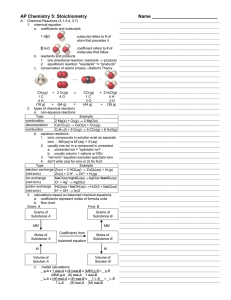
Extended Detail on Checklist items
... 10. Present “extraordinary detection claims” based on DDA mass spectrometry with high-resolution, high signal-to-noise ratio (SNR), and clearly annotated spectra. Annotated spectra (i.e. spectra with the matched peaks clearly labeled) must be provided in the supplementary material for the manuscrip ...
... 10. Present “extraordinary detection claims” based on DDA mass spectrometry with high-resolution, high signal-to-noise ratio (SNR), and clearly annotated spectra. Annotated spectra (i.e. spectra with the matched peaks clearly labeled) must be provided in the supplementary material for the manuscrip ...
Fragmentation pathway for glutamine identification: Loss of 73 da
... roles in numerous processes by analytical identification and°quantification°techniques.°Trinh°et°al.°[18]°modified Johnson’s°two-step°derivatization°method°[13]°to°distinguish glutamine and glutamic acid. Trinh’s butanol derivatization°[18]°was°performed°at°ambient°temperature and for a shorter time ...
... roles in numerous processes by analytical identification and°quantification°techniques.°Trinh°et°al.°[18]°modified Johnson’s°two-step°derivatization°method°[13]°to°distinguish glutamine and glutamic acid. Trinh’s butanol derivatization°[18]°was°performed°at°ambient°temperature and for a shorter time ...
Database Searching for Protein Identification and
... specificity of the enzyme, calculates the masses of the predicted peptides, and compares the set of calculated mass values with the set of experimental mass values. Some type of scoring is used to identify the entry in the database that gives the best match, and a report is generated. I will discuss ...
... specificity of the enzyme, calculates the masses of the predicted peptides, and compares the set of calculated mass values with the set of experimental mass values. Some type of scoring is used to identify the entry in the database that gives the best match, and a report is generated. I will discuss ...
2 Applications
... The sample is ionized at atmospheric pressure. Highly charged droplets disperse from a capillary in an electric field, evaporate and are drawn into the vacuum of the analyzer. ESI generates multiply charged ions. ESI is a soft ionization method that allows for detection of non-covalent protein compl ...
... The sample is ionized at atmospheric pressure. Highly charged droplets disperse from a capillary in an electric field, evaporate and are drawn into the vacuum of the analyzer. ESI generates multiply charged ions. ESI is a soft ionization method that allows for detection of non-covalent protein compl ...
Technologies for Proteomics
... column that is heated and points at a skimmer cone. (Too much build up on orifice) • Electrospray (ESI) – analytes are charged upon exiting the capillary tube and cross sprayed with nitrogen. The charge particles cause a “Coulomb explosion” making smaller droplets of analyte to enter the skimmer con ...
... column that is heated and points at a skimmer cone. (Too much build up on orifice) • Electrospray (ESI) – analytes are charged upon exiting the capillary tube and cross sprayed with nitrogen. The charge particles cause a “Coulomb explosion” making smaller droplets of analyte to enter the skimmer con ...
mass
... Calculating average r.a.m. from isotopes To calculate the average r.a.m. of a mixture of isotopes, multiply the percentage of each isotope( as a decimal) by its relative atomic mass and then add these together. Naturally-occurring bromine is composed of two isotopes: bromine-79 (50.5%) and bromine- ...
... Calculating average r.a.m. from isotopes To calculate the average r.a.m. of a mixture of isotopes, multiply the percentage of each isotope( as a decimal) by its relative atomic mass and then add these together. Naturally-occurring bromine is composed of two isotopes: bromine-79 (50.5%) and bromine- ...
Title: Direct Analysis of Fexofenadine and Pseudoephedrine in
... subject. Urine samples were collected 2 and 4 hours postingestion for direct analysis by LAESI-MS. A 20 µL volume of each sample was deposited in the wells of a low volume 96-well plate and analyzed by LAESI-MS using the Protea LAESI DP-1000 system integrated with a Thermo ...
... subject. Urine samples were collected 2 and 4 hours postingestion for direct analysis by LAESI-MS. A 20 µL volume of each sample was deposited in the wells of a low volume 96-well plate and analyzed by LAESI-MS using the Protea LAESI DP-1000 system integrated with a Thermo ...
A Method To Define the Carboxyl Terminal of Proteins
... of proteins.37 More recent approaches for C-terminal peptide sequence analysis include multistage MS of alkali-cationized peptides in an ion trap38 and fragmentation of intact protein ions in Fourier transform mass spectrometers.39 Notwithstanding the utility of the above-described methods, the C-te ...
