basis of antigenic characteristics. New influenza virus variants result from frequent antigenic change (i.e., antigenic drif ...
The elusive definition of pandemic influenza
... extraordinary and costly responses to what turned out to be mostly ordinary disease.29,30 This resulted in much scrutiny and controversy over the decision-making process. As future policy responses to emerging infectious diseases will not succeed without the trust and understanding of the public, of ...
... extraordinary and costly responses to what turned out to be mostly ordinary disease.29,30 This resulted in much scrutiny and controversy over the decision-making process. As future policy responses to emerging infectious diseases will not succeed without the trust and understanding of the public, of ...
Antiviral agents targeting the influenza virus: a review and
... Antiviral agents targeting the influenza virus: a review and publication analysis L. Eyer, K. Hruska Veterinary Research Institute, Brno, Czech Republic ABSTRACT: Influenza is a serious infectious disease, which is life-threatening especially in children, seniors and immunocompromised patients. In a ...
... Antiviral agents targeting the influenza virus: a review and publication analysis L. Eyer, K. Hruska Veterinary Research Institute, Brno, Czech Republic ABSTRACT: Influenza is a serious infectious disease, which is life-threatening especially in children, seniors and immunocompromised patients. In a ...
Pandemic Influenza - Facilitator`s Manual
... increased as more cases appeared in various areas of United States. Patient tracking in hospitals reveals that the virus is starting to be spread via human-to-human contact transmission between previously hospitalized patients and those who had close contact with these individuals. In fact, a signif ...
... increased as more cases appeared in various areas of United States. Patient tracking in hospitals reveals that the virus is starting to be spread via human-to-human contact transmission between previously hospitalized patients and those who had close contact with these individuals. In fact, a signif ...
Pandemic Influenza Tabletop Exercise Participant`s Manual
... increased as more cases appeared in various areas of United States. Patient tracking in hospitals reveals that the virus is starting to be spread via human-to-human contact transmission between previously hospitalized patients and those who had close contact with these individuals. In fact, a signif ...
... increased as more cases appeared in various areas of United States. Patient tracking in hospitals reveals that the virus is starting to be spread via human-to-human contact transmission between previously hospitalized patients and those who had close contact with these individuals. In fact, a signif ...
Influenza and Children with Asthma
... National Heart, Lung, and Blood Institute, includes influenza vaccine as one of its 10 “key clinical activities” aimed at reducing asthma morbidity and mortality.16 Although direct evidence of influenza vaccination benefits in patients with asthma is limited and some trials have failed to demonstrat ...
... National Heart, Lung, and Blood Institute, includes influenza vaccine as one of its 10 “key clinical activities” aimed at reducing asthma morbidity and mortality.16 Although direct evidence of influenza vaccination benefits in patients with asthma is limited and some trials have failed to demonstrat ...
Influenza vaccines
... considerably from year to year depending on the characteristics of the circulating virus strains and level of immunity in the population. On average, 85 deaths and approximately 4,000 hospitalisations are directly attributed to influenza annually in Australia, although this has long been recognised ...
... considerably from year to year depending on the characteristics of the circulating virus strains and level of immunity in the population. On average, 85 deaths and approximately 4,000 hospitalisations are directly attributed to influenza annually in Australia, although this has long been recognised ...
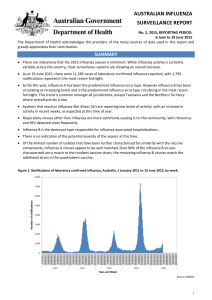
Australian Influenza Surveillance Report No 02
... The WHO6 has reported that as at 15 June 2015, global influenza activity influenza activity has decreased from its peak of influenza activity in early 2015 to low levels in the Northern Hemisphere. In the southern hemisphere, influenza activity was slightly higher in most countries but remained at l ...
... The WHO6 has reported that as at 15 June 2015, global influenza activity influenza activity has decreased from its peak of influenza activity in early 2015 to low levels in the Northern Hemisphere. In the southern hemisphere, influenza activity was slightly higher in most countries but remained at l ...
Training Bulletin Influenza Educational Review 2015/2016
... to circulate as a seasonal virus for some years to come.”4 There are programs that monitor circulating strains of the flu virus (like H1N1) and assess their sensitivity to antiviral medications. The three seasonal influenzas – A, B, and C cannot be reliably predicted and as such, paramedics must rem ...
