RLE-TR-059-047069 - DSpace@MIT - Massachusetts Institute of
... The type of frequency standard used is described in Vol. 11 of the M.I.T. Radiation Laboratory Series 1 6 and is shown schematically in Fig. 6. The frequency standard "pip" is generated by the beating of the frequencyswept signal generator of the spectroscope with the frequency standard signal. If t ...
... The type of frequency standard used is described in Vol. 11 of the M.I.T. Radiation Laboratory Series 1 6 and is shown schematically in Fig. 6. The frequency standard "pip" is generated by the beating of the frequencyswept signal generator of the spectroscope with the frequency standard signal. If t ...
Chapter_10
... One pulls at the end of the wrench (r = 1 m) with a force F = 500 N at an angle F1 = 80°; the other pulls at the middle of wrench with the same force and at an angle F2 = 90°. What is the net torque the two mechanics are applying to the screw? ...
... One pulls at the end of the wrench (r = 1 m) with a force F = 500 N at an angle F1 = 80°; the other pulls at the middle of wrench with the same force and at an angle F2 = 90°. What is the net torque the two mechanics are applying to the screw? ...
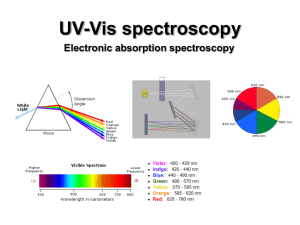
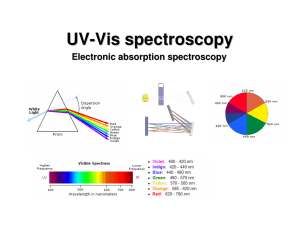
UV-Vis (electronic) spectroscopy
... • Spin-forbidden transitions – Transitions involving a change in the spin state of the molecule are forbidden – Strongly obeyed – Relaxed by effects that make spin a poor quantum number (heavy atoms) • Symmetry-forbidden transitions – Transitions between states of the same parity are forbidden – Pa ...
... • Spin-forbidden transitions – Transitions involving a change in the spin state of the molecule are forbidden – Strongly obeyed – Relaxed by effects that make spin a poor quantum number (heavy atoms) • Symmetry-forbidden transitions – Transitions between states of the same parity are forbidden – Pa ...
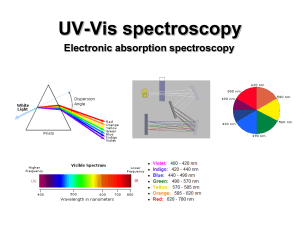
UV-Vis (electronic) spectroscopy
... • Spin-forbidden transitions – Transitions involving a change in the spin state of the molecule are forbidden – Strongly obeyed – Relaxed by effects that make spin a poor quantum number (heavy atoms) • Symmetry-forbidden transitions – Transitions between states of the same parity are forbidden – Pa ...
... • Spin-forbidden transitions – Transitions involving a change in the spin state of the molecule are forbidden – Strongly obeyed – Relaxed by effects that make spin a poor quantum number (heavy atoms) • Symmetry-forbidden transitions – Transitions between states of the same parity are forbidden – Pa ...
Homework Handout #3
... transitions. For a large number of nuclear spins, the lower and upper spin state will be nearly equally populated, since the nergy splitting is much smaller than the thermal energy (kT) at room temp. Since the intensity of the absorption is related to the difference in the population of the two stat ...
... transitions. For a large number of nuclear spins, the lower and upper spin state will be nearly equally populated, since the nergy splitting is much smaller than the thermal energy (kT) at room temp. Since the intensity of the absorption is related to the difference in the population of the two stat ...
Rotational spectroscopy

Rotational spectroscopy is concerned with the measurement of the energies of transitions between quantized rotational states of molecules in the gas phase. The spectra of polar molecules can be measured in absorption or emission by microwave spectroscopy or by far infrared spectroscopy. The rotational spectra of non-polar molecules cannot be observed by those methods, but can be observed and measured by Raman spectroscopy. Rotational spectroscopy is sometimes referred to as pure rotational spectroscopy to distinguish it from rotational-vibrational spectroscopy where changes in rotational energy occur together with changes in vibrational energy, and also from ro-vibronic spectroscopy (or just vibronic spectroscopy) where rotational, vibrational and electronic energy changes occur simultaneously.For rotational spectroscopy, molecules are classified according to symmetry into spherical top, linear and symmetric top; analytical expressions can be derived for the rotational energy terms of these molecules. Analytical expressions can be derived for the fourth category, asymmetric top, for rotational levels up to J=3, but higher energy levels need to be determined using numerical methods. The rotational energies are derived theoretically by considering the molecules to be rigid rotors and then applying extra terms to account for centrifugal distortion, fine structure, hyperfine structure and Coriolis coupling. Fitting the spectra to the theoretical expressions gives numerical values of the angular moments of inertia from which very precise values of molecular bond lengths and angles can be derived in favorable cases. In the presence of an electrostatic field there is Stark splitting which allows molecular electric dipole moments to be determined.An important application of rotational spectroscopy is in exploration of the chemical composition of the interstellar medium using radio telescopes.