... of proteins.37 More recent approaches for C-terminal peptide sequence analysis include multistage MS of alkali-cationized peptides in an ion trap38 and fragmentation of intact protein ions in Fourier transform mass spectrometers.39 Notwithstanding the utility of the above-described methods, the C-te ...
Topic 1: Quantitative chemistry
... The following laws and ideas underpin our knowledge about the relationships between chemicals involved in reactions (see PowerPoint): 1. Law of Conservation of Mass: Lavoisier (18th Century) concluded, from studying reactions involving oxygen, that during any chemical reaction no atoms were destroye ...
... The following laws and ideas underpin our knowledge about the relationships between chemicals involved in reactions (see PowerPoint): 1. Law of Conservation of Mass: Lavoisier (18th Century) concluded, from studying reactions involving oxygen, that during any chemical reaction no atoms were destroye ...
Mapping Post-Transcriptional Modifications onto Transfer
... thin‐layer chromatography, and radioisotopes [26,27]. While adequate at the time, these methods required the use of large numbers of sample and, generally, could only identify the presence of modifications outside of any tRNA sequence context. Different methods, ...
... thin‐layer chromatography, and radioisotopes [26,27]. While adequate at the time, these methods required the use of large numbers of sample and, generally, could only identify the presence of modifications outside of any tRNA sequence context. Different methods, ...
Chapter_7
... amino acids. It does not effect the search time, • Variable modifications These are modifications that may be present at some or all positions of the actual amino acid. This can (dramatically) increase the search time. If an amino acid with a possible modification occurs n times in a peptide, then t ...
... amino acids. It does not effect the search time, • Variable modifications These are modifications that may be present at some or all positions of the actual amino acid. This can (dramatically) increase the search time. If an amino acid with a possible modification occurs n times in a peptide, then t ...
Atomic Masses
... 9.5 trillion kilometers!!!!!!!!!! Why do you think we measure distances to nearby stars in light years and not kilometers????? • Consider the reaction below, In order to make one molecule of CO2 (g), each carbon atom needs how many molecules of oxygen gas???? C(s) + ...
... 9.5 trillion kilometers!!!!!!!!!! Why do you think we measure distances to nearby stars in light years and not kilometers????? • Consider the reaction below, In order to make one molecule of CO2 (g), each carbon atom needs how many molecules of oxygen gas???? C(s) + ...
Document
... Divide each by smallest (2.21 x 10-4 ): C = 1.00 Multiply each by 3: C = 3.00 = 3.0 H = 1.32 (to get ~integers) H = 3.96 = 4.0 O = 1.00 O = 3.00 = 3.0 ...
... Divide each by smallest (2.21 x 10-4 ): C = 1.00 Multiply each by 3: C = 3.00 = 3.0 H = 1.32 (to get ~integers) H = 3.96 = 4.0 O = 1.00 O = 3.00 = 3.0 ...
Thermo Scientific Solutions for Quantitative
... TO UNDERSTAND the functions of individual proteins and their place in complex biological systems, it is often necessary to measure changes in protein abundance relative to changes in the state of the system. As such, modern proteomics has evolved from an almost exclusively qualitative technique into ...
... TO UNDERSTAND the functions of individual proteins and their place in complex biological systems, it is often necessary to measure changes in protein abundance relative to changes in the state of the system. As such, modern proteomics has evolved from an almost exclusively qualitative technique into ...
Unit V The Mole
... ________________________________________________________________________ During the time that Dalton’s mass scale was just being introduced, the French chemist ______________________________ began to study how gases reacted. When Gay– Lussac reacted pairs of gases at the same temperature and pressur ...
... ________________________________________________________________________ During the time that Dalton’s mass scale was just being introduced, the French chemist ______________________________ began to study how gases reacted. When Gay– Lussac reacted pairs of gases at the same temperature and pressur ...
a rapid uplc™ - ms/ms method for determining specific
... amino acids (and their corresponding internal standards) at a concentration of 10µg/mL via syringe pump (10µL/min.) into a 50:50 mixture of mobile phases A and B flowing at 0.6mL/min. Under these conditions, the mass spectrometer was optimized to provide the greatest abundance of the leucine butyl e ...
... amino acids (and their corresponding internal standards) at a concentration of 10µg/mL via syringe pump (10µL/min.) into a 50:50 mixture of mobile phases A and B flowing at 0.6mL/min. Under these conditions, the mass spectrometer was optimized to provide the greatest abundance of the leucine butyl e ...
AP Chemistry
... Percent Yield of CO2 Gas Lab (Wear Goggles) 19. Completely react NaHCO3(s) with 6 M HCl, according to the net ionic equation HCO3- + H+ H2O + CO2(g), measure the volume of CO2 and compare this to the theoretical yield. Mass 0.6 g NaHCO3 and record its mass (m) to the nearest 0.001 g. Add the NaHCO ...