... to circulate as a seasonal virus for some years to come.”4 There are programs that monitor circulating strains of the flu virus (like H1N1) and assess their sensitivity to antiviral medications. The three seasonal influenzas – A, B, and C cannot be reliably predicted and as such, paramedics must rem ...
Swine Influenza A (H1N1 Virus): A Pandemic Disease
... Human infections with swine flu H1N1 viruses are rare. There is currently no way to differentiate antibodies produced in response to flu vaccination in pigs from antibodies made in response to pig infections with swine H1N1 influenza. While H1N1 swine viruses have been known to circulate among pig p ...
... Human infections with swine flu H1N1 viruses are rare. There is currently no way to differentiate antibodies produced in response to flu vaccination in pigs from antibodies made in response to pig infections with swine H1N1 influenza. While H1N1 swine viruses have been known to circulate among pig p ...
the registration brochure - Options IX for the Control of
... The meeting will have a more modular schedule so that attendees will have more choice in topics to design a meeting that will be personalized to their research and clinical interests. The meeting will start with an opening plenary session on Wednesday, 24 August at 5pm where we will welcome Nancy Co ...
... The meeting will have a more modular schedule so that attendees will have more choice in topics to design a meeting that will be personalized to their research and clinical interests. The meeting will start with an opening plenary session on Wednesday, 24 August at 5pm where we will welcome Nancy Co ...
Healthcare workers handbook on Influenza in SA
... Health Care Providers and Occupational Health ............................................................................. 22 Resources for further information .................................................................................................. 23 Appendix 1: Home care guidance: healt ...
... Health Care Providers and Occupational Health ............................................................................. 22 Resources for further information .................................................................................................. 23 Appendix 1: Home care guidance: healt ...
influenza surveillance in russia based on epidemiological and
... and laboratory data are entered through the internet into the electronic database at the Research Institute of Influenza (RII), where they are analyzed and then reported to the Ministry of Public Health of Russia. Simultaneously, data are introduced into WHO’s Flu Net and Euro Flu, both electronic d ...
... and laboratory data are entered through the internet into the electronic database at the Research Institute of Influenza (RII), where they are analyzed and then reported to the Ministry of Public Health of Russia. Simultaneously, data are introduced into WHO’s Flu Net and Euro Flu, both electronic d ...
Australian Influenza Surveillance Report No.03, 2013
... with fair attendance and contact with swine. The virus detected is the same as the variant viruses detected during the 2012 multi-state outbreak, which was also associated with swine exposure at agricultural fairs. The WHO has reported on an additional laboratory-confirmed case of human infection ...
... with fair attendance and contact with swine. The virus detected is the same as the variant viruses detected during the 2012 multi-state outbreak, which was also associated with swine exposure at agricultural fairs. The WHO has reported on an additional laboratory-confirmed case of human infection ...
Public Health Guidelines on the Prevention and Care Facilities in Ireland 2013/2014
... infection is lower. In elderly nursing home residents, well matched vaccines were found to be 46% effective in preventing pneumonia and 60% effective in reducing all cause mortality. ...
... infection is lower. In elderly nursing home residents, well matched vaccines were found to be 46% effective in preventing pneumonia and 60% effective in reducing all cause mortality. ...
Public Health Guidelines on the Prevention and Management of
... infection is lower. In elderly nursing home residents, well matched vaccines were found to be 46% effective in preventing pneumonia and 60% effective in reducing all-cause mortality. ...
... infection is lower. In elderly nursing home residents, well matched vaccines were found to be 46% effective in preventing pneumonia and 60% effective in reducing all-cause mortality. ...
Parallel evolution of influenza across multiple spatiotemporal scales
... We suspected that the complex dynamics of these within-host mutations might arise from competition among mutant lineages. The influenza genome consists of eight linear segments that freely reassort with one another but do not recombine (Boni et al., 2008), meaning that each segment evolves clonally. ...
... We suspected that the complex dynamics of these within-host mutations might arise from competition among mutant lineages. The influenza genome consists of eight linear segments that freely reassort with one another but do not recombine (Boni et al., 2008), meaning that each segment evolves clonally. ...
australian influenza surveillance report
... a B/Massachusetts/2/2012-like virus. Additionally, WHO recommended that quadrivalent vaccines containing two influenza B viruses contain the above three viruses and a B/Brisbane/60/2008-like virus. In comparison to the current 2013 southern hemisphere vaccine, this recommendation changed the B com ...
