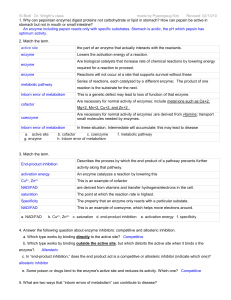
Inborn Errors of Metabolism
... “A genetic disorder that involves an enzyme deficiency. The enzyme block leads to the accumulation of a toxic substrate and/or the deficient synthesis of a product needed for normal body function” (Figure 20.1 pg. 286). Enzymes break down the fats, proteins & carbohydrates from the food we eat. ...
... “A genetic disorder that involves an enzyme deficiency. The enzyme block leads to the accumulation of a toxic substrate and/or the deficient synthesis of a product needed for normal body function” (Figure 20.1 pg. 286). Enzymes break down the fats, proteins & carbohydrates from the food we eat. ...
Reagent Grade Enzymes for Nitrate Determination
... often with attached “cofactors” that facilitate catalytic function. Some B vitamins are enzyme cofactors; others are metal complexes such as hemoglobin or chlorophyll. Enzymes are named and grouped by catalytic function. There may be hundreds of variations of an enzyme, differing by minor changes in ...
... often with attached “cofactors” that facilitate catalytic function. Some B vitamins are enzyme cofactors; others are metal complexes such as hemoglobin or chlorophyll. Enzymes are named and grouped by catalytic function. There may be hundreds of variations of an enzyme, differing by minor changes in ...
ENZYME
... In general, alkaline phosphatase is a dimer containing nearly identical subunits which each have two molecules of zinc and one molecule of magnesium ion. One molecule of zinc is tightly bound, giving the structure stability and the other is loosely bound which provides for the catalytic activity ...
... In general, alkaline phosphatase is a dimer containing nearly identical subunits which each have two molecules of zinc and one molecule of magnesium ion. One molecule of zinc is tightly bound, giving the structure stability and the other is loosely bound which provides for the catalytic activity ...
Enzymes what are they - Laurel County Schools
... The active site involves a small number of key residues that actually bind thesubstrates The rest of the protein structure is needed to maintain these residues in position ...
... The active site involves a small number of key residues that actually bind thesubstrates The rest of the protein structure is needed to maintain these residues in position ...
2. Enzyme activity - Lectures For UG-5
... When the enzyme initially introduced to the reactants and the excess substrate is steadily combining with available enzyme, the reaction rate rises. After the enzyme is saturated, the rates of product formation, release of enzyme ,and recombination with more substrate proceed linearly. 6to8 minutes ...
... When the enzyme initially introduced to the reactants and the excess substrate is steadily combining with available enzyme, the reaction rate rises. After the enzyme is saturated, the rates of product formation, release of enzyme ,and recombination with more substrate proceed linearly. 6to8 minutes ...
Enzymes POGIL 2014
... three different enzyme mutants. For each, one aspect of the active site's structure is slightly changed. The mutant enzymes are mixed with RNA in solution. The reaction rates are shown underneath each image. ...
... three different enzyme mutants. For each, one aspect of the active site's structure is slightly changed. The mutant enzymes are mixed with RNA in solution. The reaction rates are shown underneath each image. ...
Enzyme Activity
... structure to the actual substrate and so will bind temporarily with the active site. The rate of reaction will be closer to the maximum when there is more ‘real’ substrate, (e.g. arabinose competes with glucose for the active sites on glucose oxidase enzyme). Non-competitive reversible inhibitors: t ...
... structure to the actual substrate and so will bind temporarily with the active site. The rate of reaction will be closer to the maximum when there is more ‘real’ substrate, (e.g. arabinose competes with glucose for the active sites on glucose oxidase enzyme). Non-competitive reversible inhibitors: t ...
Anaerobic Respiration
... lactate production increases. The increase in Lactate also causes an interference between areas of muscle fibres which inhibit muscular contraction. The lactate threshold (LT) is the value of exercise ...
... lactate production increases. The increase in Lactate also causes an interference between areas of muscle fibres which inhibit muscular contraction. The lactate threshold (LT) is the value of exercise ...
Enzymes: Organic Catalysts What are Enzymes?
... • Enzymes can act rapidly, as in the case of carbonic anhydrase (enzymes typically end in the -ase suffix), which causes the chemicals to react 107 times faster than without the enzyme present. Carbonic anhydrase speeds up the transfer of carbon dioxide from cells to the blood. • There are over 2000 ...
... • Enzymes can act rapidly, as in the case of carbonic anhydrase (enzymes typically end in the -ase suffix), which causes the chemicals to react 107 times faster than without the enzyme present. Carbonic anhydrase speeds up the transfer of carbon dioxide from cells to the blood. • There are over 2000 ...
Protease - etcsciencestudents
... enzyme isn’t present, the lactose cannot be converted into sugars such as glucose. A lack of this enzyme causes lactose intolerance. The lactose can’t be broken down and acts as a great food source ...
... enzyme isn’t present, the lactose cannot be converted into sugars such as glucose. A lack of this enzyme causes lactose intolerance. The lactose can’t be broken down and acts as a great food source ...
Enzymes
... close together. This allows them 10 react more quickly than they would otherwise, after which they are released. Oneway to remove an enzyme from a chemicalsystem is to inhibit if; How itihibilion takes place is the subiectof the third diagram of the plate; the appropriate structures as you. complete ...
... close together. This allows them 10 react more quickly than they would otherwise, after which they are released. Oneway to remove an enzyme from a chemicalsystem is to inhibit if; How itihibilion takes place is the subiectof the third diagram of the plate; the appropriate structures as you. complete ...
