Chemistry Merit Badge
... D) Discuss the safe storage of chemicals. How does the safe storage of chemicals apply to your home, your school, your community, and the environment? 2) Do EACH of the following activities: A) Predict what would happen if you placed and iron nail in a copper sulfate solution. Then, put an iron nail ...
... D) Discuss the safe storage of chemicals. How does the safe storage of chemicals apply to your home, your school, your community, and the environment? 2) Do EACH of the following activities: A) Predict what would happen if you placed and iron nail in a copper sulfate solution. Then, put an iron nail ...
Chapter 19 Chemical Thermodynamics
... from one form to another or transferred from a system to the surroundings or vice versa. Chemical Thermodynamics © 2009, Prentice-Hall, Inc. ...
... from one form to another or transferred from a system to the surroundings or vice versa. Chemical Thermodynamics © 2009, Prentice-Hall, Inc. ...
Lab 3. Chemical Reactions
... Each element or compound has unique properties that make it distinguishable from other elements or compounds. Physical properties, such as color, luster, softness, electrical conductivity, the temperature at which it melts or boils, crystal structure etc. are characteristics that are visible or meas ...
... Each element or compound has unique properties that make it distinguishable from other elements or compounds. Physical properties, such as color, luster, softness, electrical conductivity, the temperature at which it melts or boils, crystal structure etc. are characteristics that are visible or meas ...
Notes
... or identity of a substance • Physical change - produces a recognizable difference in the appearance of a substance without causing any change in its composition or identity - conversion from one physical state to another - melting an ice cube ...
... or identity of a substance • Physical change - produces a recognizable difference in the appearance of a substance without causing any change in its composition or identity - conversion from one physical state to another - melting an ice cube ...
Chapter 4 Nomenclature and Chemical Equations
... It is a convention to write the reactants on the left and the products on the right. The letters enclosed in the parenthesis tell us the states of the substances: s denotes a solid, l denotes a liquid, g denotes a gas and aq denotes an aqueous solution, i.e. a homogeneous mixture in water. Theref ...
... It is a convention to write the reactants on the left and the products on the right. The letters enclosed in the parenthesis tell us the states of the substances: s denotes a solid, l denotes a liquid, g denotes a gas and aq denotes an aqueous solution, i.e. a homogeneous mixture in water. Theref ...
Chemical Thermodynamics : Georg Duesberg
... 2) They undergo elastic collisions 3) They are large in number and are randomly distributed 4) They can be treated as points of mass (diameter<< mean free path) Chemical Thermodynamics : Georg Duesberg ...
... 2) They undergo elastic collisions 3) They are large in number and are randomly distributed 4) They can be treated as points of mass (diameter<< mean free path) Chemical Thermodynamics : Georg Duesberg ...
Diversity-oriented synthesis - David Spring
... point was demonstrated by the results from the computational analysis of various databases. It was found that the number of chiral centers, on average, from combinatorial chemistry, natural products, and drugs was 0.4, 6.2, and 3.3 per molecule, respectively.17 Although the compound archives of phar ...
... point was demonstrated by the results from the computational analysis of various databases. It was found that the number of chiral centers, on average, from combinatorial chemistry, natural products, and drugs was 0.4, 6.2, and 3.3 per molecule, respectively.17 Although the compound archives of phar ...
Document
... I can write chemical reactions by interpreting word equations I can classify reaction types (synthesis, decomposition, single replacement, double replacement, combustion) I can predict the products of chemical reactions in writing complete chemical equations (synthesis, decomposition, single replace ...
... I can write chemical reactions by interpreting word equations I can classify reaction types (synthesis, decomposition, single replacement, double replacement, combustion) I can predict the products of chemical reactions in writing complete chemical equations (synthesis, decomposition, single replace ...
Synthesis Reaction
... I can write chemical reactions by interpreting word equations I can classify reaction types (synthesis, decomposition, single replacement, double replacement, combustion) I can predict the products of chemical reactions in writing complete chemical equations (synthesis, decomposition, single replace ...
... I can write chemical reactions by interpreting word equations I can classify reaction types (synthesis, decomposition, single replacement, double replacement, combustion) I can predict the products of chemical reactions in writing complete chemical equations (synthesis, decomposition, single replace ...
Chemistry: Matter and Change
... that when different compounds are formed by a combination of the same elements, different masses of one element combine with the same relative mass of the other element in whole number ratios. – H2O2 and H2O – Copper(I) chloride and copper(II) chloride ...
