THE STUDY OF INTERMEDIARY METABOLISM OF
... as permit accumulation of interinediates or end-products: Use of WInatural compounds, or of diets unduly enriched or poor in test-subst,ances, experiments on poisoned, or sick organisms or on isolated organs, tissues or extracts. The isotopes of the elements C, H, N, S, 0, which constitute organic m ...
... as permit accumulation of interinediates or end-products: Use of WInatural compounds, or of diets unduly enriched or poor in test-subst,ances, experiments on poisoned, or sick organisms or on isolated organs, tissues or extracts. The isotopes of the elements C, H, N, S, 0, which constitute organic m ...
E:\My Documents\sch4u\SCH4U review McKay answers.wpd
... 2) You are given a 100.0 mL sample of 0.0010 M silver nitrate and 100.0 mL of solution made by dissolving 6.4X10-4 g of sodium carbonate (not sodium bicarbonate) in enough water to make 100.0 mL of solution. If the two solutions are mixed at 25/C, will a precipitate of silver carbonate form? (Ans; ...
... 2) You are given a 100.0 mL sample of 0.0010 M silver nitrate and 100.0 mL of solution made by dissolving 6.4X10-4 g of sodium carbonate (not sodium bicarbonate) in enough water to make 100.0 mL of solution. If the two solutions are mixed at 25/C, will a precipitate of silver carbonate form? (Ans; ...
Uric Acid and Kidney Stones
... are one of the few stone types that may be A high concentration of uric acid in the urine is not only a risk factor for uric acid stones but it is also a risk factor for the development of other types of stones (e.g. calcium oxalate stones). Uric acid crystals can form very quickly under the right c ...
... are one of the few stone types that may be A high concentration of uric acid in the urine is not only a risk factor for uric acid stones but it is also a risk factor for the development of other types of stones (e.g. calcium oxalate stones). Uric acid crystals can form very quickly under the right c ...
the pdf of this lesson!
... Try a “Tums”. You will need an antacid tablet of the type that has calcium in it such as “Tums” and a little vinegar. The active ingredient in “Tums” is calcium carbonate. Place half of a “Tums” tablet in a cup and add 2 teaspoons of water. Stir with a stick. Notice that the white particles will not ...
... Try a “Tums”. You will need an antacid tablet of the type that has calcium in it such as “Tums” and a little vinegar. The active ingredient in “Tums” is calcium carbonate. Place half of a “Tums” tablet in a cup and add 2 teaspoons of water. Stir with a stick. Notice that the white particles will not ...
CH4 Student Revision Guides pdf | GCE AS/A
... sweeping. The sample is subjected to radio frequency electromagnetic radiation and a receiver can detect absorption of radiation by the sample for a given magnetic field strength and a fixed radio frequency. All factors are linked to a computer and recorder. ...
... sweeping. The sample is subjected to radio frequency electromagnetic radiation and a receiver can detect absorption of radiation by the sample for a given magnetic field strength and a fixed radio frequency. All factors are linked to a computer and recorder. ...
Chemistry - An Introduction for Medical and Hea..
... year, thousands of plant samples are collected by drug companies to find out whether they have any anti-disease activity. Many of them do. In the mean time, we continue to destroy the rain forests just to obtain teak furniture or some extra peanuts, but that is another story. This area of research is ...
... year, thousands of plant samples are collected by drug companies to find out whether they have any anti-disease activity. Many of them do. In the mean time, we continue to destroy the rain forests just to obtain teak furniture or some extra peanuts, but that is another story. This area of research is ...
Chemistry: An Introduction for Medical and Health Sciences - E
... year, thousands of plant samples are collected by drug companies to find out whether they have any anti-disease activity. Many of them do. In the mean time, we continue to destroy the rain forests just to obtain teak furniture or some extra peanuts, but that is another story. This area of research is ...
... year, thousands of plant samples are collected by drug companies to find out whether they have any anti-disease activity. Many of them do. In the mean time, we continue to destroy the rain forests just to obtain teak furniture or some extra peanuts, but that is another story. This area of research is ...
