Preparation and reactions of some lower tungsten halides and
... The tungsten(V) and (VI) halides were hydrolyzed in fifty per cent sodium hydroxide and boiled to assure complete hydrolysis. The solution was cooled and diluted to 250 ml. in a volumetric flask. Aliquots were taken for the tungsten and halide analyses. For the tungsten analysis the solution was aci ...
... The tungsten(V) and (VI) halides were hydrolyzed in fifty per cent sodium hydroxide and boiled to assure complete hydrolysis. The solution was cooled and diluted to 250 ml. in a volumetric flask. Aliquots were taken for the tungsten and halide analyses. For the tungsten analysis the solution was aci ...
PREPARATION, STRUCTURAL STUDIES AND CHEMICAL
... primary alcohols into aldehydes. However, they still have some drawbacks; IBX is insoluble in most useful organic solvents except DMSO, which is not an appropriate solvent in many cases due to its high boiling point which makes it difficult to remove. ...
... primary alcohols into aldehydes. However, they still have some drawbacks; IBX is insoluble in most useful organic solvents except DMSO, which is not an appropriate solvent in many cases due to its high boiling point which makes it difficult to remove. ...
w_4-3 Chemistry of Nitrogen Compounds
... over the pH range 5–9. The apparent activation energy is 26.6 Kcal. The acetaldehyde formed will be oxidized by free chlorine to acetic acid (CH3CHO + HOCl → CH3COOH + HCl), whereas the ammonium ion can undergo oxidation to nitrogen by breakpoint chlorination. Hydroxide ion assisted decomposition of ...
... over the pH range 5–9. The apparent activation energy is 26.6 Kcal. The acetaldehyde formed will be oxidized by free chlorine to acetic acid (CH3CHO + HOCl → CH3COOH + HCl), whereas the ammonium ion can undergo oxidation to nitrogen by breakpoint chlorination. Hydroxide ion assisted decomposition of ...
g - mrnicholsscience
... …that the one with more mass is in excess—you might need more of it. If 150 g nitrogen and 40 g hydrogen make ammonia… 8g hydrogen is left over …that the one with more moles is in excess—you might need more of it. If 5 moles oxygen and 8 moles hydrogen make water… 1 mol oxygen is left over ...
... …that the one with more mass is in excess—you might need more of it. If 150 g nitrogen and 40 g hydrogen make ammonia… 8g hydrogen is left over …that the one with more moles is in excess—you might need more of it. If 5 moles oxygen and 8 moles hydrogen make water… 1 mol oxygen is left over ...
teaching and learning materials - UNESDOC
... in many different parts of the world - with special emphasis on the developing countries to create and develop projects on chemical sciences. Professor Glen Seaborg and I, together with all our colleagues, have always tried to find the best solutions during the development stages of such projects. W ...
... in many different parts of the world - with special emphasis on the developing countries to create and develop projects on chemical sciences. Professor Glen Seaborg and I, together with all our colleagues, have always tried to find the best solutions during the development stages of such projects. W ...
Questions
... III The acidic solution in the beaker was filtered into a 250 cm3 volumetric flask. A small amount of solid impurity remained in the filter paper. The solution in the volumetric flask was carefully made up to 250 cm3 with distilled water. IV A pipette was used to transfer 25.0 cm3 portions of the ac ...
... III The acidic solution in the beaker was filtered into a 250 cm3 volumetric flask. A small amount of solid impurity remained in the filter paper. The solution in the volumetric flask was carefully made up to 250 cm3 with distilled water. IV A pipette was used to transfer 25.0 cm3 portions of the ac ...
Chemistry of CHLORINE
... Electronegativity is the ease/tendency of gaining/ acquiring electrons by an element during chemical reactions. It does not involve use of energy but theoretical arbitrary Pauling’s scale of measurements. (g) (i) 5cm3 of sodium chloride, Sodium bromide and Sodium iodide solutions were put separately ...
... Electronegativity is the ease/tendency of gaining/ acquiring electrons by an element during chemical reactions. It does not involve use of energy but theoretical arbitrary Pauling’s scale of measurements. (g) (i) 5cm3 of sodium chloride, Sodium bromide and Sodium iodide solutions were put separately ...
