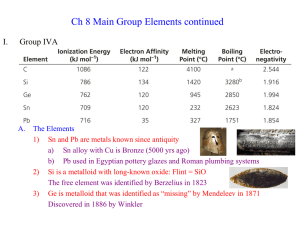
Pdf - Text of NPTEL IIT Video Lectures
... carbonyls on the surface, it has been supposed that there is a sphere on which the carbonyls are moving. The metal cluster is in fact embedded inside the sphere and is able to freely rotate, so this type offered dynamic behavior is extremely well-known in metal clusters especially with carbonyls. (R ...
... carbonyls on the surface, it has been supposed that there is a sphere on which the carbonyls are moving. The metal cluster is in fact embedded inside the sphere and is able to freely rotate, so this type offered dynamic behavior is extremely well-known in metal clusters especially with carbonyls. (R ...
Siloxane-containing polymers
... liquid hydrocarbon is to facilitate the removal, by azeo 65 tropic distillation, of any water present in the reaction mixture. Preferably, the his (haloalkyl) siloxane is grad is known in the prior art. Thus, British Pat. No. ually added under anhydrous conditions to a reaction 1,062,418, published ...
... liquid hydrocarbon is to facilitate the removal, by azeo 65 tropic distillation, of any water present in the reaction mixture. Preferably, the his (haloalkyl) siloxane is grad is known in the prior art. Thus, British Pat. No. ually added under anhydrous conditions to a reaction 1,062,418, published ...
Atomic Masses
... • To learn how we define 1 amu (atomic mass unit) • To learn how we derive atomic mass from amu (atomic mass unit) • To learn how atomic mass is calculated using the average natural abundance of isotopes ...
... • To learn how we define 1 amu (atomic mass unit) • To learn how we derive atomic mass from amu (atomic mass unit) • To learn how atomic mass is calculated using the average natural abundance of isotopes ...
Chapter 4 Carbon and the Molecular Diversity of Life
... its ability to form molecules that are large, complex, and diverse. Student understanding of this complexity and diversity is aided by naming and describing typical groups of atoms (functional groups) that are mixed and matched to construct larger carbon-based molecules. Much of the material in this ...
... its ability to form molecules that are large, complex, and diverse. Student understanding of this complexity and diversity is aided by naming and describing typical groups of atoms (functional groups) that are mixed and matched to construct larger carbon-based molecules. Much of the material in this ...
Orbitals - faculty at Chemeketa
... Two sp2-hybridized orbitals overlap to form a s bond p orbitals overlap side-to-side to formation a pi () bond sp2–sp2 s bond and 2p–2p bond result in sharing four electrons and formation of C-C double bond Electrons in the s bond are centered between nuclei Electrons in the bond occupy ...
... Two sp2-hybridized orbitals overlap to form a s bond p orbitals overlap side-to-side to formation a pi () bond sp2–sp2 s bond and 2p–2p bond result in sharing four electrons and formation of C-C double bond Electrons in the s bond are centered between nuclei Electrons in the bond occupy ...
Introduction to Organometallic Chemistry Prof. A.G. Samuelson
... There was a long gap since, from 1981 to 2001, and in 2001 Sharp less Knowles and Noyori were awarded the Nobel prize for their contributions to asymmetric catalysis. And this was completely in the field of organometallic chemistry, where chiral ligands were used by these workers to generate chiral ...
... There was a long gap since, from 1981 to 2001, and in 2001 Sharp less Knowles and Noyori were awarded the Nobel prize for their contributions to asymmetric catalysis. And this was completely in the field of organometallic chemistry, where chiral ligands were used by these workers to generate chiral ...
Liquids and Solids
... surrounded by six others. These layers do not lie directly over those in the first layer, instead they fill the indentations of the layer below. The third layer is in the same position as the first. This is called aba arrangement. ...
... surrounded by six others. These layers do not lie directly over those in the first layer, instead they fill the indentations of the layer below. The third layer is in the same position as the first. This is called aba arrangement. ...
Fp*) Fe(η5-C5Me5)(CO)2
... Synthesis of Fp*-(CtC)3-H (3). K2CO3 (215 mg, 1.56 mmol) was added to 2 (611 mg, 1.56 mmol) dissolved in a mixture of THF (10 mL) and MeOH (10 mL), and the resultant mixture was stirred for 2 h at ambient temperature. After the consumption of 2 was checked by TLC, the volatiles were removed by TLC. ...
... Synthesis of Fp*-(CtC)3-H (3). K2CO3 (215 mg, 1.56 mmol) was added to 2 (611 mg, 1.56 mmol) dissolved in a mixture of THF (10 mL) and MeOH (10 mL), and the resultant mixture was stirred for 2 h at ambient temperature. After the consumption of 2 was checked by TLC, the volatiles were removed by TLC. ...