Amino Acids, Proteins, and Enzymes
... • are most active at an optimum temperature (usually 37°C in humans). • show little activity at low temperatures. • lose activity at high temperatures as denaturation occurs. ...
... • are most active at an optimum temperature (usually 37°C in humans). • show little activity at low temperatures. • lose activity at high temperatures as denaturation occurs. ...
N .B. Aschengreen PROTEOLYTIC ENZYMES USED FOR
... This title, which I have been given and which you have Seen in the Programme for today, is a little difficult for me because we here at NOVO do not know much about restoring; but as we do know something about enzymes I shall stick to this subject and try to emphasize factors, that I would expect of ...
... This title, which I have been given and which you have Seen in the Programme for today, is a little difficult for me because we here at NOVO do not know much about restoring; but as we do know something about enzymes I shall stick to this subject and try to emphasize factors, that I would expect of ...
Basic Enzyme Structure and Function
... Fill in the gaps and annotate the diagram to explain what is happening. Enzymes act as biological ______________________. They are __________________ proteins that have a specific shape (tertiary ___________________) within which there is a functional portion known as the ____________ ___________. E ...
... Fill in the gaps and annotate the diagram to explain what is happening. Enzymes act as biological ______________________. They are __________________ proteins that have a specific shape (tertiary ___________________) within which there is a functional portion known as the ____________ ___________. E ...
... as well as what changes take place when a patient begins drinking again. The goal of these studies is to determine which alcohol-induced effects on the brain are permanent and which ones can be reversed with abstinence. PET imaging is allowing researchers to visualize, in the living brain, the damag ...
Enzymes
... Are necessary for normal activity of enzymes ;are derived from vitamins; transport small molecules needed by enzymes. ...
... Are necessary for normal activity of enzymes ;are derived from vitamins; transport small molecules needed by enzymes. ...
Enzymes
... What are Enzymes? Enzymes are protein compounds that assist chemical reactions by increasing the rate at which they occur, and lowering the amount of energy used. For example, the food that you eat is broken down by digestive enzymes into tiny pieces that are small enough to travel through your bloo ...
... What are Enzymes? Enzymes are protein compounds that assist chemical reactions by increasing the rate at which they occur, and lowering the amount of energy used. For example, the food that you eat is broken down by digestive enzymes into tiny pieces that are small enough to travel through your bloo ...
Effect aliphatic alcohols on catalytic activity of bovine pancreatic α
... organic media provide the possibility of conducting industrially important synthetic reactions that do not occur in aqueous media (peptide synthesis and esterification). The enzymatic catalysis in organic solvents is competitive and cost-saving technology for producing substances with a high optical ...
... organic media provide the possibility of conducting industrially important synthetic reactions that do not occur in aqueous media (peptide synthesis and esterification). The enzymatic catalysis in organic solvents is competitive and cost-saving technology for producing substances with a high optical ...
Unit-III Enzymes
... 1. Introduction and Properties of enzymes 2. Nomenclature and Classification 3. Mechanism of enzyme-catalyzed reactions 4. Kinetics of enzyme-catalyzed reactions 5. Inhibition of enzymes 6. Regulation of enzymes 7. Clinical applications of enzymes ...
... 1. Introduction and Properties of enzymes 2. Nomenclature and Classification 3. Mechanism of enzyme-catalyzed reactions 4. Kinetics of enzyme-catalyzed reactions 5. Inhibition of enzymes 6. Regulation of enzymes 7. Clinical applications of enzymes ...
ANSWERS TO REVIEW QUESTIONS – CHAPTER 03
... 100 000 g mole–1 = 100 kDaltons) that consist of an agglomeration of a small number of lower molecular weight subunits. These subunits are held together by intermolecular forces that are broken as the temperature is increased. The rupture of the intermolecular bonds between the subunits results in a ...
... 100 000 g mole–1 = 100 kDaltons) that consist of an agglomeration of a small number of lower molecular weight subunits. These subunits are held together by intermolecular forces that are broken as the temperature is increased. The rupture of the intermolecular bonds between the subunits results in a ...
Enzymes
... type of key that will open it, each enzyme has a particular substrate that it can act upon based on – Size, shape, and specificity of the substrate ...
... type of key that will open it, each enzyme has a particular substrate that it can act upon based on – Size, shape, and specificity of the substrate ...
Enzymes
... An enzyme is not only substrate specific, but it can only catalyze a reaction in ONE direction. So, it either builds up, or breaks down …but not both ...
... An enzyme is not only substrate specific, but it can only catalyze a reaction in ONE direction. So, it either builds up, or breaks down …but not both ...
Enzymes - Guided Notes - Flip-Flop
... How many reactants are there? ______ How many products are there? _______ ----------------------------------------------------------------- What is/are the reactant(s)? What is/are the product(s)? --------------------------------------------------- What is/are the reactant(s)? What is/are the prod ...
... How many reactants are there? ______ How many products are there? _______ ----------------------------------------------------------------- What is/are the reactant(s)? What is/are the product(s)? --------------------------------------------------- What is/are the reactant(s)? What is/are the prod ...
Alcohol dehydrogenase

Alcohol dehydrogenases (ADH) (EC 1.1.1.1) are a group of dehydrogenase enzymes that occur in many organisms and facilitate the interconversion between alcohols and aldehydes or ketones with the reduction of nicotinamide adenine dinucleotide (NAD+ to NADH). In humans and many other animals, they serve to break down alcohols that otherwise are toxic, and they also participate in generation of useful aldehyde, ketone, or alcohol groups during biosynthesis of various metabolites. In yeast, plants, and many bacteria, some alcohol dehydrogenases catalyze the opposite reaction as part of fermentation to ensure a constant supply of NAD+.