Cyanide Destruction with Chlorine Dioxide
... should be considered when levels of oxidizable cyanide are too high to be processed by a biological treatment system. Chlorine dioxide Chlorine dioxide is unique in its ability to be used at moderately basic pH's. All other oxidants require pH's greater than 12. In the case of chlorine this is neces ...
... should be considered when levels of oxidizable cyanide are too high to be processed by a biological treatment system. Chlorine dioxide Chlorine dioxide is unique in its ability to be used at moderately basic pH's. All other oxidants require pH's greater than 12. In the case of chlorine this is neces ...
The science of chemistry is concerned with the composition
... cold. Solid mercury has a density of 14.1 g cm–3 slightly greater than that of the liquid. When mercury is heated, it remains a liquid until quite a high temperature, finally boiling at 356.6°C to give an invisible vapor. Even at low concentrations gaseous mercury is extremely toxic if breathed into ...
... cold. Solid mercury has a density of 14.1 g cm–3 slightly greater than that of the liquid. When mercury is heated, it remains a liquid until quite a high temperature, finally boiling at 356.6°C to give an invisible vapor. Even at low concentrations gaseous mercury is extremely toxic if breathed into ...
The science of chemistry is concerned with the
... cold. Solid mercury has a density of 14.1 g cm–3 slightly greater than that of the liquid. When mercury is heated, it remains a liquid until quite a high temperature, finally boiling at 356.6°C to give an invisible vapor. Even at low concentrations gaseous mercury is extremely toxic if breathed into ...
... cold. Solid mercury has a density of 14.1 g cm–3 slightly greater than that of the liquid. When mercury is heated, it remains a liquid until quite a high temperature, finally boiling at 356.6°C to give an invisible vapor. Even at low concentrations gaseous mercury is extremely toxic if breathed into ...
Chemistry 139
... (2 pts) One of the main features of Dalton's atomic theory which is no longer considered valid is: All atoms of each element are identical in every respect. Which of the following is the best explanation of why this feature is no longer considered valid? ...
... (2 pts) One of the main features of Dalton's atomic theory which is no longer considered valid is: All atoms of each element are identical in every respect. Which of the following is the best explanation of why this feature is no longer considered valid? ...
Groups 2 and 7
... Electronegativity of the halogens decreases down the group due to an increase in atomic radius. Increased nuclear charge has no significant effect because there are more electron shells and more shielding. Iodine atoms therefore attract electron density in a covalent bond less strongly than fluorine ...
... Electronegativity of the halogens decreases down the group due to an increase in atomic radius. Increased nuclear charge has no significant effect because there are more electron shells and more shielding. Iodine atoms therefore attract electron density in a covalent bond less strongly than fluorine ...
Chemical Reactions
... than one reactant, and asked to calculate the amount of product formed. The quantities of reactants might be such that both react completely, or one might react completely, and the other(s) might be in excess. These are called limiting reagent problems, since the quantity of one of the reacts will l ...
... than one reactant, and asked to calculate the amount of product formed. The quantities of reactants might be such that both react completely, or one might react completely, and the other(s) might be in excess. These are called limiting reagent problems, since the quantity of one of the reacts will l ...
Mechanistic and Computational Studies of Ferroin, Simple Organic
... study was to model the oxidation of bromomalonic acid (BMA) by Fe(1,10-phenanthroline)33+ (a.k.a. ferriin) and determine the mechanism by which electrons are transferred. Following many months of unpublished work, the initial models were determined to be inadequate and the project underwent a transf ...
... study was to model the oxidation of bromomalonic acid (BMA) by Fe(1,10-phenanthroline)33+ (a.k.a. ferriin) and determine the mechanism by which electrons are transferred. Following many months of unpublished work, the initial models were determined to be inadequate and the project underwent a transf ...
Word - chemmybear.com
... When 2.0 mol of carbon disulfide and 4.0 mol of chlorine are placed in a 1.0 Liter flask, the following equilibrium system results. At equilibrium, the flask is found to contain 0.30 mol of carbon tetrachloride. What quantities of the other components are present in this equilibrium mixture? CS2(g) ...