Dr David`s Chemistry Test Answers
... 28. No (although they are very stable!) 29. Yes (when heated and plunged into liquid oxygen). 30. Solid/gas. 31. Because both form dioxides) 32. Sodium. 33. No. 34. Left. 35. Middle section. ...
... 28. No (although they are very stable!) 29. Yes (when heated and plunged into liquid oxygen). 30. Solid/gas. 31. Because both form dioxides) 32. Sodium. 33. No. 34. Left. 35. Middle section. ...
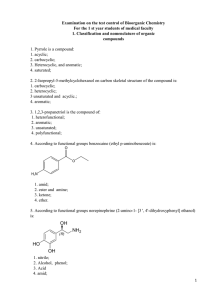
2. 2-Isopropyl-5-methylcyclohexanol on carbon skeletal
... 23. Energy of 2-chlorobutane in the eclipsed conformation more than gauche because in the eclipsed conformation: 1. The other configuration of the molecule; 2. The greater torsional stress; 3. increased Van der Waals repulsion. 4. the molecule another electronic structure; 24. The conformations of 1 ...
... 23. Energy of 2-chlorobutane in the eclipsed conformation more than gauche because in the eclipsed conformation: 1. The other configuration of the molecule; 2. The greater torsional stress; 3. increased Van der Waals repulsion. 4. the molecule another electronic structure; 24. The conformations of 1 ...
Acid + Base Class # 1
... Using this chart, let’s count how much stronger or weaker these solutions are from their partners in the chart. NOTE: acids are stronger than other acids, or weaker. Bases are compared to each other as well. We will not compare acids to bases (apples and oranges!). It can be done mathematically, but ...
... Using this chart, let’s count how much stronger or weaker these solutions are from their partners in the chart. NOTE: acids are stronger than other acids, or weaker. Bases are compared to each other as well. We will not compare acids to bases (apples and oranges!). It can be done mathematically, but ...
1. (a) Propan-1ol, C2H5CH2OH can be oxidised to propanoic acid
... to alleviate sunburn. Benzocaine reacts with dilute acids to form the ion C9H12O2N+ and with ethanoyl chloride to form C11H13O3N. When benzocaine is heated under reflux with aqueous sodium hydroxide and the solution obtained is neutralised, two compounds X and Y are formed. X has a formula of ...
... to alleviate sunburn. Benzocaine reacts with dilute acids to form the ion C9H12O2N+ and with ethanoyl chloride to form C11H13O3N. When benzocaine is heated under reflux with aqueous sodium hydroxide and the solution obtained is neutralised, two compounds X and Y are formed. X has a formula of ...
Strecker Degradation Products of Aspartic and Glutamic Acids and
... the same methods. It was found that the degradation reactions of amino acids are complex. Amino acids are principally degraded via the corresponding α-keto acids to Strecker aldehydes (aspartic acid to oxalacetic and 3-oxopropionic acids and glutamic acid to α-ketoglutaric and 4-oxobutyric acids), w ...
... the same methods. It was found that the degradation reactions of amino acids are complex. Amino acids are principally degraded via the corresponding α-keto acids to Strecker aldehydes (aspartic acid to oxalacetic and 3-oxopropionic acids and glutamic acid to α-ketoglutaric and 4-oxobutyric acids), w ...
Supplementary Information
... carbonaceous chondrites was estimated in the range from 0.22 % to 3% in weight. At least 70% of the total carbon content is the insoluble fraction (Kerogen-like material), which can be detected only after dissolution of the inorganic matrix with HF-HCl mixture. The resting 30% corresponds to the sol ...
... carbonaceous chondrites was estimated in the range from 0.22 % to 3% in weight. At least 70% of the total carbon content is the insoluble fraction (Kerogen-like material), which can be detected only after dissolution of the inorganic matrix with HF-HCl mixture. The resting 30% corresponds to the sol ...
Acid‒base reaction
... reactions, devised by Gilbert N. Lewis in 1923,[12] in the same year as Brønsted–Lowry, but it was not elaborated by him until 1938.[2] Instead of defining acid–base reactions in terms of protons or other bonded substances, the Lewis definition defines a base (referred to as a Lewis base) to be a co ...
