69. A general approach to the enantioselective -oxidation of aldehydes via synergistic catalysis
... strategies continue to expand the range of starting materials or functional groups from which this important stereogenicity can be created. Recently, the enantioselective a-oxidation of aldehydes has garnered substantial attention as a novel catalytic approach to asymmetric oxygen-bearing stereocent ...
... strategies continue to expand the range of starting materials or functional groups from which this important stereogenicity can be created. Recently, the enantioselective a-oxidation of aldehydes has garnered substantial attention as a novel catalytic approach to asymmetric oxygen-bearing stereocent ...
13-4 Ligands in Organometallic Chemistry
... Organometallic chemistry is the study of chemical compounds containing bonds between carbon and a metal. Organometallic chemistry combines aspects of inorganic chemistry and organic chemistry. Organometallic compounds find practical use in stoichiometric and catalytically active compounds. Electron ...
... Organometallic chemistry is the study of chemical compounds containing bonds between carbon and a metal. Organometallic chemistry combines aspects of inorganic chemistry and organic chemistry. Organometallic compounds find practical use in stoichiometric and catalytically active compounds. Electron ...
Asymmetric Synthesis: Substrate and Auxiliary Control
... ▪ Enantiomers have identical physical properties (assuming they are in an achiral environment), e.g. melting/boiling point, IR and NMR spectra, density, viscosity, etc. ▪ Diastereo(iso)mers have different physical properties. diastereoisomers (in red) ...
... ▪ Enantiomers have identical physical properties (assuming they are in an achiral environment), e.g. melting/boiling point, IR and NMR spectra, density, viscosity, etc. ▪ Diastereo(iso)mers have different physical properties. diastereoisomers (in red) ...
Efficient hydrogenation of organic carbonates, carbamates and
... blocks such as alcohols and amines. Much progress has been made in the hydrogenation of ketones and aldehydes and, more recently, rare examples of the significantly more difficult hydrogenation of esters4,5 and amides6,7 have also been reported. However, the hydrogenation of organic carbonates and car ...
... blocks such as alcohols and amines. Much progress has been made in the hydrogenation of ketones and aldehydes and, more recently, rare examples of the significantly more difficult hydrogenation of esters4,5 and amides6,7 have also been reported. However, the hydrogenation of organic carbonates and car ...
optical isomerism
... Synthesis of 2-hydroxypropanoic acid (lactic acid) LACTIC ACID can be formed from ethanal in a two stage process. 1. Nucleophilic addition of hydrogen cyanide to ethanal 2 Hydrolysis of the nitrile group ...
... Synthesis of 2-hydroxypropanoic acid (lactic acid) LACTIC ACID can be formed from ethanal in a two stage process. 1. Nucleophilic addition of hydrogen cyanide to ethanal 2 Hydrolysis of the nitrile group ...
Reaction Mechanisms
... Eliminations are the reverse reaction of migratory insertion and can occur one after the other. The group being eliminated does not have to be the one that participated in the insertion. There are several types of eliminations. α-Hydride Elimination (AHE) Elimination of an α-hydrogen from metal alky ...
... Eliminations are the reverse reaction of migratory insertion and can occur one after the other. The group being eliminated does not have to be the one that participated in the insertion. There are several types of eliminations. α-Hydride Elimination (AHE) Elimination of an α-hydrogen from metal alky ...
Hydrogenation for Low Trans and High Conjugated Fatty Acids
... isomerizes the natural cis double bonds to trans double bonds during hydrogenation. The trans fatty acids of hydrogenated vegetable oil by precious metal catalysts were lower than those of hydrogenated oil by nickel catalysts. Nickel catalysts are not very active below 120 °C. Precious metal catalys ...
... isomerizes the natural cis double bonds to trans double bonds during hydrogenation. The trans fatty acids of hydrogenated vegetable oil by precious metal catalysts were lower than those of hydrogenated oil by nickel catalysts. Nickel catalysts are not very active below 120 °C. Precious metal catalys ...
Recent developments in the applications of palladium complexes
... The structural features and catalytic applications of metal complexes bearing polyNHCs have also been reviewed.19-23 Some excellent reviews have appeared dealing with the design of chiral-NHC systems and their applications in asymmetric catalysis.24-27 Moreover, monographs28, 29 and special issues30 ...
... The structural features and catalytic applications of metal complexes bearing polyNHCs have also been reviewed.19-23 Some excellent reviews have appeared dealing with the design of chiral-NHC systems and their applications in asymmetric catalysis.24-27 Moreover, monographs28, 29 and special issues30 ...