... a B/Massachusetts/2/2012-like virus. Additionally, WHO recommended that quadrivalent vaccines containing two influenza B viruses contain the above three viruses and a B/Brisbane/60/2008-like virus. In comparison to the current 2013 southern hemisphere vaccine, this recommendation changed the B com ...
PPT - Institute for Mathematical Sciences
... “Moreover, confinement to bed may reduce or increase an individual’s opportunities for transmission, depending on living conditions and hygiene.” - Yes! Exactly the point. If friends come to visit (especially in boarding schools), e.g. room-mates, etc., a degree of secondary infection can continue – ...
... “Moreover, confinement to bed may reduce or increase an individual’s opportunities for transmission, depending on living conditions and hygiene.” - Yes! Exactly the point. If friends come to visit (especially in boarding schools), e.g. room-mates, etc., a degree of secondary infection can continue – ...
National Preparedness Plan
... high collective immunity, and are thus capable of causing an epidemic among the population. Such changes in the dominant virus are termed “antigenic drift”. Typical influenza outbreaks will cause an increase in the incidence of pneumonia, reflecting in an increased number of hospitalisations and hig ...
... high collective immunity, and are thus capable of causing an epidemic among the population. Such changes in the dominant virus are termed “antigenic drift”. Typical influenza outbreaks will cause an increase in the incidence of pneumonia, reflecting in an increased number of hospitalisations and hig ...
COMMITTEE ON INFECTIOUS DISEASES ; originally published online September 2, 2013; Pediatrics
... Cardiovascular disease includes include conditions such as coronary heart disease, cardiac valve disorders, congestive heart failure, pulmonary hypertension, and aortic stenosis. It does not include hypertension disease only. Chronic lung disease includes conditions such as bronchitis obliterans, ch ...
... Cardiovascular disease includes include conditions such as coronary heart disease, cardiac valve disorders, congestive heart failure, pulmonary hypertension, and aortic stenosis. It does not include hypertension disease only. Chronic lung disease includes conditions such as bronchitis obliterans, ch ...
Guidance for Infection Prevention and Control
... Influenza (commonly referred to as ‘the flu’) is a virus that causes respiratory disease. Typical symptoms include fever, cough and muscle ache. In some people, complications can cause pneumonia and death. Due to its genetic make-up, the influenza virus can change and adapt, so new strains that are ...
... Influenza (commonly referred to as ‘the flu’) is a virus that causes respiratory disease. Typical symptoms include fever, cough and muscle ache. In some people, complications can cause pneumonia and death. Due to its genetic make-up, the influenza virus can change and adapt, so new strains that are ...
Oseltamivir
Oseltamivir INN /ɒsəlˈtæmɨvɪər/, marketed under the trade name Tamiflu, is an antiviral medication used to treat influenza A and influenza B (flu), and to prevent flu after exposure. The medication is taken orally.The Infectious Disease Society of America, the United States' Centers for Disease Control and Prevention (CDC), and the United Kingdom's National Institute for Health and Care Excellence recommend the use of oseltamavir for people who have complications or are at high risk for complications who present within 48 hours of first symptoms of infection. They recommend its use to prevent infection in at-risk people but not the general population. The CDC recommends that clinicians use their discretion to treat those at lower risk who present within 48 hours of first symptoms of infection. However, these recommendations are controversial as are criticisms of the recommendations. A Cochrane review concluded that oseltamivir does not reduce hospitalizations, and that there is no evidence of reduction in complications of influenza (such as pneumonia) because of a lack of diagnostic definitions, or reduction of the spread of the virus. Two meta-analyses have concluded that benefits in those who are otherwise healthy do not outweigh its risks. They also found little evidence regarding whether treatment changes the risk of hospitalization or death in high risk populations. However, another meta-analysis found that oseltamivir was effective for prevention of influenza at the individual and household levels.Side effects include psychiatric symptoms, increased rates of vomiting, and headaches. It is pregnancy category C in the United States and category B in Australia meaning that it has been taken by a small number of women without signs of problems and in animal studies it looks safe. Dose adjustment may be needed in those with kidney problems.It was the first orally administered neuraminidase inhibitor commercially developed. It was discovered and developed by US-based Gilead Sciences, which licensed the exclusive rights to Roche in 1996. It is on the World Health Organization's List of Essential Medicines, a list of the most important medications needed in a basic health system.