... When 2.0 mol of carbon disulfide and 4.0 mol of chlorine are placed in a 1.0 Liter flask, the following equilibrium system results. At equilibrium, the flask is found to contain 0.30 mol of carbon tetrachloride. What quantities of the other components are present in this equilibrium mixture? CS2(g) ...
South Pasadena · AP Chemistry
... When 2.0 mol of carbon disulfide and 4.0 mol of chlorine are placed in a 1.0 Liter flask, the following equilibrium system results. At equilibrium, the flask is found to contain 0.30 mol of carbon tetrachloride. What quantities of the other components are present in this equilibrium mixture? CS2(g) ...
... When 2.0 mol of carbon disulfide and 4.0 mol of chlorine are placed in a 1.0 Liter flask, the following equilibrium system results. At equilibrium, the flask is found to contain 0.30 mol of carbon tetrachloride. What quantities of the other components are present in this equilibrium mixture? CS2(g) ...
GQ2613291336
... temperature (as illustrated in the fig. (1)) after thermal equilibrium has been reached, bromine (Br2) 10.77ml (0.015mol, 1.392mol dm-3) added which is also allowed in 298K thermostat to come to temperature equilibrium before use. A pure nitrogen gas with constant speed 10 dm3/hr. by using gas flow ...
... temperature (as illustrated in the fig. (1)) after thermal equilibrium has been reached, bromine (Br2) 10.77ml (0.015mol, 1.392mol dm-3) added which is also allowed in 298K thermostat to come to temperature equilibrium before use. A pure nitrogen gas with constant speed 10 dm3/hr. by using gas flow ...
Worksheet 1 - Oxidation/Reduction Reactions Oxidation number
... ___Br- + ___H+ + ___MnO4- ___Br2 + ___Mn2+ + ___H2O Which compound is the oxidizing agent? Which compound is the reducing agent? Notice that there are protons (H+) present in the reactants. This indicates that the reaction is carried out in an acidic solution. To carry this out in a basic solution ...
... ___Br- + ___H+ + ___MnO4- ___Br2 + ___Mn2+ + ___H2O Which compound is the oxidizing agent? Which compound is the reducing agent? Notice that there are protons (H+) present in the reactants. This indicates that the reaction is carried out in an acidic solution. To carry this out in a basic solution ...
The GENESIS Device TM
... THE TECHNOLOGY TRU BLU is a chemical that is 98% pure sodium bromide. Sodium bromide -“NaBr”-is the kissing cousin of sodium chloride – “NaCl” – otherwise known as table salt). TRU BLU dissolves when added to your spa’s water. When your spa is circulating, the TRU BLU circulates through the GENESIS ...
... THE TECHNOLOGY TRU BLU is a chemical that is 98% pure sodium bromide. Sodium bromide -“NaBr”-is the kissing cousin of sodium chloride – “NaCl” – otherwise known as table salt). TRU BLU dissolves when added to your spa’s water. When your spa is circulating, the TRU BLU circulates through the GENESIS ...
Compounds of Chlorine
... Dichlorine tetraoxide (Cl2 O4 ) is commonly called chlorine perchlorate as a consequence of its structure (Figure 4c). Dichlorine hexaoxide (Cl2 O6 ) is an unstable red oil that has the ionic structure in the solid state: [ClO2 ]+ [ClO4 ]- . Dichlorine heptoxide (Cl2 O7 ) is a relatively stable oil, ...
... Dichlorine tetraoxide (Cl2 O4 ) is commonly called chlorine perchlorate as a consequence of its structure (Figure 4c). Dichlorine hexaoxide (Cl2 O6 ) is an unstable red oil that has the ionic structure in the solid state: [ClO2 ]+ [ClO4 ]- . Dichlorine heptoxide (Cl2 O7 ) is a relatively stable oil, ...
inorganic chemistry
... Halogens are highly reactive, and as such can be harmful or lethal to biological organisms in sufficient quantities. This high reactivity is due to the atoms being highly electronegative due to their high effective nuclear charge. They can gain an electron by reacting with atoms of other elements. F ...
... Halogens are highly reactive, and as such can be harmful or lethal to biological organisms in sufficient quantities. This high reactivity is due to the atoms being highly electronegative due to their high effective nuclear charge. They can gain an electron by reacting with atoms of other elements. F ...