... that when different compounds are formed by a combination of the same elements, different masses of one element combine with the same relative mass of the other element in whole number ratios. – H2O2 and H2O – Copper(I) chloride and copper(II) chloride ...
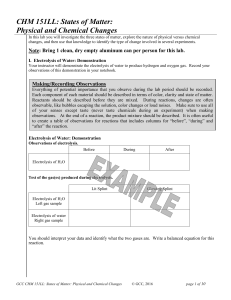
CHM 151LL: States of Matter: Physical and Chemical Changes
... In a pure substance every particle in the sample would look the same. In a drawing of a pure substance, every particle in the material looks the same. In a drawing of a mixture, different components of the mixture will look different. A mixture contains different atoms, molecules, and/or compounds a ...
... In a pure substance every particle in the sample would look the same. In a drawing of a pure substance, every particle in the material looks the same. In a drawing of a mixture, different components of the mixture will look different. A mixture contains different atoms, molecules, and/or compounds a ...
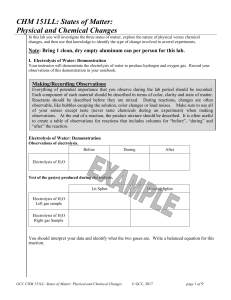
CHM 151LL: States of Matter: Physical and Chemical Changes
... In a pure substance every particle in the sample would look the same. In a drawing of a pure substance, every particle in the material looks the same. In a drawing of a mixture, different components of the mixture will look different. A mixture contains different atoms, molecules, and/or compounds a ...
... In a pure substance every particle in the sample would look the same. In a drawing of a pure substance, every particle in the material looks the same. In a drawing of a mixture, different components of the mixture will look different. A mixture contains different atoms, molecules, and/or compounds a ...
2011-2012 ACAD REVIEW SHEET Chapter 2
... What is the relationship between the kinetic energy of molecules and their physical state? (ANS: Gases have the greatest amount of kinetic energy; solids have the least.) ...
... What is the relationship between the kinetic energy of molecules and their physical state? (ANS: Gases have the greatest amount of kinetic energy; solids have the least.) ...
Chemistry Mid-Term Review Guide
... that when different compounds are formed by a combination of the same elements, different masses of one element combine with the same relative mass of the other element in whole number ratios. – H2O2 and H2O – Copper(I) chloride and copper(II) chloride ...
... that when different compounds are formed by a combination of the same elements, different masses of one element combine with the same relative mass of the other element in whole number ratios. – H2O2 and H2O – Copper(I) chloride and copper(II) chloride ...
Chemistry: Matter and Change
... that when different compounds are formed by a combination of the same elements, different masses of one element combine with the same relative mass of the other element in whole number ratios. – H2O2 and H2O – Copper(I) chloride and copper(II) chloride ...
... that when different compounds are formed by a combination of the same elements, different masses of one element combine with the same relative mass of the other element in whole number ratios. – H2O2 and H2O – Copper(I) chloride and copper(II) chloride ...
Chapter 19 Chemical Thermodynamics
... • What is enthalpy, H, and how is Hess’s Law used to calculate enthalpy changes in chemical reactions? Chemical Thermodynamics ...
... • What is enthalpy, H, and how is Hess’s Law used to calculate enthalpy changes in chemical reactions? Chemical Thermodynamics ...
Instruments for Radiation Detection and Measurement
... One should be familiar with the color and state of a radiopharmaceutical A true solution should not contain any particulate matter Any deviation from the original color and clarity should be viewed with concern because it may reflect changes in the radiopharmaceutical that would alter its biologic b ...
... One should be familiar with the color and state of a radiopharmaceutical A true solution should not contain any particulate matter Any deviation from the original color and clarity should be viewed with concern because it may reflect changes in the radiopharmaceutical that would alter its biologic b ...
Theories in the Evolution of Chemical Equilibrium: Impli
... In the early years of the 18th century, Newton tried to find a theoretical explanation for why some substances reacted with others. In the thirty-first Query of his book Optics he considered that in chemistry there would be forces similar to the gravitational ones. Within this theoretical basis, New ...
... In the early years of the 18th century, Newton tried to find a theoretical explanation for why some substances reacted with others. In the thirty-first Query of his book Optics he considered that in chemistry there would be forces similar to the gravitational ones. Within this theoretical basis, New ...
ppt
... – Translational: Movement of the entire molecule from one place to another. – Vibrational: Periodic motion of atoms within a molecule. – Rotational: Rotation of the molecule on about an axis or ...
