The History of the Atom - Brookville Local Schools
... of hydrogen combine with 16 grams of oxygen. In another compound, 2 grams of hydrogen combine with 32 grams of hydrogen. The ratio of oxygen in one compound to another is 32:16 = 2:1. Why is this important? It leads to the far more important understanding that atoms always combine in whole number ...
... of hydrogen combine with 16 grams of oxygen. In another compound, 2 grams of hydrogen combine with 32 grams of hydrogen. The ratio of oxygen in one compound to another is 32:16 = 2:1. Why is this important? It leads to the far more important understanding that atoms always combine in whole number ...
Lecture 02 Post. Rutherford Model
... NB. mass number ≠ atomic mass (average mass number is not normally used ) General Formula: x1 I1 + x2 I2 + ... = E where, x ...fraction, abundance; x1 +x2 + ... = 1 I atomic mass of isotope E ... atomic mass of “element” ...
... NB. mass number ≠ atomic mass (average mass number is not normally used ) General Formula: x1 I1 + x2 I2 + ... = E where, x ...fraction, abundance; x1 +x2 + ... = 1 I atomic mass of isotope E ... atomic mass of “element” ...
Chapter 4 - Elements and the Periodic Table I. Introduction to atoms
... B. Families of nonmetals 1. Carbon family, Group 14 a. Can gain, lose or share 4 electrons b. Carbon only nonmetals in group c. Important element for life 2. Nitrogen family, Group 15 a. Contains 2 nonmetals, nitrogen and phosphorus b. Share 3 electrons c. Does not react with other elements d. Occu ...
... B. Families of nonmetals 1. Carbon family, Group 14 a. Can gain, lose or share 4 electrons b. Carbon only nonmetals in group c. Important element for life 2. Nitrogen family, Group 15 a. Contains 2 nonmetals, nitrogen and phosphorus b. Share 3 electrons c. Does not react with other elements d. Occu ...
Chapter 1000A - U of L Class Index
... • Atoms of the same element must have the same atomic number but different mass numbers are possible. • These are . Most elements have more than one naturally occurring isotope: H H H ...
... • Atoms of the same element must have the same atomic number but different mass numbers are possible. • These are . Most elements have more than one naturally occurring isotope: H H H ...
elements and isotopes - vocabulary
... the nucleus which is the same for all atoms of the element. isotope A species of atom; each atom of a particular isotope has a specific number of protons and a specific number of neutrons in the nucleus which are the same for all atoms of the isotope, but are not necessarily equal to each other. ato ...
... the nucleus which is the same for all atoms of the element. isotope A species of atom; each atom of a particular isotope has a specific number of protons and a specific number of neutrons in the nucleus which are the same for all atoms of the isotope, but are not necessarily equal to each other. ato ...
Atoms, Molecules and Ions Part 2
... Isotopes • Dalton, had proposed that all atoms of the same element must have the same mass. • One of the 2 flaws in Dalton’s theory was, atoms of the same element can have DIFFERENT masses. • The mass of an atom is due to the mass of the protons and the neutrons in that atom. • Isotopes are atoms o ...
... Isotopes • Dalton, had proposed that all atoms of the same element must have the same mass. • One of the 2 flaws in Dalton’s theory was, atoms of the same element can have DIFFERENT masses. • The mass of an atom is due to the mass of the protons and the neutrons in that atom. • Isotopes are atoms o ...
Atomic Structure Worksheet
... 1. The extremely low-mass, negatively charged particle found outside the nucleus is called an electron. 2. The number of protons in the nucleus of the atom is indicated by the atomic number. 3. The symbol for an element is either a capital letter or possibly a capital letter followed by a lower case ...
... 1. The extremely low-mass, negatively charged particle found outside the nucleus is called an electron. 2. The number of protons in the nucleus of the atom is indicated by the atomic number. 3. The symbol for an element is either a capital letter or possibly a capital letter followed by a lower case ...
AS II Rutherford Model
... ∴ atomic mass of Ag = 0.5184 × 106.905 u + 0.4816 × 108.905 u = 107.87 u NB. mass number ≠ atomic mass (average mass number is not normally used ) General Formula: x1 I1 + x2 I2 + ... = E where, x ...fraction, abundance; x1 +x2 + ... = 1 I atomic mass of isotope E ... atomic mass of “element” ...
... ∴ atomic mass of Ag = 0.5184 × 106.905 u + 0.4816 × 108.905 u = 107.87 u NB. mass number ≠ atomic mass (average mass number is not normally used ) General Formula: x1 I1 + x2 I2 + ... = E where, x ...fraction, abundance; x1 +x2 + ... = 1 I atomic mass of isotope E ... atomic mass of “element” ...
