(pdf)
... if the energy of the particle at the exobase is lower than the escape energy, the particle is assumed to start a ballistic trajectory in the exosphere. This trajectory will lead it back to crossing the exobase a second time, but in the downward direction. Although particles crossing the exobase upwa ...
... if the energy of the particle at the exobase is lower than the escape energy, the particle is assumed to start a ballistic trajectory in the exosphere. This trajectory will lead it back to crossing the exobase a second time, but in the downward direction. Although particles crossing the exobase upwa ...
Modeling of Energy Transfer From Vibrationally Excited CO2
... semiempirical formulation that allows the colliding molecules to be stretchable, rather than frozen at their equilibrium geometry. In a previous work, the same potential energy surface has been used to show that modifications in the geometry (and in physical properties such as polarizability and char ...
... semiempirical formulation that allows the colliding molecules to be stretchable, rather than frozen at their equilibrium geometry. In a previous work, the same potential energy surface has been used to show that modifications in the geometry (and in physical properties such as polarizability and char ...
Unit 5 1 Thermodynamics Intro / Thermochemistry/ Entropy
... driven by a decrease in enthalpy or Essential knowledge 5.E.2: Some physical or chemical processes involve both a decrease in the internal an increase in entropy, or both. energy of the components (ΔH ° < 0) under consideration and an increase in the entropy of those components (ΔS ° > 0). These pro ...
... driven by a decrease in enthalpy or Essential knowledge 5.E.2: Some physical or chemical processes involve both a decrease in the internal an increase in entropy, or both. energy of the components (ΔH ° < 0) under consideration and an increase in the entropy of those components (ΔS ° > 0). These pro ...
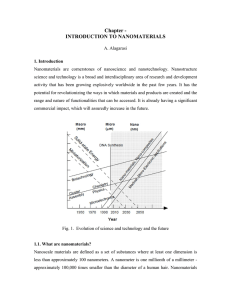
Chapter - INTRODUCTION TO NANOMATERIALS
... products and processes. They can be found in such things as sunscreens, cosmetics, sporting goods, stain-resistant clothing, tires, electronics, as well as many other everyday items, and are used in medicine for purposes of diagnosis, imaging and drug delivery. Engineered nanomaterials are resources ...
... products and processes. They can be found in such things as sunscreens, cosmetics, sporting goods, stain-resistant clothing, tires, electronics, as well as many other everyday items, and are used in medicine for purposes of diagnosis, imaging and drug delivery. Engineered nanomaterials are resources ...
Accurate van der Waals interactions from groundstate
... (50 atoms and molecules – 1225 interaction pairs) ...
... (50 atoms and molecules – 1225 interaction pairs) ...
Energetics - chemistryatdulwich
... size of ion: the smaller the ion , the larger the electrostatic attraction and the more energy is needed to sublime the ions. charge of the ion: the larger the charge, the greater the electrostatic attraction. The smaller the ionic radius and the greater the charge of the ion the greater the charg ...
... size of ion: the smaller the ion , the larger the electrostatic attraction and the more energy is needed to sublime the ions. charge of the ion: the larger the charge, the greater the electrostatic attraction. The smaller the ionic radius and the greater the charge of the ion the greater the charg ...
AP_PPT_ch_17
... LO 2.15 The student is able to explain observations regarding the solubility of ionic solids and molecules in water and other solvents on the basis of particle views that include intermolecular interactions and entropic effects. LO 5.12 The student is able to use representations and models to predic ...
... LO 2.15 The student is able to explain observations regarding the solubility of ionic solids and molecules in water and other solvents on the basis of particle views that include intermolecular interactions and entropic effects. LO 5.12 The student is able to use representations and models to predic ...
Material Equilibrium
... ENTROPY AND EQUILIBRIUM Example: In isolated system (not in material equilibrium) The spontaneous chemical reaction or transport of matter are irreversible process that increase the ENTROPY The process was continued until the system’s entropy is maximized. Once it is maximized, any further pr ...
... ENTROPY AND EQUILIBRIUM Example: In isolated system (not in material equilibrium) The spontaneous chemical reaction or transport of matter are irreversible process that increase the ENTROPY The process was continued until the system’s entropy is maximized. Once it is maximized, any further pr ...
