Microsoft Word
... if either of the new compounds is insoluble (or slightly soluble) precipitation occurs. if both new compounds are insoluble, two precipitation reactions occurs if both new compounds are soluble, no precipitation occurs. ...
... if either of the new compounds is insoluble (or slightly soluble) precipitation occurs. if both new compounds are insoluble, two precipitation reactions occurs if both new compounds are soluble, no precipitation occurs. ...
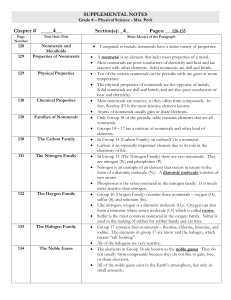
Chemistry of the Non
... Hydrogen has a 1s1 electron configuration so it is placed above Li in the periodic table. • However, H is significantly less reactive than the alkali metals. Hydrogen can gain an electron to form the hydride ion (H+1) which has a He electron configuration. • Therefore, H could be placed above the ha ...
... Hydrogen has a 1s1 electron configuration so it is placed above Li in the periodic table. • However, H is significantly less reactive than the alkali metals. Hydrogen can gain an electron to form the hydride ion (H+1) which has a He electron configuration. • Therefore, H could be placed above the ha ...
Chemical Changes and Structure Homework Booklet
... Draw the electron cloud diagrams to show how the electrons are shared in the following molecules. a. Hydrogen joined to carbon b. Nitrogen joined to chlorine c. Hydrogen joined to oxygen d. Nitrogen joined to nitrogen Hence, write the chemical formula for the molecules in Q.7 Describe how the covale ...
... Draw the electron cloud diagrams to show how the electrons are shared in the following molecules. a. Hydrogen joined to carbon b. Nitrogen joined to chlorine c. Hydrogen joined to oxygen d. Nitrogen joined to nitrogen Hence, write the chemical formula for the molecules in Q.7 Describe how the covale ...
SNC1D Periodic Table and Atomic Structure Package
... chemical symbols that we use today was first proposed by the Swedish chemist Jons Jakob Berzelius (1779-1848). Eventually this system was accepted all around the world. It was accepted not only because it provided symbols for all the known elements, but also because it showed how to create symbols ...
... chemical symbols that we use today was first proposed by the Swedish chemist Jons Jakob Berzelius (1779-1848). Eventually this system was accepted all around the world. It was accepted not only because it provided symbols for all the known elements, but also because it showed how to create symbols ...
1 - Montville.net
... 24. What does the law of definite composition say? All samples of a pure compound contain the same elements in the same proportion by mass 25. What does the law of multiple proportions say? Elements combine in small, whole number ratios to form pure compounds 26. An element “E” is present as 10E wit ...
... 24. What does the law of definite composition say? All samples of a pure compound contain the same elements in the same proportion by mass 25. What does the law of multiple proportions say? Elements combine in small, whole number ratios to form pure compounds 26. An element “E” is present as 10E wit ...
CHEM 120 WEEK 11 LECTURES (INORGANIC WEEK 2) Dr. MD
... atomic number. • Each halogen is the most electronegative element in its row. • Halogens exist as diatomic molecules. • In solids and liquids, the molecules are held together by weak London-dispersion forces • Iodine has the highest melting point and the strongest intermolecular forces. • At room te ...
... atomic number. • Each halogen is the most electronegative element in its row. • Halogens exist as diatomic molecules. • In solids and liquids, the molecules are held together by weak London-dispersion forces • Iodine has the highest melting point and the strongest intermolecular forces. • At room te ...
The Representative Elements: Group 5A Through 8A
... Important Trends in The Chemical Behaviors of the Group 5A Elements Elements of Group 5A overwhelmingly form covalent compounds. Whereas nitrogen can form a maximum of four covalent bonds, other elements in the group can form more than four covalent bonds by utilizing one or more of the nd orbitals. ...
... Important Trends in The Chemical Behaviors of the Group 5A Elements Elements of Group 5A overwhelmingly form covalent compounds. Whereas nitrogen can form a maximum of four covalent bonds, other elements in the group can form more than four covalent bonds by utilizing one or more of the nd orbitals. ...
National 5 Unit 1 Homework Booklet
... (c) Name the two pollutant gases changed by the catalyst and describe what they are changed into. 4. Explain why solid citric acid does not conduct electricity yet when it dissolves in water it does conduct. 5. Electrolysis of acids can be used to confirm the presence of hydrogen ions. (a) At which ...
... (c) Name the two pollutant gases changed by the catalyst and describe what they are changed into. 4. Explain why solid citric acid does not conduct electricity yet when it dissolves in water it does conduct. 5. Electrolysis of acids can be used to confirm the presence of hydrogen ions. (a) At which ...