... reactions, devised by Gilbert N. Lewis in 1923,[12] in the same year as Brønsted–Lowry, but it was not elaborated by him until 1938.[2] Instead of defining acid–base reactions in terms of protons or other bonded substances, the Lewis definition defines a base (referred to as a Lewis base) to be a co ...
View PDF - CiteSeerX
... relates gene products that encode enzymes to the reactions these enzymes catalyze. EcoCyc also includes transport reactions and signaling pathways. 1.1 Metabolic Analysis and Nutrition-Related Studies Metabolic analysis is usually carried out through quantitative calculation of the fluxes of chemica ...
... relates gene products that encode enzymes to the reactions these enzymes catalyze. EcoCyc also includes transport reactions and signaling pathways. 1.1 Metabolic Analysis and Nutrition-Related Studies Metabolic analysis is usually carried out through quantitative calculation of the fluxes of chemica ...
the Main-Group Metals - McQuarrie General Chemistry
... The alkaline-earth metals—beryllium, magnesium, calcium, strontium, barium, and radium—occur in Group 2 in the periodic table (Figure I.1). Beryllium is a relatively rare element but occurs as localized surface deposits in the mineral beryl (Figure I.2). Essentially unlimited quantities of magnesium ...
... The alkaline-earth metals—beryllium, magnesium, calcium, strontium, barium, and radium—occur in Group 2 in the periodic table (Figure I.1). Beryllium is a relatively rare element but occurs as localized surface deposits in the mineral beryl (Figure I.2). Essentially unlimited quantities of magnesium ...
1.02 x 10 = 3 mol lit 3.4 x 10
... The colour of halogens is due to the fact that their molecules absorb radiations from visible light and the outer electrons are easily excited to higher energy levels. The amount of energy required for excitation depends upon the size of the atom. Fluorine atom is the smallest and the force of attra ...
... The colour of halogens is due to the fact that their molecules absorb radiations from visible light and the outer electrons are easily excited to higher energy levels. The amount of energy required for excitation depends upon the size of the atom. Fluorine atom is the smallest and the force of attra ...
Fluorinated Butatrienes - diss.fu-berlin.de
... wurde ein Enin-Isomer entdeckt, das erstaunlicherweise stabiler als sein Butatrien Isomer ist, obwohl es an der Dreifachbindung fluoriert ist. Eben jene Fluorierung an der Dreifachbindung ist eigentlich notwendig um die Energie fluorierter But-1-en-3-ine relativ ...
... wurde ein Enin-Isomer entdeckt, das erstaunlicherweise stabiler als sein Butatrien Isomer ist, obwohl es an der Dreifachbindung fluoriert ist. Eben jene Fluorierung an der Dreifachbindung ist eigentlich notwendig um die Energie fluorierter But-1-en-3-ine relativ ...
getting started 3.1 hydrocarbons
... condense. As the gases ascend higher, the smaller molecules also condense. As each fraction condenses, the liquid formed is collected on trays. This fraction boils at 10°C; that is, it is a liquid below 10°C because it has not yet boiled, and is a gas above 10°C because it has already boiled. Theref ...
... condense. As the gases ascend higher, the smaller molecules also condense. As each fraction condenses, the liquid formed is collected on trays. This fraction boils at 10°C; that is, it is a liquid below 10°C because it has not yet boiled, and is a gas above 10°C because it has already boiled. Theref ...
Energy Matters - Perth Grammar
... The number of H+ (aq) ions in the beaker decreased. The pH of the solution decreased. The number of SO42−(aq) ions in the beaker decreased. Water molecules formed during the reaction. A precipitate formed during the reaction. The final solution contained equal numbers of H+(aq) and OH− (aq) ions. ...
... The number of H+ (aq) ions in the beaker decreased. The pH of the solution decreased. The number of SO42−(aq) ions in the beaker decreased. Water molecules formed during the reaction. A precipitate formed during the reaction. The final solution contained equal numbers of H+(aq) and OH− (aq) ions. ...