... Percent Yield of CO2 Gas Lab (Wear Goggles) 19. Completely react NaHCO3(s) with 6 M HCl, according to the net ionic equation HCO3- + H+ H2O + CO2(g), measure the volume of CO2 and compare this to the theoretical yield. Mass 0.6 g NaHCO3 and record its mass (m) to the nearest 0.001 g. Add the NaHCO ...
mole concept a
... 12 gram of the carbon-12 isotope. This definition gives us a method by which we can find out the amount of a substance (in moles) if we know the number of elementary entities present in it or vice versa. Now the question arises how many atoms are there in exactly 12 g of carbon-12. This number is de ...
... 12 gram of the carbon-12 isotope. This definition gives us a method by which we can find out the amount of a substance (in moles) if we know the number of elementary entities present in it or vice versa. Now the question arises how many atoms are there in exactly 12 g of carbon-12. This number is de ...
Atomic Mass: The atomic mass of an element is the mass average of
... x Molar mass of (Cl 2 ) mass of (Cl 2 ) = Molar mass of H 2 1(Cl 2 ) 4.770g of H 2 1( H 2 ) x 71.0g / mole ...
... x Molar mass of (Cl 2 ) mass of (Cl 2 ) = Molar mass of H 2 1(Cl 2 ) 4.770g of H 2 1( H 2 ) x 71.0g / mole ...
JSUNIL TUTORIAL , SAMASTIPUR, BIHAR
... Here, 12 gm of carbon combine with 16g and 32gm of Oxygen to form Carbon monoxide and Carbon dioxide respectively. The ratio of oxygen combining with 12 gm of Carbon is 16: 32 or, 1:2 which is in a simple ratio Dalton’s explanation for the law of conservation of mass and the law of definite proporti ...
... Here, 12 gm of carbon combine with 16g and 32gm of Oxygen to form Carbon monoxide and Carbon dioxide respectively. The ratio of oxygen combining with 12 gm of Carbon is 16: 32 or, 1:2 which is in a simple ratio Dalton’s explanation for the law of conservation of mass and the law of definite proporti ...
Metabolomics Research Core
... In partnership with the North Carolina Translational & Clinical Sciences (NCTraCS) Institute, RTI International offers opportunities to collaborate with our metabolomics researchers to discover biomarkers, reveal mechanistic insights, synthesize metabolite standards, determine characteristics of exp ...
... In partnership with the North Carolina Translational & Clinical Sciences (NCTraCS) Institute, RTI International offers opportunities to collaborate with our metabolomics researchers to discover biomarkers, reveal mechanistic insights, synthesize metabolite standards, determine characteristics of exp ...
AP Chemistry
... oxygen, in which a hydrocarbon or related compound reacts with O2 to form CO2 and H2O; (4) electron exchange reactions in which reactants exchange electrons; (5) ion exchange reactions, in which ions exchange "partners"; and (6) proton exchange reactions, in which atoms exchange H+. The last three r ...
... oxygen, in which a hydrocarbon or related compound reacts with O2 to form CO2 and H2O; (4) electron exchange reactions in which reactants exchange electrons; (5) ion exchange reactions, in which ions exchange "partners"; and (6) proton exchange reactions, in which atoms exchange H+. The last three r ...
PC235: 2008 Lecture 5 Quantitation
... •Quantitation is normally relative (between samples), but can also be absolute. •Software for analysis of this type of data is still being developed. ...
... •Quantitation is normally relative (between samples), but can also be absolute. •Software for analysis of this type of data is still being developed. ...
Mass spectrometry
Mass spectrometry (MS) is an analytical chemistry technique that helps identify the amount and type of chemicals present in a sample by measuring the mass-to-charge ratio and abundance of gas-phase ions.A mass spectrum (plural spectra) is a plot of the ion signal as a function of the mass-to-charge ratio. The spectra are used to determine the elemental or isotopic signature of a sample, the masses of particles and of molecules, and to elucidate the chemical structures of molecules, such as peptides and other chemical compounds. Mass spectrometry works by ionizing chemical compounds to generate charged molecules or molecule fragments and measuring their mass-to-charge ratios.In a typical MS procedure, a sample, which may be solid, liquid, or gas, is ionized, for example by bombarding it with electrons. This may cause some of the sample's molecules to break into charged fragments. These ions are then separated according to their mass-to-charge ratio, typically by accelerating them and subjecting them to an electric or magnetic field: ions of the same mass-to-charge ratio will undergo the same amount of deflection. The ions are detected by a mechanism capable of detecting charged particles, such as an electron multiplier. Results are displayed as spectra of the relative abundance of detected ions as a function of the mass-to-charge ratio. The atoms or molecules in the sample can be identified by correlating known masses to the identified masses or through a characteristic fragmentation pattern.