Periodic Table and the Atom Answers
... Guch’s Helpdesk” section of my website (http://www.chemfiesta.com). If you’re having problems with stoichiometry problems, I would highly suggest consulting this section of the site before answering these questions. When doing stoichiometry problems, people are frequently worried by statements such ...
... Guch’s Helpdesk” section of my website (http://www.chemfiesta.com). If you’re having problems with stoichiometry problems, I would highly suggest consulting this section of the site before answering these questions. When doing stoichiometry problems, people are frequently worried by statements such ...
Comparison of 2008 to 2000 SCH3U_ud
... compounds can be released into the environment, including cadmium, arsenic, sulphur dioxide, and mercury, all of which can endanger the health and safety of local populations. Sample questions: What are some chemical reactions used in the manufacture of paper? How might the reactants or products of ...
... compounds can be released into the environment, including cadmium, arsenic, sulphur dioxide, and mercury, all of which can endanger the health and safety of local populations. Sample questions: What are some chemical reactions used in the manufacture of paper? How might the reactants or products of ...
AP Chemistry Summer Assignment
... About the AP Chemistry Course: Since this is a college level course taught in high school, it is very demanding, both in time and effort required. Much of the work involves solving math-type story problems. Homework is assigned each day through all three trimesters. The three weeks before the AP Exa ...
... About the AP Chemistry Course: Since this is a college level course taught in high school, it is very demanding, both in time and effort required. Much of the work involves solving math-type story problems. Homework is assigned each day through all three trimesters. The three weeks before the AP Exa ...
GCSE - WordPress.com
... 4. Explain the following in terms of bonding and structure ideas :. (i) Silicon dioxide and carbon dioxide both contain covalent bonds but the former melts at 1700oC whereas the latter is a gas at 0oC. (ii) Sodium oxide, carbon dioxide and silicon dioxide are all poor conductors of electricity ...
... 4. Explain the following in terms of bonding and structure ideas :. (i) Silicon dioxide and carbon dioxide both contain covalent bonds but the former melts at 1700oC whereas the latter is a gas at 0oC. (ii) Sodium oxide, carbon dioxide and silicon dioxide are all poor conductors of electricity ...
Homework Booklet [4,S]
... 4. Explain the following in terms of bonding and structure ideas :. (i) Silicon dioxide and carbon dioxide both contain covalent bonds but the former melts at 1700oC whereas the latter is a gas at 0oC. (ii) Sodium oxide, carbon dioxide and silicon dioxide are all poor conductors of electricity ...
... 4. Explain the following in terms of bonding and structure ideas :. (i) Silicon dioxide and carbon dioxide both contain covalent bonds but the former melts at 1700oC whereas the latter is a gas at 0oC. (ii) Sodium oxide, carbon dioxide and silicon dioxide are all poor conductors of electricity ...
LESSON 23: Exploding Bags
... the structure or composition of the materials change. Chemical reactions occur around us all the time. When a chemical change is complete, the resulting substance(s) is/are different from the original substance(s). The substance or substances that start a chemical reaction are called reactants. The ...
... the structure or composition of the materials change. Chemical reactions occur around us all the time. When a chemical change is complete, the resulting substance(s) is/are different from the original substance(s). The substance or substances that start a chemical reaction are called reactants. The ...
664
... trace concentrations. The gas is used as an anesthetic, especially in dentistry and minor surgery. It produces mild hysteria and laughter preceding the anesthetic effect, for which reason it also is called “laughing gas.” It is used as an aerosol propellant, an aerating agent for whipped cream, and ...
... trace concentrations. The gas is used as an anesthetic, especially in dentistry and minor surgery. It produces mild hysteria and laughter preceding the anesthetic effect, for which reason it also is called “laughing gas.” It is used as an aerosol propellant, an aerating agent for whipped cream, and ...
workbook Chem (WP)
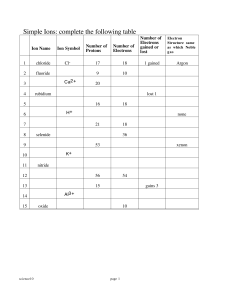
... 1. identify the theories and their authors represented by the following: a. atom looks like this b. atom looks like a planetary system c. the electrons move around the nucleus in an unknown path. d. key was the “Gold foil experiment”. Ions 1. Why do ions form? 2. Draw the Bohr diagram for the atom a ...
... 1. identify the theories and their authors represented by the following: a. atom looks like this b. atom looks like a planetary system c. the electrons move around the nucleus in an unknown path. d. key was the “Gold foil experiment”. Ions 1. Why do ions form? 2. Draw the Bohr diagram for the atom a ...
CHEMISTRY SEC 06 SYLLABUS
... 4. Scheme of Assessment The examination will consist of two written papers, each of two hours’ duration, and an assessment of practical work in chemistry. Questions will be set in English and must be answered in English. The examination will be structured as follows: Paper I consists of a written pa ...
... 4. Scheme of Assessment The examination will consist of two written papers, each of two hours’ duration, and an assessment of practical work in chemistry. Questions will be set in English and must be answered in English. The examination will be structured as follows: Paper I consists of a written pa ...