Homework Booklet Unit 1 Feb14
... (c) Name the two pollutant gases changed by the catalyst and describe what they are changed into. 4. Explain why solid citric acid does not conduct electricity yet when it dissolves in water it does conduct. 5. Electrolysis of acids can be used to confirm the presence of hydrogen ions. (a) At which ...
... (c) Name the two pollutant gases changed by the catalyst and describe what they are changed into. 4. Explain why solid citric acid does not conduct electricity yet when it dissolves in water it does conduct. 5. Electrolysis of acids can be used to confirm the presence of hydrogen ions. (a) At which ...
Name - TeacherWeb
... The elements in Group 18 are known as the noble gases. They do not usually form compounds because they do not like to gain, lose, or share electrons. All of the noble gases exist in the Earth’s atmosphere, but only in small amounts. ...
... The elements in Group 18 are known as the noble gases. They do not usually form compounds because they do not like to gain, lose, or share electrons. All of the noble gases exist in the Earth’s atmosphere, but only in small amounts. ...
IT IS ELEMENTARY - the OLLI at UCI Blog
... and animal origin • These elements or their very simple compounds can kill—most commonly by interfering with cellular access to oxygen • Nitrogen N2 • Carbon dioxide CO2 • Carbon monoxide CO • Hydrogen cyanide HCN ...
... and animal origin • These elements or their very simple compounds can kill—most commonly by interfering with cellular access to oxygen • Nitrogen N2 • Carbon dioxide CO2 • Carbon monoxide CO • Hydrogen cyanide HCN ...
AP Chemistry - School Webmasters
... 53. To prevent a condition called the “bends”, deep sea divers breathe a mixture containing, ...
... 53. To prevent a condition called the “bends”, deep sea divers breathe a mixture containing, ...
Chapter 20 – The Representative Elements
... A complex series of reactions occur between NO2, O3, and unburned hydrocarbons in gasoline fumes to form peroxyacylnitrates (PANs), a group of atmospheric pollutants responsible for the brown smog and potent nose and eye irritants. Oxo-acids and Oxo-anions of Nitrogen Nitric acid (HNO3) and nitrous ...
... A complex series of reactions occur between NO2, O3, and unburned hydrocarbons in gasoline fumes to form peroxyacylnitrates (PANs), a group of atmospheric pollutants responsible for the brown smog and potent nose and eye irritants. Oxo-acids and Oxo-anions of Nitrogen Nitric acid (HNO3) and nitrous ...
inorganic chemistry
... organisms in sufficient quantities. This high reactivity is due to the atoms being highly electronegative due to their high effective nuclear charge. They can gain an electron by reacting with atoms of other elements. Fluorine is one of the most reactive elements in existence, attacking otherwise in ...
... organisms in sufficient quantities. This high reactivity is due to the atoms being highly electronegative due to their high effective nuclear charge. They can gain an electron by reacting with atoms of other elements. Fluorine is one of the most reactive elements in existence, attacking otherwise in ...
Fluorine in medicine (PDF Available)
... molecules exercise an influence on their properties and activities. The replacement of a hydrogen atom and/or a hydroxyl group by a fluorine atom are common strategies in drug development (Alkorta et al. 2000). Fluorine substitution in a drug molecule can influence not only pharmacokinetic propertie ...
... molecules exercise an influence on their properties and activities. The replacement of a hydrogen atom and/or a hydroxyl group by a fluorine atom are common strategies in drug development (Alkorta et al. 2000). Fluorine substitution in a drug molecule can influence not only pharmacokinetic propertie ...
AP Chemistry Summer Assignment
... For those students who have just taken Chemistry 1, much of the material in the summer packet will be familiar to you. For those students who have not taken Chemistry for a while the problems will help you rebuild a foundation in chemistry and insure all students are on a relatively even plane. It w ...
... For those students who have just taken Chemistry 1, much of the material in the summer packet will be familiar to you. For those students who have not taken Chemistry for a while the problems will help you rebuild a foundation in chemistry and insure all students are on a relatively even plane. It w ...
Ionic Bonding
... 13. How many elements are there in a tertiary compound? 14. Use each of the following terms correctly in a sentence about the formation of compounds: (a) polyatomic ion (b) oxyanion (c) hydrate 15. Write the IUPAC name for each of the following ionic compounds: (a) NaNO3(s) (found in tobacco) (b) Na ...
... 13. How many elements are there in a tertiary compound? 14. Use each of the following terms correctly in a sentence about the formation of compounds: (a) polyatomic ion (b) oxyanion (c) hydrate 15. Write the IUPAC name for each of the following ionic compounds: (a) NaNO3(s) (found in tobacco) (b) Na ...
Answers
... (c) Lithium would float on water, [1] producing gas steadily. [1] (d) Potassium would melt to a silvery ball [1] which moves about very quickly on the water surface, [1] producing a hissing sound, [1] burning spontaneously with a lilac flame [1] before finally disappearing completely. [1] (e) It wou ...