PHY–309 L. Solutions for homework set # 10. Textbook question Q
... This wikipedia page shows a diagram of a color CRT tube. In any CRT tube — a TV, a monitor, an oscilloscope, or an X-ray tube — a beam of electrons hits the anode (a screen, or just a piece of metal) at high speed. When the atoms in the anode are hit by fast electrons, sometimes the inner electrons ...
... This wikipedia page shows a diagram of a color CRT tube. In any CRT tube — a TV, a monitor, an oscilloscope, or an X-ray tube — a beam of electrons hits the anode (a screen, or just a piece of metal) at high speed. When the atoms in the anode are hit by fast electrons, sometimes the inner electrons ...
Cobalt isotopes in industry 60Co is used to irradiate food sources as
... from a 60Co source. This technique of sterilization is generally much cheaper and more effective than steam-heat sterilization because it is a cold process. For example, it can be performed on packaged items, such as disposable syringes. This sterilization technique is applicable to a wide range of ...
... from a 60Co source. This technique of sterilization is generally much cheaper and more effective than steam-heat sterilization because it is a cold process. For example, it can be performed on packaged items, such as disposable syringes. This sterilization technique is applicable to a wide range of ...
PS 2.2 - S2TEM Centers SC
... Easter Egg Isotopes Introduction to the lesson: Isotopes have the same atomic number and hence nearly identical chemical behavior but different atomic masses. Most elements found in nature are mixtures of several isotopes; tin, for example, has 10 isotopes. In most cases, only stable isotopes of ele ...
... Easter Egg Isotopes Introduction to the lesson: Isotopes have the same atomic number and hence nearly identical chemical behavior but different atomic masses. Most elements found in nature are mixtures of several isotopes; tin, for example, has 10 isotopes. In most cases, only stable isotopes of ele ...
September 20th, 2012
... Magnesium has three isotopes, with mass numbers 24, 25, and 26. a) write the complete chemical symbol (subscript and superscript) of each. b) how many neutrons are in an atom of ...
... Magnesium has three isotopes, with mass numbers 24, 25, and 26. a) write the complete chemical symbol (subscript and superscript) of each. b) how many neutrons are in an atom of ...
The Atom - VCE Chemistry
... • These findings provided evidence that the atom could in fact consist of smaller particles, that is, it is not indivisible. • Radioactivity was also a useful tool with which to probe the structure of the atom. • We now know that the stability of the nucleus depends upon the ratio of protons to neut ...
... • These findings provided evidence that the atom could in fact consist of smaller particles, that is, it is not indivisible. • Radioactivity was also a useful tool with which to probe the structure of the atom. • We now know that the stability of the nucleus depends upon the ratio of protons to neut ...
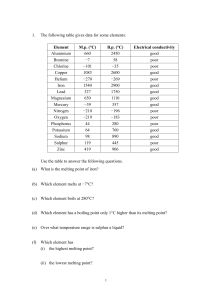
Answers
... (a) Group I [1] (b) All are soft metals. [1] (c) Lithium would float on water, [1] producing gas steadily. [1] (d) Potassium would melt to a silvery ball [1] which moves about very quickly on the water surface, [1] producing a hissing sound, [1] burning spontaneously with a lilac flame [1] before fi ...
... (a) Group I [1] (b) All are soft metals. [1] (c) Lithium would float on water, [1] producing gas steadily. [1] (d) Potassium would melt to a silvery ball [1] which moves about very quickly on the water surface, [1] producing a hissing sound, [1] burning spontaneously with a lilac flame [1] before fi ...
Atomic structure - ISA DP Chemistry with Ms Tsui
... • The mass spectrometer is used to determine the relative atomic mass of an element from its isotopic composition. ...
... • The mass spectrometer is used to determine the relative atomic mass of an element from its isotopic composition. ...
File
... The carbon-14 which is formed is radioactive and decays producing nitrogen again – there is therefore a fixed amount of carbon-14 in the environment which is a balance between the rate at which it is formed and the rate at which it decays ...
... The carbon-14 which is formed is radioactive and decays producing nitrogen again – there is therefore a fixed amount of carbon-14 in the environment which is a balance between the rate at which it is formed and the rate at which it decays ...
Nuclear Chemistry I: Radioactivity Reading: Moore chapter 20
... There are certain stable proton/neutron configurations in nuclei. In a graph of number of neutrons N (x-axis) vs. number of protons Z (y-axis), the line of maximum nuclear stability has a slope corresponding to a 1:1 proton:neutron ratio for the lightest nuclei (==> 20Ca). Examples: 24He, 714N, 2040 ...
... There are certain stable proton/neutron configurations in nuclei. In a graph of number of neutrons N (x-axis) vs. number of protons Z (y-axis), the line of maximum nuclear stability has a slope corresponding to a 1:1 proton:neutron ratio for the lightest nuclei (==> 20Ca). Examples: 24He, 714N, 2040 ...