For detailed information on Aluminium Production
... A modern aluminum reduction cell, commonly called a pot, is made of a rectangular steel shell, (At Balco15.780m long by 4.180 m wide by 1.372m high), lined with refractory thermal insulation. Inside the shell there is an inner lining of SiC to contain the highly corrosive molten fluoride electrolyte ...
... A modern aluminum reduction cell, commonly called a pot, is made of a rectangular steel shell, (At Balco15.780m long by 4.180 m wide by 1.372m high), lined with refractory thermal insulation. Inside the shell there is an inner lining of SiC to contain the highly corrosive molten fluoride electrolyte ...
CHEMISTRY SEC 06 SYLLABUS
... 4. Scheme of Assessment The examination will consist of two written papers, each of two hours’ duration, and an assessment of practical work in chemistry. Questions will be set in English and must be answered in English. The examination will be structured as follows: Paper I consists of a written pa ...
... 4. Scheme of Assessment The examination will consist of two written papers, each of two hours’ duration, and an assessment of practical work in chemistry. Questions will be set in English and must be answered in English. The examination will be structured as follows: Paper I consists of a written pa ...
CHEMISTRY SEC 06 SYLLABUS
... 4. Scheme of Assessment The examination will consist of two written papers, each of two hours’ duration, and an assessment of practical work in chemistry. Questions will be set in English and must be answered in English. The examination will be structured as follows: Paper I consists of a written pa ...
... 4. Scheme of Assessment The examination will consist of two written papers, each of two hours’ duration, and an assessment of practical work in chemistry. Questions will be set in English and must be answered in English. The examination will be structured as follows: Paper I consists of a written pa ...
CHEMISTRY SEC 06 SYLLABUS
... 4. Scheme of Assessment The examination will consist of two written papers, each of two hours’ duration, and an assessment of practical work in chemistry. Questions will be set in English and must be answered in English. The examination will be structured as follows: Paper I consists of a written pa ...
... 4. Scheme of Assessment The examination will consist of two written papers, each of two hours’ duration, and an assessment of practical work in chemistry. Questions will be set in English and must be answered in English. The examination will be structured as follows: Paper I consists of a written pa ...
aq - HCC Learning Web
... Balance the Chemical Equations • Aqueous solutions of lead(II) nitrate and potassium iodide produce a yellow precipitate of lead(II) iodide and an aqueous solution of potassium nitrate Pb(NO3)2(aq) + KI(aq) PbI2(s) + KNO3(aq) • Aqueous solutions of calcium nitrate and sodium carbonate react to gi ...
... Balance the Chemical Equations • Aqueous solutions of lead(II) nitrate and potassium iodide produce a yellow precipitate of lead(II) iodide and an aqueous solution of potassium nitrate Pb(NO3)2(aq) + KI(aq) PbI2(s) + KNO3(aq) • Aqueous solutions of calcium nitrate and sodium carbonate react to gi ...
Reaction of niobium with water
... Tantalum is extracted from the ores by first fusing the ore with alkali, and then extracting the resultant mixture into hydrofluoric acid, HF. Current methodology involves the separation of tantalum from these acid solutions using a liquid-liquid extraction technique. In this process tantalum salts ...
... Tantalum is extracted from the ores by first fusing the ore with alkali, and then extracting the resultant mixture into hydrofluoric acid, HF. Current methodology involves the separation of tantalum from these acid solutions using a liquid-liquid extraction technique. In this process tantalum salts ...
chem equation Pkt Student2
... A reactant or product in the solid state Alternative to (s); used only for a precipitate (solid) falling out of solution A reactant or product in the liquid state A reactant or product in aqueous solution (dissolved in water) A reactant or product in the gaseous state Alternative to (g); used only f ...
... A reactant or product in the solid state Alternative to (s); used only for a precipitate (solid) falling out of solution A reactant or product in the liquid state A reactant or product in aqueous solution (dissolved in water) A reactant or product in the gaseous state Alternative to (g); used only f ...
Chemical and physical changes
... ……………….. and in their place new ones appear. C. The ……………….. changes are called chemical ……………….. . D. Pure substances can be: ……………….. substances and ……………….. . E. ……………….. substance is which ……………….. not disappear and does not give rise to other different ones by ……………….. or ……………….. . F. ……………….. ...
... ……………….. and in their place new ones appear. C. The ……………….. changes are called chemical ……………….. . D. Pure substances can be: ……………….. substances and ……………….. . E. ……………….. substance is which ……………….. not disappear and does not give rise to other different ones by ……………….. or ……………….. . F. ……………….. ...
Stoichiometry Notes
... Definition: Titration is an analytical technique used to determine the concentration of an unknown solution. To carry out this analytical method, the chemist would measure accurately the volume of a solution of unknown concentration and react with a solution of known concentration. The volume of th ...
... Definition: Titration is an analytical technique used to determine the concentration of an unknown solution. To carry out this analytical method, the chemist would measure accurately the volume of a solution of unknown concentration and react with a solution of known concentration. The volume of th ...
