PDF (Free)
... titanium is still used to a much lesser extent than iron (Fe) or aluminum (Al), mainly because of low productivity and high production costs involved at all processing stages.1) For example, the cost of processing titanium ore is 15 times higher than that required for processing iron ore.25) To red ...
... titanium is still used to a much lesser extent than iron (Fe) or aluminum (Al), mainly because of low productivity and high production costs involved at all processing stages.1) For example, the cost of processing titanium ore is 15 times higher than that required for processing iron ore.25) To red ...
CHAPTER 20 METALLURGY AND THE CHEMISTRY OF METALS
... The trick in this process centers on the fact that TiCl4 is a liquid with a boiling point (136.4°C), a little higher than that of water. The tetrachloride can be formed by treating the oxide (rutile) with chlorine gas at high temperature. The balanced equation is: TiO2(s) + 2Cl2(g) → TiCl4(l) + O2(g ...
... The trick in this process centers on the fact that TiCl4 is a liquid with a boiling point (136.4°C), a little higher than that of water. The tetrachloride can be formed by treating the oxide (rutile) with chlorine gas at high temperature. The balanced equation is: TiO2(s) + 2Cl2(g) → TiCl4(l) + O2(g ...
Module-2-s-and-d-elements - Львівський національний медичний
... Рецензент: проф. Й.Д. Комариця – професор кафедри фармацевтичної, органічної та біоорганічної хімії ЛНМУ імені Данила Галицького. ...
... Рецензент: проф. Й.Д. Комариця – професор кафедри фармацевтичної, органічної та біоорганічної хімії ЛНМУ імені Данила Галицького. ...
What is a Mineral?
... • Mine reclamation is the process by which land used for mining is returned to its original state or better. • Has been required by law since the mid-1970’s. • To reduce the effects of mining, reduce our need for minerals, reuse, and recycle. ...
... • Mine reclamation is the process by which land used for mining is returned to its original state or better. • Has been required by law since the mid-1970’s. • To reduce the effects of mining, reduce our need for minerals, reuse, and recycle. ...
The Alkaline Earth Metals (Group 2) - Chemwiki
... The only other alkaline earth that is widely used as the metal is beryllium, which is extremely toxic. Ingestion of beryllium or exposure to berylliumcontaining dust causes a syndrome called berylliosis, characterized by severe inflammation of the respiratory tract or other tissues. A small percenta ...
... The only other alkaline earth that is widely used as the metal is beryllium, which is extremely toxic. Ingestion of beryllium or exposure to berylliumcontaining dust causes a syndrome called berylliosis, characterized by severe inflammation of the respiratory tract or other tissues. A small percenta ...
s-BLOCK ELEMENTS - einstein classes
... alkali metals. Because they aremore closely packed due to increasing nuclear charge. (iii) Atomic size : The atomic size of alkaline earth metals are smaller than the corresponding alkali metals. Alkaline earth metals have a higher nuclear charge and therefore, electrons are attracted more towards t ...
... alkali metals. Because they aremore closely packed due to increasing nuclear charge. (iii) Atomic size : The atomic size of alkaline earth metals are smaller than the corresponding alkali metals. Alkaline earth metals have a higher nuclear charge and therefore, electrons are attracted more towards t ...
KISS Notes
... Before metals, people used tools mainly made from a)............................. or ................................. The first metal used was probably b)................................., because it occurs in the elemental state in nature. However, it is too soft to be used for tools, so was just ...
... Before metals, people used tools mainly made from a)............................. or ................................. The first metal used was probably b)................................., because it occurs in the elemental state in nature. However, it is too soft to be used for tools, so was just ...
Slide 1
... CuFeS2(s) + 4O2(g) → CuSO4(s) + FeSO4(s) 4CuFeS2(s) + 13O2(g) → 4CuO(s) + 2Fe2O3(s) + 8SO2(g) • Reactions are exothermic, roasting is an autogenous process requiring little or no additional fuel. • NB, not all the sulphides are oxidised, only around 1/3. Rest remain as sulphide minerals. • The gas ...
... CuFeS2(s) + 4O2(g) → CuSO4(s) + FeSO4(s) 4CuFeS2(s) + 13O2(g) → 4CuO(s) + 2Fe2O3(s) + 8SO2(g) • Reactions are exothermic, roasting is an autogenous process requiring little or no additional fuel. • NB, not all the sulphides are oxidised, only around 1/3. Rest remain as sulphide minerals. • The gas ...
Determination of the Empirical Formula of an
... While a few metals, such as gold and silver, can be found in their elemental forms in nature, the majority of metals are naturally found in mineral ores in the form of compounds.1 These metals must be extracted from their ores through a process known as smelting.2 The first metals to be smelted, mor ...
