Periodic Table
... ability to gain or lose eIV. Chemical Families and their Characteristics – A. Alkali Metals (Group 1, or IA) ...
... ability to gain or lose eIV. Chemical Families and their Characteristics – A. Alkali Metals (Group 1, or IA) ...
Single Replacement Reactions
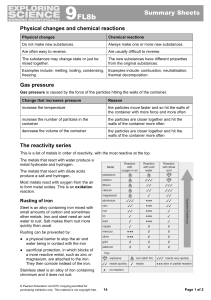
... In nature, elements can occur either free, meaning uncombined with other elements, or chemically combined in a compound. The tendency of a particular element to combine with other substances is a measure of the activity of the element. The more active an element is, the more likely it is to combine. ...
... In nature, elements can occur either free, meaning uncombined with other elements, or chemically combined in a compound. The tendency of a particular element to combine with other substances is a measure of the activity of the element. The more active an element is, the more likely it is to combine. ...
File
... Lanthanide series are part of the inner transition elements. 6. Actinide series- transition metals that follow the element actinium in Period 7 of the periodic table; all actinides are radioactive. Actinide series are part of the inner transition. 7. Noble gases- any of the gaseous elements helium, ...
... Lanthanide series are part of the inner transition elements. 6. Actinide series- transition metals that follow the element actinium in Period 7 of the periodic table; all actinides are radioactive. Actinide series are part of the inner transition. 7. Noble gases- any of the gaseous elements helium, ...
Metals, Nonmetals, and Metalloids (Vocabulary)
... are arranged by properties and are represented by one or two letter chemical symbols. ...
... are arranged by properties and are represented by one or two letter chemical symbols. ...
The subject of " Engineering Materials " deals with the study of
... All metals are capable of thermionic emission ( the emission of electrons when being heated , they are good reflectors of light and lend them selves to plastic deformation . Pure metals are of low string the and do not possess the required physiochemical and technological properties for some definit ...
... All metals are capable of thermionic emission ( the emission of electrons when being heated , they are good reflectors of light and lend them selves to plastic deformation . Pure metals are of low string the and do not possess the required physiochemical and technological properties for some definit ...
9F Reactivity - Parrs Wood High School
... Steel is an alloy containing iron mixed with small amounts of carbon and sometimes other metals. Iron and steel need air and water to rust. Salt makes them rust more quickly than usual. Rusting can be prevented by: ● a physical barrier to stop the air and water being in contact with the iron ● sacri ...
... Steel is an alloy containing iron mixed with small amounts of carbon and sometimes other metals. Iron and steel need air and water to rust. Salt makes them rust more quickly than usual. Rusting can be prevented by: ● a physical barrier to stop the air and water being in contact with the iron ● sacri ...
Power point on the Periodic Table
... Number of protons that an atom of any element has is called the atomic number The sum of the protons and neutrons is called the mass number Protons and neutrons make up nearly all the mass of an atom ...
... Number of protons that an atom of any element has is called the atomic number The sum of the protons and neutrons is called the mass number Protons and neutrons make up nearly all the mass of an atom ...
lecture_CH1-2review_chem121pikul
... • Brittle, dull • Insulators, nonconductors of electricity and heat • Chemical reactivity varies • Exist mostly as compounds rather then pure elements • Many are gases, some are solids at room temp, only Br2 is a liquid. ...
... • Brittle, dull • Insulators, nonconductors of electricity and heat • Chemical reactivity varies • Exist mostly as compounds rather then pure elements • Many are gases, some are solids at room temp, only Br2 is a liquid. ...