The Impact of Ligand Design on the Coordination Chemistry and
... Second, for carbonylrhodium(I) complexes, (NNN)Rh(CO), substitution at the para-aryl positions predictably modulates the electronic properties and chemical reactivity. Oxidative addition reactions of the (NNN)Rh(CO) with iodoalkanes proceed about three orders of magnitude faster than those reported ...
... Second, for carbonylrhodium(I) complexes, (NNN)Rh(CO), substitution at the para-aryl positions predictably modulates the electronic properties and chemical reactivity. Oxidative addition reactions of the (NNN)Rh(CO) with iodoalkanes proceed about three orders of magnitude faster than those reported ...
Sam P. de Visser,* Jan-Uwe Rohde,* Yong
... hydroxylation, halogenation, and other reactions involving C H bond activation for a variety of purposes, including biosynthetic functions, DNA repair, and cellular oxygen sensing. Many of these enzymes, including several ˛-ketoglutarate- (˛KG) and pterin-dependent oxygenases for which such a high-v ...
... hydroxylation, halogenation, and other reactions involving C H bond activation for a variety of purposes, including biosynthetic functions, DNA repair, and cellular oxygen sensing. Many of these enzymes, including several ˛-ketoglutarate- (˛KG) and pterin-dependent oxygenases for which such a high-v ...
The kinetics of the conversion of DHA to MGO
... MGO gain in mānuka honey during the exponential growth period showed similar behaviour but in mānuka honey the rate of loss of DHA was much higher and increased much more with increased temperature than in the clover honey. This indicates that the conversion of DHA to MGO in mānuka honey is less eff ...
... MGO gain in mānuka honey during the exponential growth period showed similar behaviour but in mānuka honey the rate of loss of DHA was much higher and increased much more with increased temperature than in the clover honey. This indicates that the conversion of DHA to MGO in mānuka honey is less eff ...
Thiele et al.: `Genome-scale reconstruction of E. coli`s transcriptional
... Dus_gen = DusA – C. These proteins are responsible for dihydrouridine formation in E.coli’s tRNA [15]. Based on Blast results of the sequence, the dus genes have homology to genes that carry out NADPH dependent reactions. Furthermore, the reaction was reported to be NADH or NADPH dependent in yeast ...
... Dus_gen = DusA – C. These proteins are responsible for dihydrouridine formation in E.coli’s tRNA [15]. Based on Blast results of the sequence, the dus genes have homology to genes that carry out NADPH dependent reactions. Furthermore, the reaction was reported to be NADH or NADPH dependent in yeast ...
Short-term regulation of the mammalian pyruvate dehydrogenase
... For a long time it has been suggested that mammalian PDHC loses TDP almost completely during isolation, because the measured PDHC activity was usually very low in the absence of added TDP (Walsh et al., 1976; Sumegi & Alkonyi, 1983). The measurements were based on the initial rate of the reaction ca ...
... For a long time it has been suggested that mammalian PDHC loses TDP almost completely during isolation, because the measured PDHC activity was usually very low in the absence of added TDP (Walsh et al., 1976; Sumegi & Alkonyi, 1983). The measurements were based on the initial rate of the reaction ca ...
c00kieee - Ritter Illustration
... is used for the in situ leaching of uranium ore except that the initial treatment is performed on bores with a slightly acidic and high oxygenated aqueous solution that is raised to the surface and allowed to go through an extraction process to remove the uranium. No matter which mining process is u ...
... is used for the in situ leaching of uranium ore except that the initial treatment is performed on bores with a slightly acidic and high oxygenated aqueous solution that is raised to the surface and allowed to go through an extraction process to remove the uranium. No matter which mining process is u ...
Biosynthesis of Glucosyl Glycerol, a Compatible Solute, Using
... the intermolecular transglycosylation activity of ASase has gained increasing attention because it uses a relatively cheap substrate, sucrose, as well as its broad range of acceptor specificity [7, 8]. ASase can employ not only various glycones such as salicin [9] and arbutin [10] but also numerous ...
... the intermolecular transglycosylation activity of ASase has gained increasing attention because it uses a relatively cheap substrate, sucrose, as well as its broad range of acceptor specificity [7, 8]. ASase can employ not only various glycones such as salicin [9] and arbutin [10] but also numerous ...
Peter Ertl - American Chemical Society
... methods. To estimate this number, we used a very simple computational experiment, namely to determine how many molecules of the general formula R1-X-R2 may be constructed. R1 and R2 in this formula are substituents, and X is a scaffold with two attachment points. We analyzed our database of 3 millio ...