... While a few metals, such as gold and silver, can be found in their elemental forms in nature, the majority of metals are naturally found in mineral ores in the form of compounds.1 These metals must be extracted from their ores through a process known as smelting.2 The first metals to be smelted, mor ...
s - Wits Structural Chemistry
... CuFeS2(s) + 4O2(g) → CuSO4(s) + FeSO4(s) 4CuFeS2(s) + 13O2(g) → 4CuO(s) + 2Fe2O3(s) + 8SO2(g) • Reactions are exothermic, roasting is an autogenous process requiring little or no additional fuel. • NB, not all the sulphides are oxidised, only around 1/3. Rest remain as sulphide minerals. • The gas ...
... CuFeS2(s) + 4O2(g) → CuSO4(s) + FeSO4(s) 4CuFeS2(s) + 13O2(g) → 4CuO(s) + 2Fe2O3(s) + 8SO2(g) • Reactions are exothermic, roasting is an autogenous process requiring little or no additional fuel. • NB, not all the sulphides are oxidised, only around 1/3. Rest remain as sulphide minerals. • The gas ...
Document
... of electrons are available for bonding. There is only a ____________________ in successive ionization energies of the d electrons. Thus when elements of the first transition series react to form compounds, they can form ions of roughly the ___________________ by losing different numbers of electrons ...
... of electrons are available for bonding. There is only a ____________________ in successive ionization energies of the d electrons. Thus when elements of the first transition series react to form compounds, they can form ions of roughly the ___________________ by losing different numbers of electrons ...
Chapter 23 Metals and Metallurgy
... Steel • The impurities are oxidized by O2 (except phosphorus, which reacts with CaO) to compounds easily separated from the molten iron. • Purified molten steel is poured into molds. ...
... Steel • The impurities are oxidized by O2 (except phosphorus, which reacts with CaO) to compounds easily separated from the molten iron. • Purified molten steel is poured into molds. ...
Group 1: The Alkali Metals
... Atomic number: 11 Atomic weight: 22.9897 soft silvery metal. extremely reactive metal Electron configuration: [Ne]3s1 used in nuclear reactors because of its low boiling point. Sodium is reacted with chlorine to produce the ionic halide, NaCl o Sodium chloride is an important part of human diet It ...
... Atomic number: 11 Atomic weight: 22.9897 soft silvery metal. extremely reactive metal Electron configuration: [Ne]3s1 used in nuclear reactors because of its low boiling point. Sodium is reacted with chlorine to produce the ionic halide, NaCl o Sodium chloride is an important part of human diet It ...
Chapter 23 Metals and Metallurgy
... compounds known as minerals. • Minerals are named by common, not chemical, names. ...
... compounds known as minerals. • Minerals are named by common, not chemical, names. ...
The s-Block Elements - GCG-42
... BeCl2 is essentially covalent, with comparatively low m.pt. The lower members in group II form essentially ionic chlorides, with Mg having intermediate properties. ...
... BeCl2 is essentially covalent, with comparatively low m.pt. The lower members in group II form essentially ionic chlorides, with Mg having intermediate properties. ...
the Main-Group Metals - McQuarrie General Chemistry
... The alkaline-earth metals—beryllium, magnesium, calcium, strontium, barium, and radium—occur in Group 2 in the periodic table (Figure I.1). Beryllium is a relatively rare element but occurs as localized surface deposits in the mineral beryl (Figure I.2). Essentially unlimited quantities of magnesium ...
... The alkaline-earth metals—beryllium, magnesium, calcium, strontium, barium, and radium—occur in Group 2 in the periodic table (Figure I.1). Beryllium is a relatively rare element but occurs as localized surface deposits in the mineral beryl (Figure I.2). Essentially unlimited quantities of magnesium ...
01.CN_Other pages/p1-9
... • Heating the mixture / increasing the temperature would increase the rate of chemical reaction. This is because there is a larger number of particles with ...
... • Heating the mixture / increasing the temperature would increase the rate of chemical reaction. This is because there is a larger number of particles with ...
MATИ” РГТУ им. К.Э. Циолковского
... • Modification of traditional materials to make them active biocidal properties: different kinds of packaging materials and packaging, textiles and textile products, paints and other building materials, paper and paperboard, bandages, plastics and film, ceramics and glass, other materials and coatin ...
... • Modification of traditional materials to make them active biocidal properties: different kinds of packaging materials and packaging, textiles and textile products, paints and other building materials, paper and paperboard, bandages, plastics and film, ceramics and glass, other materials and coatin ...
Learning objectives C8.2 Chemical Reactions Reactivity of Metals
... Be able to know a variety of methods of extracting a salt from reactions of acids Acid + metal Acid + metal oxide Acid + metal carbonate ...