AP Thermodynamics ppt.
... • Chemical systems in equilibrium are reversible. • In any spontaneous process, the path between reactants and products is irreversible. • Thermodynamics gives us the direction of a process but it cannot predict the speed at which the process will occur. Prentice Hall © 2003 ...
... • Chemical systems in equilibrium are reversible. • In any spontaneous process, the path between reactants and products is irreversible. • Thermodynamics gives us the direction of a process but it cannot predict the speed at which the process will occur. Prentice Hall © 2003 ...
Fulltext PDF - Indian Academy of Sciences
... Abstract. We give a detailed description of the use of explicit as well as implicit solvation treatments to compute the reduction potentials of biomolecules in a medium. The explicit solvent method involves quantum mechanical/molecular mechanics (QM/MM) treatment of the solvated moiety followed by a ...
... Abstract. We give a detailed description of the use of explicit as well as implicit solvation treatments to compute the reduction potentials of biomolecules in a medium. The explicit solvent method involves quantum mechanical/molecular mechanics (QM/MM) treatment of the solvated moiety followed by a ...
Mechanistic Studies on the Galvanic Replacement Reaction
... of various noble metals could be synthesized via galvanic replacement reaction in an aqueous solution by reacting sacrificial Ag template with a precursor compound of the desired metal such as Au, Pd, or Pt. For example, Ag templates with a variety of shapes, including nanocubes, nanoplates, nanosph ...
... of various noble metals could be synthesized via galvanic replacement reaction in an aqueous solution by reacting sacrificial Ag template with a precursor compound of the desired metal such as Au, Pd, or Pt. For example, Ag templates with a variety of shapes, including nanocubes, nanoplates, nanosph ...
L-12 Spontaneity of chemical reactions
... (ii) the gaseous state is the most disordered state, hence its entropy is the maximum, and (iii) the disorder in the liquid state is intermediate between the solid and the gaseous state. When a system changes from one state to another, the change of entropy S is given by S = ...
... (ii) the gaseous state is the most disordered state, hence its entropy is the maximum, and (iii) the disorder in the liquid state is intermediate between the solid and the gaseous state. When a system changes from one state to another, the change of entropy S is given by S = ...
Chemical Thermodynamics - Winona State University
... Spontaneous Processes • A process that is spontaneous in one direction is not spontaneous in the opposite direction. • The direction of a spontaneous process can depend on temperature: Ice turning to water is spontaneous at T > 0C, Water turning to ice is spontaneous at T < 0C. Reversible and Irr ...
... Spontaneous Processes • A process that is spontaneous in one direction is not spontaneous in the opposite direction. • The direction of a spontaneous process can depend on temperature: Ice turning to water is spontaneous at T > 0C, Water turning to ice is spontaneous at T < 0C. Reversible and Irr ...
Thermodynamics PPT
... amount of work, at a given temperature and pressure, that can be done on the surroundings by a system. Never really achieved because some of the free energy is changed to heat during a change, so it can’t be used to do work. Wmax = G For a spontaneous process: Maximum amount of energy release ...
... amount of work, at a given temperature and pressure, that can be done on the surroundings by a system. Never really achieved because some of the free energy is changed to heat during a change, so it can’t be used to do work. Wmax = G For a spontaneous process: Maximum amount of energy release ...
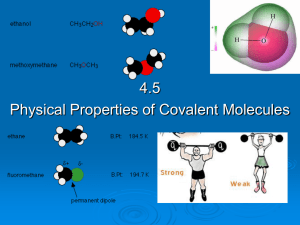
4.5 Physical properties of molecular covalent
... As the non-polar hydrocarbon chain of a polar molecule increases, the solubility of the molecule decreases because the non-polar carbon chain outweighs the polar part. E.g. ethanol (CH3CH2OH) is more soluble in water than butanol (CH3CH2CH2CH2OH) Non-polar covalent molecules are generally insoluble ...
... As the non-polar hydrocarbon chain of a polar molecule increases, the solubility of the molecule decreases because the non-polar carbon chain outweighs the polar part. E.g. ethanol (CH3CH2OH) is more soluble in water than butanol (CH3CH2CH2CH2OH) Non-polar covalent molecules are generally insoluble ...