... methods. To estimate this number, we used a very simple computational experiment, namely to determine how many molecules of the general formula R1-X-R2 may be constructed. R1 and R2 in this formula are substituents, and X is a scaffold with two attachment points. We analyzed our database of 3 millio ...
Aldehydes and Ketones
... because individuals have characteristic odor prints that are as unique as their fingerprints or DNA. Forensic scientists are now making the next step in developing this technology for more accurate detection of people associated with a crime scene. The first step is to identify the components of the ...
... because individuals have characteristic odor prints that are as unique as their fingerprints or DNA. Forensic scientists are now making the next step in developing this technology for more accurate detection of people associated with a crime scene. The first step is to identify the components of the ...
Mole Concept - Shailendra Kumar Chemistry
... (c) 200 ml of 3.0 M NaCl is added to 300 ml of 4.0 M NaCl. (d) 200 ml of 2.0 M BaCl2 is added to 400 ml of 3.0 M BaCl2 and 400 ml of water. (e) 300 ml of 3.0 M NaCl is added to 200 ml of 4.0 M BaCl2. (f) 400 ml of 2.0 M HCl is added to 150 ml of 4.0 M NaOH. (g) 100 ml of 2.0 M HCl and 200 ml of 1.5 ...
... (c) 200 ml of 3.0 M NaCl is added to 300 ml of 4.0 M NaCl. (d) 200 ml of 2.0 M BaCl2 is added to 400 ml of 3.0 M BaCl2 and 400 ml of water. (e) 300 ml of 3.0 M NaCl is added to 200 ml of 4.0 M BaCl2. (f) 400 ml of 2.0 M HCl is added to 150 ml of 4.0 M NaOH. (g) 100 ml of 2.0 M HCl and 200 ml of 1.5 ...
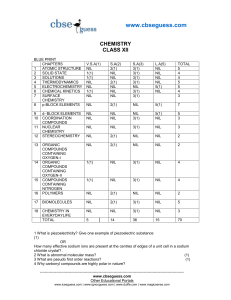
guess paper class xii
... 7 Write balanced equations for the following reactions; (i) SnO is treated with dilute HNO3 (ii) Aqueous sodium hydroxide is added dropwise to a solution of gallium chloride in water. A precipitate is formed initially which dissolves on further addition of NaOH solution. ...
... 7 Write balanced equations for the following reactions; (i) SnO is treated with dilute HNO3 (ii) Aqueous sodium hydroxide is added dropwise to a solution of gallium chloride in water. A precipitate is formed initially which dissolves on further addition of NaOH solution. ...
The polydentate ligands include polyaminopolycarbonic acids, such
... Chapters are helpfully signposted throughout, informing the reader how topics are related, which is especially important in such a multidisciplinary subject. Topics are also presented clearly and with a logical progression culminating in the main points, questions and reading sections at the beginni ...
... Chapters are helpfully signposted throughout, informing the reader how topics are related, which is especially important in such a multidisciplinary subject. Topics are also presented clearly and with a logical progression culminating in the main points, questions and reading sections at the beginni ...
Theoretical Investigation of the Water
... The Water-Gas Shift (WGS: CO+H2O↔CO2+H2) reaction is a key step in hydrogen fuel processing for mobile fuel cell applications. Since the reaction is equilibrium-limited and exothermic, high conversions are favored by low temperatures. However, conventional low-temperature shift catalysts are not act ...
... The Water-Gas Shift (WGS: CO+H2O↔CO2+H2) reaction is a key step in hydrogen fuel processing for mobile fuel cell applications. Since the reaction is equilibrium-limited and exothermic, high conversions are favored by low temperatures. However, conventional low-temperature shift catalysts are not act ...
102MSJc14 - Louisiana Tech University
... 4. Obtain equilibrium constant expressions for related reactions from the Expression for one or more known reactions (Section 14.2). 5. Calculate Kp from K, or Kc from Kp for the same equilibrium (Section 14.2). 6. Calculate a value of K, for an equilibrium system, given information about initial co ...
... 4. Obtain equilibrium constant expressions for related reactions from the Expression for one or more known reactions (Section 14.2). 5. Calculate Kp from K, or Kc from Kp for the same equilibrium (Section 14.2). 6. Calculate a value of K, for an equilibrium system, given information about initial co ...