... Be able to know a variety of methods of extracting a salt from reactions of acids Acid + metal Acid + metal oxide Acid + metal carbonate ...
Elements – (Metals)
... Bronsted Lowry acid and base also fits Lewis definition but there is also a more general definition Acid Base ...
... Bronsted Lowry acid and base also fits Lewis definition but there is also a more general definition Acid Base ...
Unit 10
... Extracting metal – getting metal from ores. What are the metal compounds from mineral ores? Are they soluble in water? Insoluble metal oxides, carbonates and sulphides – found in ores Which metals are found free in nature? Unreactive metals such as gold and platinum found free (as elements) in natur ...
... Extracting metal – getting metal from ores. What are the metal compounds from mineral ores? Are they soluble in water? Insoluble metal oxides, carbonates and sulphides – found in ores Which metals are found free in nature? Unreactive metals such as gold and platinum found free (as elements) in natur ...
Types of Materials
... Aluminium is a metallic element. It is a soft metal with a silvery appearance. Aluminium exists in a metal lattice structure as Al3+ ions in a 'sea' of electrons where the bonding between the ions and the free electrons is very strong. This gives the aluminium a tightly bound metallic lattice making ...
... Aluminium is a metallic element. It is a soft metal with a silvery appearance. Aluminium exists in a metal lattice structure as Al3+ ions in a 'sea' of electrons where the bonding between the ions and the free electrons is very strong. This gives the aluminium a tightly bound metallic lattice making ...
Single Replacement Reactions
... chemically combined in a compound. The tendency of a particular element to combine with other substances is a measure of the activity of the element. The more active an element is, the more likely it is to combine. In a single replacement reaction, an uncombined element replaces a less active elemen ...
... chemically combined in a compound. The tendency of a particular element to combine with other substances is a measure of the activity of the element. The more active an element is, the more likely it is to combine. In a single replacement reaction, an uncombined element replaces a less active elemen ...
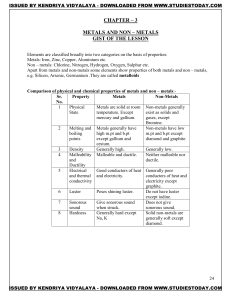
METALS AND NON – METALS Concepts
... 4. Conduction of electricity:ionic compounds in solid state-----does not conduct electricity. Reason—Ions can not move due to rigid solid structure. Ionic compounds conduct electricity in molten state. Reason-- Ions can move freely since the electrostatic forces of attraction between the oppositely ...
... 4. Conduction of electricity:ionic compounds in solid state-----does not conduct electricity. Reason—Ions can not move due to rigid solid structure. Ionic compounds conduct electricity in molten state. Reason-- Ions can move freely since the electrostatic forces of attraction between the oppositely ...
De re metallica

De re metallica (Latin for On the Nature of Metals (Minerals)) is a book cataloguing the state of the art of mining, refining, and smelting metals, published a year posthumously in 1556 due to a delay in preparing woodcuts for the text. The author was Georg Bauer, whose pen name was the Latinized Georgius Agricola. The book remained the authoritative text on mining for 180 years after its publication. It was also an important chemistry text for the period and is significant in the history of chemistry.Agricola had spent nine years in the Bohemian town of Joachimsthal, now in the Czech Republic. (Joachimsthal is famous for its silver mines and the origin of the word ""Thaler"" and, ultimately, ""dollar."") After Joachimsthal, he spent the rest of his life in Chemnitz, a prominent mining town in Saxony. Both Joachimsthal and Chemnitz are in the Erzgebirge, or Ore Mountains.The book was greatly influential, and for more than a century after it was published, De Re Metallica remained a standard treatise used throughout Europe. The German mining technology it portrayed was acknowledged as the most advanced at the time, and the metallic wealth produced in German mining districts was the envy of many other European nations. The book was reprinted in a number of Latin editions, as well as in German and Italian translations. Publication in Latin meant that it could be read by any educated European of the time. The numerous woodcuts and detailed descriptions of machinery made it a practical reference for those wishing to replicate the latest in mining technology.In 1912, the first English translation of De Re Metallica was privately published in London by subscription. The translators were Herbert Hoover, a mining engineer (and later President of the United States), and his wife, Lou Henry Hoover, a geologist and Latinist. The translation is notable not only for its clarity of language, but for the extensive footnotes, which detail the classical references to mining and metals. Subsequent translations into other languages, including German, owe much to the Hoover translations, as their footnotes detail their difficulties with Agricola's invention of several hundred Latin expressions to cover Mediaeval German mining and milling terms that were unknown to classical Latin.