... (c) Lithium would float on water, [1] producing gas steadily. [1] (d) Potassium would melt to a silvery ball [1] which moves about very quickly on the water surface, [1] producing a hissing sound, [1] burning spontaneously with a lilac flame [1] before finally disappearing completely. [1] (e) It wou ...
High temperature semiconductor sensor for the detection of fluorine
... Keywords: Fluorine; Lanthanum fluoride; Silicon carbide ...
... Keywords: Fluorine; Lanthanum fluoride; Silicon carbide ...
Biosteres and You
... encased in a network of water molecules, if the cluster around the molecule is more compact it is more favourable and therefore the compound is more soluble. ...
... encased in a network of water molecules, if the cluster around the molecule is more compact it is more favourable and therefore the compound is more soluble. ...
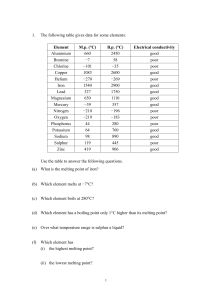
Key to Review Questions - Dixie State University
... Each of the following statements is FALSE. Correct it so that it is true. A mole of atoms is a certain mass of atoms. A mole of atoms is a certain number of atoms. All boron atoms have a mass of 10.81 Daltons. The weighted average of the naturally occurring isotopes of boron is 10.81 Daltons. All of ...
... Each of the following statements is FALSE. Correct it so that it is true. A mole of atoms is a certain mass of atoms. A mole of atoms is a certain number of atoms. All boron atoms have a mass of 10.81 Daltons. The weighted average of the naturally occurring isotopes of boron is 10.81 Daltons. All of ...
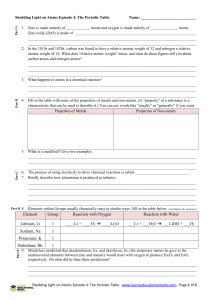
Element Group Reaction with Oxygen Reaction with Water Lithium
... atomic weight of 14. What does “relative atomic weight” mean, and what do these figures tell you about carbon atoms and nitrogen atoms? ______________________________________________________________________________________ _____________________________________________________________________________ ...
... atomic weight of 14. What does “relative atomic weight” mean, and what do these figures tell you about carbon atoms and nitrogen atoms? ______________________________________________________________________________________ _____________________________________________________________________________ ...
Nitrogen`s oxidation states
... and then a to a deep red when stored under water for prolonged periods of time. The color change corresponds to a slow conversion to a more stable allotropic form, red phosphorus. Pure red phosphorus is polymeric material with a structure consisting of joined tetrahedra in which one of the six P—P b ...
... and then a to a deep red when stored under water for prolonged periods of time. The color change corresponds to a slow conversion to a more stable allotropic form, red phosphorus. Pure red phosphorus is polymeric material with a structure consisting of joined tetrahedra in which one of the six P—P b ...
Fluorine
Fluorine is a chemical element with symbol F and atomic number 9. It is the lightest halogen and exists as a highly toxic pale yellow diatomic gas at standard conditions. As the most electronegative element, it is extremely reactive: almost all other elements, including some noble gases, form compounds with fluorine.Among the elements, fluorine ranks 24th in universal abundance and 13th in terrestrial abundance. Fluorite, the primary mineral source of fluorine, was first described in 1529; as it was added to metal ores to lower their melting points for smelting, the Latin verb fluo meaning ""flow"" became associated with it. Proposed as an element in 1810, fluorine proved difficult and dangerous to separate from its compounds, and several early experimenters died or sustained injuries from their attempts. Only in 1886 did French chemist Henri Moissan isolate elemental fluorine using low-temperature electrolysis, a process still employed for modern production. Industrial synthesis of fluorine gas for uranium enrichment, its largest application, began during the Manhattan Project in World War II.Owing to the expense of refining pure fluorine, most commercial applications of the element involve the use of its compounds, with about half of mined fluorite used in steelmaking. The rest is converted into corrosive hydrogen fluoride en route to various organic fluorides, or into cryolite which plays a key role in aluminium refining. Organic fluorides have very high chemical and thermal stability; their major uses are as refrigerants, electrical insulation and cookware, the last as PTFE (Teflon). Pharmaceuticals such as atorvastatin and fluoxetine also contain fluorine, and the fluoride ion inhibits dental cavities, and so finds use in toothpaste and water fluoridation. Global fluorochemical sales amount to over US$15 billion a year.Fluorocarbon gases are generally greenhouse gases with global-warming potentials 100 to 20,000 times that of carbon dioxide. Organofluorine compounds persist in the environment due to the strength of the carbon–fluorine bond. Fluorine has no known metabolic role in mammals; a few plants synthesize organofluorine poisons which deter herbivores.