Section A oxide in molten cryolite?
... Q2 A cheap carbon monoxide detector for a gas heater consists of a patch containing palladium chloride crystals. When carbon monoxide is present, the crystals turn from orange to black as the following reaction takes place. CO(g) + PdCl2(s) + H2O(l) → CO2(g) + Pd(s) + 2HCl(aq) orange black Which is ...
... Q2 A cheap carbon monoxide detector for a gas heater consists of a patch containing palladium chloride crystals. When carbon monoxide is present, the crystals turn from orange to black as the following reaction takes place. CO(g) + PdCl2(s) + H2O(l) → CO2(g) + Pd(s) + 2HCl(aq) orange black Which is ...
17.2.3 Interhalogen compounds(65-67)
... Diatomic interhalogens, XY All six possible diatomic compounds between F, C1, Br and I are known. Indeed, IC1 was first made (independently) by J. L. Gay Lussac and H. Davy in 1813-4 soon after the isolation of the parent halogens themselves, and its existence led J. von Liebig to m i s s the discov ...
... Diatomic interhalogens, XY All six possible diatomic compounds between F, C1, Br and I are known. Indeed, IC1 was first made (independently) by J. L. Gay Lussac and H. Davy in 1813-4 soon after the isolation of the parent halogens themselves, and its existence led J. von Liebig to m i s s the discov ...
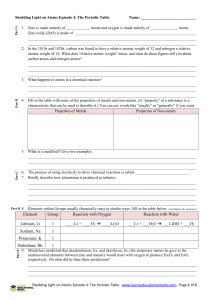
Element Group Reaction with Oxygen Reaction with Water Lithium
... atomic weight of 14. What does “relative atomic weight” mean, and what do these figures tell you about carbon atoms and nitrogen atoms? ______________________________________________________________________________________ _____________________________________________________________________________ ...
... atomic weight of 14. What does “relative atomic weight” mean, and what do these figures tell you about carbon atoms and nitrogen atoms? ______________________________________________________________________________________ _____________________________________________________________________________ ...
Bromine

Bromine (from Greek: βρῶμος, brómos, meaning ""strong-smelling"" or ""stench"") is a chemical element with symbol Br, and atomic number 35. It is a halogen. The element was isolated independently by two chemists, Carl Jacob Löwig and Antoine Jerome Balard, in 1825–1826. Elemental bromine is a fuming red-brown liquid at room temperature, corrosive and toxic, with properties between those of chlorine and iodine. Free bromine does not occur in nature, but occurs as colorless soluble crystalline mineral halide salts, analogous to table salt.Bromine is rarer than about three-quarters of elements in the Earth's crust. The high solubility of bromide ions has caused its accumulation in the oceans, and commercially the element is easily extracted from brine pools, mostly in the United States, Israel and China. About 556,000 tonnes were produced in 2007, an amount similar to the far more abundant element magnesium.At high temperatures, organobromine compounds readily convert to free bromine atoms, a process which has the effect of stopping free radical chemical chain reactions. This effect makes organobromine compounds useful as fire retardants; more than half the bromine produced industrially worldwide each year is put to this use. Unfortunately, the same property causes sunlight to convert volatile organobromine compounds to free bromine atoms in the atmosphere, and an unwanted side effect of this process is ozone depletion. As a result, many organobromide compounds that were formerly in common use—such as the pesticide, methyl bromide—have been abandoned. Bromine compounds are still used for purposes such as in well drilling fluids, in photographic film, and as an intermediate in the manufacture of organic chemicals.Bromine has been long believed to have no essential function in mammals, but recent research suggests that bromine is necessary for tissue development. In addition, bromine is used preferentially over chlorine by one antiparasitic enzyme in the human immune system. Organobromides are needed and produced enzymatically from bromide by some lower life forms in the sea, particularly algae, and the ash of seaweed was one source of bromine's discovery. As a pharmaceutical, the simple bromide ion, Br−, has inhibitory effects on the central nervous system, and bromide salts were once a major medical sedative, before being replaced by shorter-acting drugs. They retain niche uses as antiepileptics.