Original powerpoint (~1.9 MB)
... regardless of the steps we add together. Change in entropy DS will work exactly the same way! As long as we get from the same initial state to the same final state then DS will be the same regardless of the steps we add together. ...
... regardless of the steps we add together. Change in entropy DS will work exactly the same way! As long as we get from the same initial state to the same final state then DS will be the same regardless of the steps we add together. ...
Chemical Thermodynamics presentation 1
... Entropy Changes – Recall entropy increases if the number of gas molecules increases; – In this reaction the number of gas molecules decrease so this is an example of a decrease in entropy. ...
... Entropy Changes – Recall entropy increases if the number of gas molecules increases; – In this reaction the number of gas molecules decrease so this is an example of a decrease in entropy. ...
spontaneous processes
... Points of note: 1. Whenever a chemical system is in equilibrium, we can go reversibly between reactants and products. 2. In any spontaneous process, the path between reactants and products is irreversible. 3. Thermodynamics refers to the direction of a reaction, not its speed. ...
... Points of note: 1. Whenever a chemical system is in equilibrium, we can go reversibly between reactants and products. 2. In any spontaneous process, the path between reactants and products is irreversible. 3. Thermodynamics refers to the direction of a reaction, not its speed. ...
PREPARATIONS AND APPLICATION OF METAL NANOPARTICLES
... is known as overlap of atomic orbitals to give nearly continuous electronic energy levels. The energy difference between the valence and conduction is called band gap (Eg). Metals that are electronically categorized as semiconductor have partially filled band (the valence band) separated from the (m ...
... is known as overlap of atomic orbitals to give nearly continuous electronic energy levels. The energy difference between the valence and conduction is called band gap (Eg). Metals that are electronically categorized as semiconductor have partially filled band (the valence band) separated from the (m ...
Cause of Chirality Consensus
... but their rates rj were varied randomly (up to 10%) during each time step. Chemical potentials were recalculated after every step of synthesis and degradation to obtain updated free energy differences. At each time step the entropy according to eq. 1 and the degree of handedness were calculated from ...
... but their rates rj were varied randomly (up to 10%) during each time step. Chemical potentials were recalculated after every step of synthesis and degradation to obtain updated free energy differences. At each time step the entropy according to eq. 1 and the degree of handedness were calculated from ...
Chemical Bonding (short)
... A) KINETICS – is the study of reaction Mechanisms and rates of chemical reactions Recall that Molecules are constantly colliding with each Other? ...
... A) KINETICS – is the study of reaction Mechanisms and rates of chemical reactions Recall that Molecules are constantly colliding with each Other? ...
Slide 1
... with increase in entropy (S positive). However, there are some processes occur spontaneously with decrease in entropy! And most of them are highly exothermic processes (H negative) Thus, the spontaneity of a reaction seems to relate both thermodynamic quantity namely Enthalpy and Entropy! Willard ...
... with increase in entropy (S positive). However, there are some processes occur spontaneously with decrease in entropy! And most of them are highly exothermic processes (H negative) Thus, the spontaneity of a reaction seems to relate both thermodynamic quantity namely Enthalpy and Entropy! Willard ...
Self-assembly of nanoparticles

Self-assembly is a phenomenon where the components of a system assemble themselves spontaneously via an interaction to form a larger functional unit. This spontaneous organization can be due to direct specific interaction and/or indirectly through their environment. Due to the increasing technological advancements, the study of materials in the nanometre scale is becoming more important. The ability to assemble nanoparticles into well-defined configuration in space is crucial to the development of electronic devices that are small but can contain plenty of information. The spatial arrangements of these self-assembled nanoparticles can be potentially used to build increasingly complex structures leading to a wide variety of materials that can be used for different purposes.At the molecular level, intermolecular force hold the spontaneous gathering of molecules into a well-defined and stable structure together. In chemical solutions, self-assembly is an outcome of random motion of molecules and the affinity of their binding sites for one another. In the area of nanotechnology, developing a simple, efficient method to organize molecules and molecular clusters into precise, pre-determined structure is crucial.