WHAT YOU EAT - Montana State University Extended University
... burn fat-‐-‐if there is enough oxygen in your tissues. If your tissues are short of oxygen (usually because of high exertion or if you are holding your breath for a long time) your cells burn ...
... burn fat-‐-‐if there is enough oxygen in your tissues. If your tissues are short of oxygen (usually because of high exertion or if you are holding your breath for a long time) your cells burn ...
09_Lecture
... • Some antacids are mixtures of aluminum hydroxide and magnesium hydroxide. – Aluminum hydroxide produces constipation and binds phosphate in the intestinal tract, which may cause weakness and loss of appetite. – Magnesium hydroxide has a laxative effect. ...
... • Some antacids are mixtures of aluminum hydroxide and magnesium hydroxide. – Aluminum hydroxide produces constipation and binds phosphate in the intestinal tract, which may cause weakness and loss of appetite. – Magnesium hydroxide has a laxative effect. ...
Questions
... Calculate the volume of gas produced if 3.0 g of sodium reacts with an excess of water. (One mole of any gas at the temperature and pressure of the experiment occupies 24 dm3.) ...
... Calculate the volume of gas produced if 3.0 g of sodium reacts with an excess of water. (One mole of any gas at the temperature and pressure of the experiment occupies 24 dm3.) ...
advanced chemistry may 2011 marking scheme
... (ii) Explain why all bond angles in the carbonate ion are equal. Explanation in terms of delocalized electrons (or resonance hybrid) to produce three equal bonds, repulsion between electron density of which is equal thus generating a species with bond angle of 120o. (2 marks) (d) Excess hydrochloric ...
... (ii) Explain why all bond angles in the carbonate ion are equal. Explanation in terms of delocalized electrons (or resonance hybrid) to produce three equal bonds, repulsion between electron density of which is equal thus generating a species with bond angle of 120o. (2 marks) (d) Excess hydrochloric ...
Biological aspects of fluorine

Fluorine, a poisonous gas in its elemental form at biological temperatures, has been a subject of significant interest for a broad range of biological applications, including ecology, medical science, and biochemical engineering.Among the most reactive of the elements, it has proved valuable in many potent industrial compounds, such as the weak (but very toxic) acid hydrogen fluoride, which are quite dangerous to living organisms. Fluorine is a component of so-called ""1080"" poison, a mammal-killer banned in much of the world but still used to control populations of Australian foxes and American coyotes.Because carbon-fluorine bonds are difficult to form, they are seldom found in nature. A few species of plants and bacteria found in the tropics make fluorine-containing poisons to deter predators from eating them. The same bond makes fluorination a powerful lever for new drug design, allowing the tweaking of organic molecules in innovative ways which has led to several blockbuster commercial successes, such as Lipitor and Prozac.In dental products, when applied topically the fluoride ion chemically binds to surface tooth enamel, making it marginally more acid-resistant. Although politically controversial, fluoridation of public water supplies has shown consistent benefits to dental hygiene, especially for poor children.Manmade fluorinated compounds have also played roles in several noteworthy environmental concerns. Chlorofluorocarbons, once major components of numerous commercial aerosol products, have proven damaging to the Earth's ozone layer and resulted in the wide-reaching Montreal Protocol (though in truth the chlorine in CFCs is the destructive actor, fluorine is an important part of these molecules because it makes them very stable and long-lived). Similarly, the stability of many organofluorines has raised the issue of biopersistence. Long-lived molecules from waterproofing sprays, PFOA and PFOS, are found worldwide in wildlife and humans, including newborn children.Fluorine biology is also relevant to a number of cutting-edge technologies. PFCs (perfluorocarbons) are capable of holding enough oxygen to support human liquid breathing. Several works of science fiction have touched on this, but in the real world, researchers have experimented with PFCs for burned lung care and as blood substitutes. Fluorine in the form of its radioisotope F-18 is also at the heart of a modern medical imaging technique known as positron emission tomography (PET). A PET scan produces three-dimensional colored images of parts of the body that use a lot of sugar, particularly the brain or tumors.