Chapter 1 Organoaluminum Reagents for Selective Organic
... In chapter 2 we discussed several excellent methods of discriminating various functional groups using bulky aluminum reagents. In this section we focus on the reactions promoted with bulky aluminum reagents which could not be achieved with ordinary Lewis acid catalysts. The following is a typical ex ...
... In chapter 2 we discussed several excellent methods of discriminating various functional groups using bulky aluminum reagents. In this section we focus on the reactions promoted with bulky aluminum reagents which could not be achieved with ordinary Lewis acid catalysts. The following is a typical ex ...
Module 2 Asymmetric Carbon-Carbon Bond Forming Reactions
... Joint initiative of IITs and IISc – Funded by MHRD ...
... Joint initiative of IITs and IISc – Funded by MHRD ...
Today`s literature presentation = 2/5th trivia + 2/5th
... Heavy metals used in most of theses cause problems in removal, must be present <10 ppm levels. Many oxidizing agents are high energy species, giving rise to thermal hazards at scale. ...
... Heavy metals used in most of theses cause problems in removal, must be present <10 ppm levels. Many oxidizing agents are high energy species, giving rise to thermal hazards at scale. ...
The influence of organic ligands on trace metal speciation
... dissolved trace metal. Fig. 1 shows the results for U(VI) complexation with oxalate, EDTA and humic substances as a function of pH and log pCO2. The latter quantity is the logarithm of the CO2 partial The 'backdoor approach' pressure the water being in equilibrium with. Surface In the past, it was t ...
... dissolved trace metal. Fig. 1 shows the results for U(VI) complexation with oxalate, EDTA and humic substances as a function of pH and log pCO2. The latter quantity is the logarithm of the CO2 partial The 'backdoor approach' pressure the water being in equilibrium with. Surface In the past, it was t ...
Organic - UCLA Chemistry and Biochemistry
... the characteristic apparent quartet (J = 12 Hz) for Ha that is also seen in the spectrum of Id.' Likewise the stereochemistry of 9b was determined primarily from its highfield 'H NMR spectrum which showed the expected coupling constants for the conformation drawn, namely: Ha dd, J = 11.8, 6.8 Hz;Hb ...
... the characteristic apparent quartet (J = 12 Hz) for Ha that is also seen in the spectrum of Id.' Likewise the stereochemistry of 9b was determined primarily from its highfield 'H NMR spectrum which showed the expected coupling constants for the conformation drawn, namely: Ha dd, J = 11.8, 6.8 Hz;Hb ...
Synopsis
... The thesis entitled, “Preparation, Application of Sulfilimines as Intramolecular Nucleophiles in the Synthesis of AHDA, AHPBA, (+)Desoxoprosophylline and (-)-Deoxocassine” is divided into three chapters. Chapter-I comprises two sections, ‘A’ and ‘B’. Section A concerns with a brief introduction to s ...
... The thesis entitled, “Preparation, Application of Sulfilimines as Intramolecular Nucleophiles in the Synthesis of AHDA, AHPBA, (+)Desoxoprosophylline and (-)-Deoxocassine” is divided into three chapters. Chapter-I comprises two sections, ‘A’ and ‘B’. Section A concerns with a brief introduction to s ...
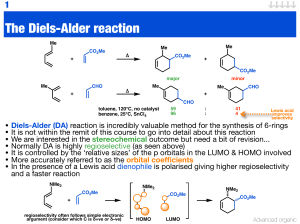
The Diels-Alder reaction
... • The reaction is related to much of Pd chemistry you have covered • Below is an example of a chiral variant of the Schrock metathesis catalyst • The reaction involves desymmetrisation by selective reaction if one disubstituted ...
... • The reaction is related to much of Pd chemistry you have covered • Below is an example of a chiral variant of the Schrock metathesis catalyst • The reaction involves desymmetrisation by selective reaction if one disubstituted ...
Enantioselective Synthesis of Cyclic Ethers through a Vanadium
... spite of the chiral ligand employed, the epoxidation/ringopening reaction gave racemic 2,5-cis-THF rings as the major product. In accord with this report, our own attempts to introduce enantioselectivity in this reaction by resolution of bishomoallylic alcohols by catalytic epoxidation[4] resulted i ...
... spite of the chiral ligand employed, the epoxidation/ringopening reaction gave racemic 2,5-cis-THF rings as the major product. In accord with this report, our own attempts to introduce enantioselectivity in this reaction by resolution of bishomoallylic alcohols by catalytic epoxidation[4] resulted i ...