Chemical Thermodynamics : Georg Duesberg
... 2) They undergo elastic collisions 3) They are large in number and are randomly distributed 4) They can be treated as points of mass (diameter<< mean free path) Chemical Thermodynamics : Georg Duesberg ...
... 2) They undergo elastic collisions 3) They are large in number and are randomly distributed 4) They can be treated as points of mass (diameter<< mean free path) Chemical Thermodynamics : Georg Duesberg ...
1. You should review balancing equations and identifying types of
... 1. You should review balancing equations and identifying types of reactions from the worksheets. In addition you should be able to write balanced chemical equations for reactions. Try to write, balance, and identify the types of the following reactions: a. the decomposition of ammonium nitrate to ni ...
... 1. You should review balancing equations and identifying types of reactions from the worksheets. In addition you should be able to write balanced chemical equations for reactions. Try to write, balance, and identify the types of the following reactions: a. the decomposition of ammonium nitrate to ni ...
File
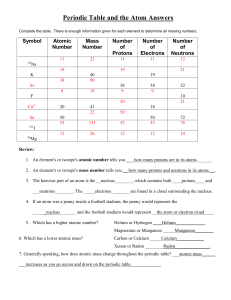
... All metals (on the left side of the periodic table) form cations and nonmetals (on the left side of the periodic table) form anions primarily. In order to determine the formula of the compound they create you must make sure their ions sum to zero. For example, table salt is sodium chloride. Using th ...
... All metals (on the left side of the periodic table) form cations and nonmetals (on the left side of the periodic table) form anions primarily. In order to determine the formula of the compound they create you must make sure their ions sum to zero. For example, table salt is sodium chloride. Using th ...
Fluorinated Butatrienes - diss.fu-berlin.de
... wurde ein Enin-Isomer entdeckt, das erstaunlicherweise stabiler als sein Butatrien Isomer ist, obwohl es an der Dreifachbindung fluoriert ist. Eben jene Fluorierung an der Dreifachbindung ist eigentlich notwendig um die Energie fluorierter But-1-en-3-ine relativ ...
... wurde ein Enin-Isomer entdeckt, das erstaunlicherweise stabiler als sein Butatrien Isomer ist, obwohl es an der Dreifachbindung fluoriert ist. Eben jene Fluorierung an der Dreifachbindung ist eigentlich notwendig um die Energie fluorierter But-1-en-3-ine relativ ...
wahideh chemistry eportfolio hw
... isotopes of sodium with measured half lives are also known. Two radioactive isotopes of sodium—sodium-22 and sodium-24— are used in medicine and other applications. They can be used as tracers to follow sodium in a person’s body. A tracer is a radioactive isotope whose presence in a system can easil ...
... isotopes of sodium with measured half lives are also known. Two radioactive isotopes of sodium—sodium-22 and sodium-24— are used in medicine and other applications. They can be used as tracers to follow sodium in a person’s body. A tracer is a radioactive isotope whose presence in a system can easil ...
Chemical Equations
... A reactant or product in the solid state Alternative to (s); used only for a precipitate (solid) falling out of solution A reactant or product in the liquid state A reactant or product in aqueous solution (dissolved in water) A reactant or product in the gaseous state Alternative to (g); used only f ...
... A reactant or product in the solid state Alternative to (s); used only for a precipitate (solid) falling out of solution A reactant or product in the liquid state A reactant or product in aqueous solution (dissolved in water) A reactant or product in the gaseous state Alternative to (g); used only f ...
SODIUM HYDROGEN CARBONATE
... agents such as starch, and weak acids such as alum or tartaric acid. These weak acids react with sodium bicarbonate, releasing CO2 gas, which causes cake batter and bread dough to rise and produces the tiny holes in cakes and breads. Sodium hydrogen carbonate dehydrates when it is heated, forming so ...
... agents such as starch, and weak acids such as alum or tartaric acid. These weak acids react with sodium bicarbonate, releasing CO2 gas, which causes cake batter and bread dough to rise and produces the tiny holes in cakes and breads. Sodium hydrogen carbonate dehydrates when it is heated, forming so ...
Fluorochemical industry
The global market for chemicals from fluorine was about US$16 billion per year as of 2006. The industry was predicted to reach 2.6 million metric tons per year by 2015. The largest market is the United States. Western Europe is the second largest. Asia Pacific is the fastest growing region of production. China in particular has experienced significant growth as a fluorochemical market and is becoming a producer of them as well. Fluorite mining (the main source of fluorine) was estimated in 2003 to be a $550 million industry, extracting 4.5 million tons per year.Mined fluorite is separated into two main grades, with about equal production of each. Acidspar is at least 97% CaF2; metspar is much lower purity, 60–85%. (A small amount of the intermediate, ceramic, grade is also made.) Metspar is used almost exclusively for iron smelting. Acidspar is primarily converted to hydrofluoric acid (by reaction with sulfuric acid). The resultant HF is mostly used to produce organofluorides and synthetic cryolite.


![Homework Booklet [4,S]](http://s1.studyres.com/store/data/010355871_1-63c750e3d1b58eaaebbb3f5d45651c44-300x300.png)