Dynamic Earth Unit 2 lesson 3 Absolute Dating
... • Scientists use many different isotopes for radiometric dating. • The type of isotope used depends on the type of material being dated. • The half-life of the isotope used is also very important. It can’t be too short or too long compared to the age of the sample. ...
... • Scientists use many different isotopes for radiometric dating. • The type of isotope used depends on the type of material being dated. • The half-life of the isotope used is also very important. It can’t be too short or too long compared to the age of the sample. ...
- EdShare - University of Southampton
... We quote carbon to the same PDB (Pee Dee Belemnite) standard as for oxygen isotopes ...
... We quote carbon to the same PDB (Pee Dee Belemnite) standard as for oxygen isotopes ...
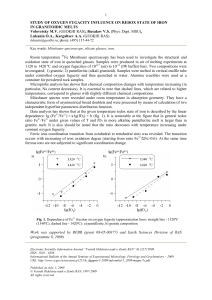
Study of oxygen fugacity influence on redox state of iron in
... investigated: 1) granitic; 2) pantelleritic (alkali granitoid). Samples were melted in vertical muffle tube under controlled oxygen fugacity and then quenched in water. Alumina crucibles were used as a container for powdered rock samples. Microprobe analysis has shown that chemical composition chang ...
... investigated: 1) granitic; 2) pantelleritic (alkali granitoid). Samples were melted in vertical muffle tube under controlled oxygen fugacity and then quenched in water. Alumina crucibles were used as a container for powdered rock samples. Microprobe analysis has shown that chemical composition chang ...
Practice problems for chapter 1, 2 and 3 1) A small amount of salt
... B) 2Mg + O2 → 2MgO C) 2N2 + 3 H2 → 2NH3 D) 2CH4 + 4O2 → 2CO2 + 4H2O E) Cd(NO3)O2 + Na2S → CdS + 2NaNO3 4) Write the product of this combination reaction? Al (s) + I2 (s) → __________ 5) Which of the following are combustion reactions? 1) CH4 (g) + O2 (g) → CO2 (g) + H2O (l) 2) CaO (s) + CO2 (g) → Ca ...
... B) 2Mg + O2 → 2MgO C) 2N2 + 3 H2 → 2NH3 D) 2CH4 + 4O2 → 2CO2 + 4H2O E) Cd(NO3)O2 + Na2S → CdS + 2NaNO3 4) Write the product of this combination reaction? Al (s) + I2 (s) → __________ 5) Which of the following are combustion reactions? 1) CH4 (g) + O2 (g) → CO2 (g) + H2O (l) 2) CaO (s) + CO2 (g) → Ca ...
Isotopes and Radioactive Decay
... NUCLEAR REACTION: Reaction that can change an _______________________into a new ___________________. It requires a change in an atom’s _______________________________. ...
... NUCLEAR REACTION: Reaction that can change an _______________________into a new ___________________. It requires a change in an atom’s _______________________________. ...
PRACTICE PROBLEMS EXAM 1,2 and 3 1311
... B) 2Mg + O2 → 2MgO C) 2N2 + 3 H2 → 2NH3 D) 2CH4 + 4O2 → 2CO2 + 4H2O E) Cd(NO3)O2 + Na2S → CdS + 2NaNO3 4) Write the product of this combination reaction? Al (s) + I2 (s) → __________ 5) Which of the following are combustion reactions? 1) CH4 (g) + O2 (g) → CO2 (g) + H2O (l) 2) CaO (s) + CO2 (g) → Ca ...
... B) 2Mg + O2 → 2MgO C) 2N2 + 3 H2 → 2NH3 D) 2CH4 + 4O2 → 2CO2 + 4H2O E) Cd(NO3)O2 + Na2S → CdS + 2NaNO3 4) Write the product of this combination reaction? Al (s) + I2 (s) → __________ 5) Which of the following are combustion reactions? 1) CH4 (g) + O2 (g) → CO2 (g) + H2O (l) 2) CaO (s) + CO2 (g) → Ca ...
Isotope analysis

Isotope analysis is the identification of isotopic signature, the distribution of certain stable isotopes and chemical elements within chemical compounds. This can be applied to a food web to make it possible to draw direct inferences regarding diet, trophic level, and subsistence. Variations in isotope ratios from isotopic fractionation are measured using mass spectrometry, which separates the different isotopes of an element on the basis of their mass-to-charge ratio.The ratios of isotopic oxygen are also differentially affected by global weather patterns and regional topography as moisture is transported. Areas of lower humidity cause the preferential loss of 18O water in the form of vapor and precipitation. Furthermore, evaporated 16O water returns preferentially to the atmospheric system as it evaporates and 18O remains in liquid form or is incorporated into the body water of plants and animals.