Materials - Hodder Education
... example high-density polythene (up to 70 per cent). Amorphous (disordered) materials have their particles randomly arranged; there is little order or symmetry. Glass is a familiar example of an amorphous material. Some plastics are also amorphous or have amorphous regions, for example, low-density p ...
... example high-density polythene (up to 70 per cent). Amorphous (disordered) materials have their particles randomly arranged; there is little order or symmetry. Glass is a familiar example of an amorphous material. Some plastics are also amorphous or have amorphous regions, for example, low-density p ...
Effect of soil compositions on the electrochemical corrosion behavior
... Specimens for electrochemical tests were made from Q235 carbon steel whose chemical composition was shown in Table 1. Samples were cut into small squares of 10 mm 6 10 mm, and then covered with epoxy resin except test surface, with a working area of 1 cm2. Each sample was successively polished using ...
... Specimens for electrochemical tests were made from Q235 carbon steel whose chemical composition was shown in Table 1. Samples were cut into small squares of 10 mm 6 10 mm, and then covered with epoxy resin except test surface, with a working area of 1 cm2. Each sample was successively polished using ...
TOPIC 1. ELEMENTS, COMPOUNDS AND MIXTURES.
... occur naturally as the diatomic molecules of their atoms. Most elements including all metals do not occur as discrete molecules at all but instead consist of extremely large numbers of atoms bonded together. The number of constituent atoms in a given sample of such elem ents depends only on the size ...
... occur naturally as the diatomic molecules of their atoms. Most elements including all metals do not occur as discrete molecules at all but instead consist of extremely large numbers of atoms bonded together. The number of constituent atoms in a given sample of such elem ents depends only on the size ...
3.0 Properties of Phosgene
... chlorine is an oxidizer, and methane, hydrogen and carbon monoxide are fuels, a fire can occur in the pipeline without oxygen. At temperatures above 250oF, chlorine will start reacting with steel, weakening the piping and vessels. At 483oF, chlorine will ignite iron and produce a fire. Detection of ...
... chlorine is an oxidizer, and methane, hydrogen and carbon monoxide are fuels, a fire can occur in the pipeline without oxygen. At temperatures above 250oF, chlorine will start reacting with steel, weakening the piping and vessels. At 483oF, chlorine will ignite iron and produce a fire. Detection of ...
oxidation number
... Assign bonding electrons: O is more electronegative than C, so in any bond between C and O, O is assigned or given all of the bonding electrons. For CO2, C shares eight bonding electrons with O. O gets all = 8. C = 0. Assign nonbonding electrons. C has 0, each O = 4. Sum of assigned electrons for C ...
... Assign bonding electrons: O is more electronegative than C, so in any bond between C and O, O is assigned or given all of the bonding electrons. For CO2, C shares eight bonding electrons with O. O gets all = 8. C = 0. Assign nonbonding electrons. C has 0, each O = 4. Sum of assigned electrons for C ...
InorgCh8.2
... Nitrogen is a nonmetal diatomic gas making up 78.1% of Earths’ atmosphere a) Isolated from air in 1772 by Rutherford, Cavendish, and Scheele b) Colorless, odorless, nonreactive gas i. :N≡N: structure has strong stable triple bond ii. Used as inert atmosphere for performing chemical reactions iii. N ...
... Nitrogen is a nonmetal diatomic gas making up 78.1% of Earths’ atmosphere a) Isolated from air in 1772 by Rutherford, Cavendish, and Scheele b) Colorless, odorless, nonreactive gas i. :N≡N: structure has strong stable triple bond ii. Used as inert atmosphere for performing chemical reactions iii. N ...
(cobalamin)-dependent enzymes
... precursor as haem and chlorophyll, but has undergone further elaborate tailoring. Indeed, it has the most complex structure of any biological cofactor, requiring more than 20 genes for its biosynthesis [6]. At the heart of the coenzyme is the cobalt atom that is chelated by a macrocyclic ring called ...
... precursor as haem and chlorophyll, but has undergone further elaborate tailoring. Indeed, it has the most complex structure of any biological cofactor, requiring more than 20 genes for its biosynthesis [6]. At the heart of the coenzyme is the cobalt atom that is chelated by a macrocyclic ring called ...
CBSE Sample Paper for Class 11 Chemistry
... OH¯ ions can combine with H ions more readily than CH3COO¯ ions can do. Hence, OH¯ is a strong base. Alternatively, the conjugate acid of CH3COO¯ and OH¯ are CH3COOH and H2O. As CH3COOH is stronger acid than H2O, the conjugate base of CH3COOH, viz CH3COO¯ will be a weaker base. ...
... OH¯ ions can combine with H ions more readily than CH3COO¯ ions can do. Hence, OH¯ is a strong base. Alternatively, the conjugate acid of CH3COO¯ and OH¯ are CH3COOH and H2O. As CH3COOH is stronger acid than H2O, the conjugate base of CH3COOH, viz CH3COO¯ will be a weaker base. ...