... – Translational: Movement of the entire molecule from one place to another. – Vibrational: Periodic motion of atoms within a molecule. – Rotational: Rotation of the molecule on about an axis or ...
Chemistry: Matter and Change
... that when different compounds are formed by a combination of the same elements, different masses of one element combine with the same relative mass of the other element in whole number ratios. – H2O2 and H2O – Copper(I) chloride and copper(II) chloride ...
... that when different compounds are formed by a combination of the same elements, different masses of one element combine with the same relative mass of the other element in whole number ratios. – H2O2 and H2O – Copper(I) chloride and copper(II) chloride ...
Science Focus 9 Matter and Chemical Change Class Notes Topic 1
... distillation, and they described the properties of many different materials. They also thought they could change lead and copper into gold. They used special symbols to prevent others from finding out their secrets. The current view of matter began with Sir Francis Bacon, who stated that all science ...
... distillation, and they described the properties of many different materials. They also thought they could change lead and copper into gold. They used special symbols to prevent others from finding out their secrets. The current view of matter began with Sir Francis Bacon, who stated that all science ...
CHEMICAL ENGINEERING CHE
... Chemical Reaction Engineering Spring. 3(3-0) P:M: (CHE 311 and CHE 312 or concurrently and CHE 321 or concurrently) Design and analysis of homogeneous flow and batch reactors. Chemical kinetics and equilibria. Reaction rate expressions from mechanisms and experimental data. Mass and heat transfer in ...
... Chemical Reaction Engineering Spring. 3(3-0) P:M: (CHE 311 and CHE 312 or concurrently and CHE 321 or concurrently) Design and analysis of homogeneous flow and batch reactors. Chemical kinetics and equilibria. Reaction rate expressions from mechanisms and experimental data. Mass and heat transfer in ...
chem equation Pkt Student2
... 3) Write a balanced chemical equation by adding_____________________, NOT subscripts (this will require trial and error, the following guidelines may be helpful) a) balance the different types of atoms ________________ b) first, balance the atoms of elements that are combined and that appear only _ ...
... 3) Write a balanced chemical equation by adding_____________________, NOT subscripts (this will require trial and error, the following guidelines may be helpful) a) balance the different types of atoms ________________ b) first, balance the atoms of elements that are combined and that appear only _ ...
Chemical Equations
... Answer the questions 1-8, using this equation: 2 Na (s) + 2 H2O (l) Æ 2 NaOH (aq) + H2 1. What are the reactant(s)? _____________________ 2. What are the product(s)? _______________________ 3. How many molecules of H2 are present as a product? ___________ 4. How many moles of NaOH are present? _____ ...
... Answer the questions 1-8, using this equation: 2 Na (s) + 2 H2O (l) Æ 2 NaOH (aq) + H2 1. What are the reactant(s)? _____________________ 2. What are the product(s)? _______________________ 3. How many molecules of H2 are present as a product? ___________ 4. How many moles of NaOH are present? _____ ...
the properties and structure of matter
... • Define physical change and list several common physical changes. • Define chemical change and list several indications that a chemical change has taken place • Explain the gas, liquid and solid states in terms of particles • Distinguish between mixtures, elements and ...
... • Define physical change and list several common physical changes. • Define chemical change and list several indications that a chemical change has taken place • Explain the gas, liquid and solid states in terms of particles • Distinguish between mixtures, elements and ...
Al-Shifa pharmaceutical factory

The Al-Shifa (الشفاء, Arabic for ""healing"") pharmaceutical factory in Khartoum North, Sudan, was constructed between 1992 and 1996 with components imported from the United States, Sweden, Italy, Switzerland, Germany, India, and Thailand. It was officially opened on July 12, 1997.The industrial complex was composed of around four buildings. It was the largest pharmaceutical factory in Khartoum and employed over 300 workers, producing medicine both for human and veterinary use.The factory was destroyed in 1998 by a missile attack launched by the United States government, killing one employee and wounding eleven. Critics of the attack have estimated that up to tens of thousands of Sudanese civilians died throughout Sudan as the supply of necessary drugs was cut off. The U.S. government stated several reasons for its attack: The alleged use of the factory for the processing of VX nerve agent. For alleged ties between the owners of the plant and al-Qaeda.These justifications for the bombing were disputed by the owners of the plant, the Sudanese government, and other governments.