Problem 1-2
... The problems to be solved in the 1st round are sent to all high schools. To solve the problems the students may use all resources available, e.g. textbooks etc. All those students who solve about 70% of the tasks correctly will receive the problems of the 2nd round, which are to be solved in the sam ...
... The problems to be solved in the 1st round are sent to all high schools. To solve the problems the students may use all resources available, e.g. textbooks etc. All those students who solve about 70% of the tasks correctly will receive the problems of the 2nd round, which are to be solved in the sam ...
coordination compounds
... Chapters are helpfully signposted throughout, informing the reader how topics are related, which is especially important in such a multidisciplinary subject. Topics are also presented clearly and with a logical progression culminating in the main points, questions and reading sections at the beginni ...
... Chapters are helpfully signposted throughout, informing the reader how topics are related, which is especially important in such a multidisciplinary subject. Topics are also presented clearly and with a logical progression culminating in the main points, questions and reading sections at the beginni ...
An Introduction to Enzyme Science
... a more appropriate appellation, ‘‘interaction spectroscopists’’ – focusing on the spectrum of interactions of proteins and enzymes with other proteins, nucleic acids, membranes, and low molecular-weight metabolites, most often in terms of location, specificity, affinity, and catalysis. And because e ...
... a more appropriate appellation, ‘‘interaction spectroscopists’’ – focusing on the spectrum of interactions of proteins and enzymes with other proteins, nucleic acids, membranes, and low molecular-weight metabolites, most often in terms of location, specificity, affinity, and catalysis. And because e ...
On-surface derivatisation of aromatic molecules
... seen that even with a large aromatic core, 1 can still be removed unless in a densely packed configuration which allows intermolecular forces to stabilise the film. While the decrease in packing density observed by Raman spectroscopy indicates an increase in the chain length of the end-groups of 1, ...
... seen that even with a large aromatic core, 1 can still be removed unless in a densely packed configuration which allows intermolecular forces to stabilise the film. While the decrease in packing density observed by Raman spectroscopy indicates an increase in the chain length of the end-groups of 1, ...
chem 102 class notes - Louisiana Tech University
... 4. Obtain equilibrium constant expressions for related reactions from the Expression for one or more known reactions (Section 14.2). 5. Calculate Kp from K, or Kc from Kp for the same equilibrium (Section 14.2). 6. Calculate a value of K, for an equilibrium system, given information about initial co ...
... 4. Obtain equilibrium constant expressions for related reactions from the Expression for one or more known reactions (Section 14.2). 5. Calculate Kp from K, or Kc from Kp for the same equilibrium (Section 14.2). 6. Calculate a value of K, for an equilibrium system, given information about initial co ...
Chemical Reactions and Stoichiometry
... HE AMOUNT OF PRODUCT FORMED IN A CHEMICAL REACTION is related to the amount of reactant that is consumed. This concept makes sense intuitively, but how do we describe and understand this relationship more ...
... HE AMOUNT OF PRODUCT FORMED IN A CHEMICAL REACTION is related to the amount of reactant that is consumed. This concept makes sense intuitively, but how do we describe and understand this relationship more ...
Mechanism of Succinyl
... Citrate Synthase is an enzyme that catalyzes the first step in the citric acid cycle. Oxaloacetate and acetyl-CoA bind to Citrate Synthase, which then catalyzes the reaction which joins the two compounds together In eukaryotes, Citrate Synthase is a dimer, meaning that it is a protein which is compo ...
... Citrate Synthase is an enzyme that catalyzes the first step in the citric acid cycle. Oxaloacetate and acetyl-CoA bind to Citrate Synthase, which then catalyzes the reaction which joins the two compounds together In eukaryotes, Citrate Synthase is a dimer, meaning that it is a protein which is compo ...
The Stereochemistry of Enzymatic Transamination“
... The observation of a small kinetic isotope effect in the apoglutamate-oxaloacetate transaminase catalyzed transamination of D2-pyridoxamine is expected. The magnitude of the true isotope effect for CH bond breaking is certain to be reduced by the reversible formation of the anion of the cofactor sub ...
... The observation of a small kinetic isotope effect in the apoglutamate-oxaloacetate transaminase catalyzed transamination of D2-pyridoxamine is expected. The magnitude of the true isotope effect for CH bond breaking is certain to be reduced by the reversible formation of the anion of the cofactor sub ...
step by step Stoichiometry
... Or 80.3 divided by 55.847, multiplied by 3, divided by 2, multiplied by 28.01015 ...
... Or 80.3 divided by 55.847, multiplied by 3, divided by 2, multiplied by 28.01015 ...