Chem_chap3b
... Calculate the mass of carbon dioxide gas that is required to react with a solution containing 10.0 grams of sodium hydroxide. Calculate the mass of sodium carbonate that is produced when 10.0 grams of sodium hydroxide reacts with an excess of carbon dioxide. ...
... Calculate the mass of carbon dioxide gas that is required to react with a solution containing 10.0 grams of sodium hydroxide. Calculate the mass of sodium carbonate that is produced when 10.0 grams of sodium hydroxide reacts with an excess of carbon dioxide. ...
Organic Molecules
... laboratory, but were still unable to produce the complex molecules that they found in living organisms. It was around this time that a Swedish chemist called Jons Jakob Berzelius suggested that compounds found only in living organisms (the organic compounds) should be grouped separately from those f ...
... laboratory, but were still unable to produce the complex molecules that they found in living organisms. It was around this time that a Swedish chemist called Jons Jakob Berzelius suggested that compounds found only in living organisms (the organic compounds) should be grouped separately from those f ...
Computational study of interaction of alkali metals with C3N nanotubes
... the nanostructure surfaces have been widely studied for several applications such as substantial activity in gasification reactions [1], improved gas sensing of the substrate [2–4], and rechargeable batteries [5, 6]. Furthermore, AMs have good thermal and electrical conductivity; however, the doping ...
... the nanostructure surfaces have been widely studied for several applications such as substantial activity in gasification reactions [1], improved gas sensing of the substrate [2–4], and rechargeable batteries [5, 6]. Furthermore, AMs have good thermal and electrical conductivity; however, the doping ...
Document
... carbonyl determines the type of reactions the carbonyl compound will undergo. • Carbonyl carbons are sp2 hybridized, trigonal planar, and have bond angles that are ~1200. In these ways, the carbonyl group resembles the trigonal planar sp2 hybridized carbons of a C=C. ...
... carbonyl determines the type of reactions the carbonyl compound will undergo. • Carbonyl carbons are sp2 hybridized, trigonal planar, and have bond angles that are ~1200. In these ways, the carbonyl group resembles the trigonal planar sp2 hybridized carbons of a C=C. ...
Carboxylic Acids and the Acidity of the O—H Bond
... carbonyl determines the type of reactions the carbonyl compound will undergo. • Carbonyl carbons are sp2 hybridized, trigonal planar, and have bond angles that are ~1200. In these ways, the carbonyl group resembles the trigonal planar sp2 hybridized carbons of a C=C. ...
... carbonyl determines the type of reactions the carbonyl compound will undergo. • Carbonyl carbons are sp2 hybridized, trigonal planar, and have bond angles that are ~1200. In these ways, the carbonyl group resembles the trigonal planar sp2 hybridized carbons of a C=C. ...
Pdf - Text of NPTEL IIT Video Lectures
... compounds. Since, the chemistry is extensive; we had to split this into 2 lectures. In the first lecture, we covered the fact that most metal carbonyl compounds are in fact most metal carbonyl compounds are in fact typically 18 electron systems. So, they tend to be 18 electron complexes. They also a ...
... compounds. Since, the chemistry is extensive; we had to split this into 2 lectures. In the first lecture, we covered the fact that most metal carbonyl compounds are in fact most metal carbonyl compounds are in fact typically 18 electron systems. So, they tend to be 18 electron complexes. They also a ...
CHAPTER 2 Introduction SECTION A
... Both ion and electron transfer reactions entail the transfer of charge through the interface, which can be measured as the electric current. Based on the definition of the electric double layer, Helmholtz7 proposed a simple geometric model of the interface. This model gave rise to the concept of the ...
... Both ion and electron transfer reactions entail the transfer of charge through the interface, which can be measured as the electric current. Based on the definition of the electric double layer, Helmholtz7 proposed a simple geometric model of the interface. This model gave rise to the concept of the ...
Pdf - Text of NPTEL IIT Video Lectures
... electrons which are available for each of these metal atoms you will always end up with a total of 18 electrons. This is true of nickel, it is true of iron, chromium and even the vanadium case, where you have 5 valence electrons on the vanadium and so you have to add an extra electron to the vanadiu ...
... electrons which are available for each of these metal atoms you will always end up with a total of 18 electrons. This is true of nickel, it is true of iron, chromium and even the vanadium case, where you have 5 valence electrons on the vanadium and so you have to add an extra electron to the vanadiu ...
Allotropes of carbon

Carbon is capable of forming many allotropes due to its valency. Well-known forms of carbon include diamond and graphite. In recent decades many more allotropes and forms of carbon have been discovered and researched including ball shapes such as buckminsterfullerene and sheets such as graphene. Larger scale structures of carbon include nanotubes, nanobuds and nanoribbons. Other unusual forms of carbon exist at very high temperature or extreme pressures.