Şenol, O.İ., Viljava, T.-R., Krause, AOI
... Methyl heptanoate and methyl hexanoate did not react at 300 8C without catalysts. The alumina support converted about 13% of the esters at 300 8C and the identified products included only the alcohols and the acids. Although the alumina support had some activity for deesterification and dehydration ...
... Methyl heptanoate and methyl hexanoate did not react at 300 8C without catalysts. The alumina support converted about 13% of the esters at 300 8C and the identified products included only the alcohols and the acids. Although the alumina support had some activity for deesterification and dehydration ...
ENZYME MIMIC ASYMMETRIC ALDOL REACTIONS
... The class I aldolases activate the ketone donor via enamine formation using active site lysine residue and concomitantly activate the aldehyde acceptor with an active site acidic residue. It would be promising if the catalyst simultaneously activates both the ketone donor and aldehyde acceptor. Amin ...
... The class I aldolases activate the ketone donor via enamine formation using active site lysine residue and concomitantly activate the aldehyde acceptor with an active site acidic residue. It would be promising if the catalyst simultaneously activates both the ketone donor and aldehyde acceptor. Amin ...
Catalysis for a Sustainable World
... Recent advances in modern organic synthesis allowed for an efficient access to a broad range of complex molecules in a highly stereocontrolled manner. The methodologies, however, usually require the stoichiometric use of activating reagents to achieve high reactivity and chemo/stereoselectivity, the ...
... Recent advances in modern organic synthesis allowed for an efficient access to a broad range of complex molecules in a highly stereocontrolled manner. The methodologies, however, usually require the stoichiometric use of activating reagents to achieve high reactivity and chemo/stereoselectivity, the ...
Pseudoasymmetry as a Means for Distinguishing Meso
... chirality is a useful complement to the use of pseudoasymmetry since chemical-shift nonequivalence points unequivocally to the dl isomer, while the creation of a pseudoasymmetric center provides unambiguous evidence of the meso isomer. Thus, the combination can be used to make unambiguous assignment ...
... chirality is a useful complement to the use of pseudoasymmetry since chemical-shift nonequivalence points unequivocally to the dl isomer, while the creation of a pseudoasymmetric center provides unambiguous evidence of the meso isomer. Thus, the combination can be used to make unambiguous assignment ...
Lecture 31 Homogeneous catalysis
... organometallic complex catalysts, the reaction begins with the reactant molecule getting attached to a metal centre in the catalyst. Hence, the metallic centers must have vacant coordination sites. However, it is difficult to maintain vacant sites on the metallic center as the molecules are always i ...
... organometallic complex catalysts, the reaction begins with the reactant molecule getting attached to a metal centre in the catalyst. Hence, the metallic centers must have vacant coordination sites. However, it is difficult to maintain vacant sites on the metallic center as the molecules are always i ...
Chapter 7 Hydrosilylation of Carbon
... with high regioselectivity to produce benzylic silanes, 1-aryl-1-silylethanes, due to the participation of π-benzylic palladium intermediates [1, 2]. It is known that bisphosphine-palladium complexes are catalytically much less active than monophosphine-palladium complexes and hence asymmetric synth ...
... with high regioselectivity to produce benzylic silanes, 1-aryl-1-silylethanes, due to the participation of π-benzylic palladium intermediates [1, 2]. It is known that bisphosphine-palladium complexes are catalytically much less active than monophosphine-palladium complexes and hence asymmetric synth ...
Lecture12
... because it is much less expensive than palladium. Nickel-based catalysts tend to be less active and general, however. Nickel is better at activating aryl chlorides than palladium in some cases, however. Platinum has shown no activity in crosscoupling chemistry. ...
... because it is much less expensive than palladium. Nickel-based catalysts tend to be less active and general, however. Nickel is better at activating aryl chlorides than palladium in some cases, however. Platinum has shown no activity in crosscoupling chemistry. ...
Asymmetric hydrogenation

Asymmetric hydrogenation is a chemical reaction that adds two atoms of hydrogen preferentially to one of two faces of an unsaturated substrate molecule, such as an alkene or ketone. The selectivity derives from the manner that the substrate binds to the chiral catalysts. In jargon, this binding transmits spatial information (what chemists refer to as chirality) from the catalyst to the target, favoring the product as a single enantiomer. This enzyme-like selectivity is particularly applied to bioactive products such as pharmaceutical agents and agrochemicals.