SCIENCE OF SUN PHOTOMETRY
... Bird, R., and C. Riordan (1994) Simple Solar Spectral Model for Direct and Diffuse Irradiance on Horizontal and Tilted Planes at the Earth’s Surface for Cloudless Atmosphere, Solar Energy Research Institute SERI/TR-215-2436. Brooks, D. R., and Forrest M. Mims III (2001) Development of an inexpensive ...
... Bird, R., and C. Riordan (1994) Simple Solar Spectral Model for Direct and Diffuse Irradiance on Horizontal and Tilted Planes at the Earth’s Surface for Cloudless Atmosphere, Solar Energy Research Institute SERI/TR-215-2436. Brooks, D. R., and Forrest M. Mims III (2001) Development of an inexpensive ...
Meteorology Frameworks Kindergarten Students know
... The atmosphere and surface of Earth are heated unevenly, giving rise to both local and global temperature differences. For example, the direct heat absorbed by the surface of the ocean, land, and air may result in different temperatures. Furthermore, the amount of heat varies with latitude, primaril ...
... The atmosphere and surface of Earth are heated unevenly, giving rise to both local and global temperature differences. For example, the direct heat absorbed by the surface of the ocean, land, and air may result in different temperatures. Furthermore, the amount of heat varies with latitude, primaril ...
Lectures 5
... Below what pH will precipitation occur given that solubility is 4.78X10-3 M and pka is 7.48 and mol. Weight is 254 gm/mol. [HA]T is 1gm/100ml convert to molar expression =10gm/1000ml number of mole = weight / mol.weight ...
... Below what pH will precipitation occur given that solubility is 4.78X10-3 M and pka is 7.48 and mol. Weight is 254 gm/mol. [HA]T is 1gm/100ml convert to molar expression =10gm/1000ml number of mole = weight / mol.weight ...
Fractional Distillation
... important to keep in mind the entire column is in dynamic equilibrium and it is not possible to identify an actual “plate” associated with a theoretical plate. Finally, very few liquid mixtures are truly Ideal. Some exhibit a positive deviation from Raoult’s Law and others a negative deviation. Acet ...
... important to keep in mind the entire column is in dynamic equilibrium and it is not possible to identify an actual “plate” associated with a theoretical plate. Finally, very few liquid mixtures are truly Ideal. Some exhibit a positive deviation from Raoult’s Law and others a negative deviation. Acet ...
Stability and Moisture lectures
... • For saturated air, the effects of latent heat release when water changes phase are important. • Consider an air parcel, rising adiabatically: • Condensation takes place and latent heat is released: the (saturated) adiabatic lapse rate is lower than the dry adiabatic lapse rate, i.e. Γs < Γd. ...
... • For saturated air, the effects of latent heat release when water changes phase are important. • Consider an air parcel, rising adiabatically: • Condensation takes place and latent heat is released: the (saturated) adiabatic lapse rate is lower than the dry adiabatic lapse rate, i.e. Γs < Γd. ...
Meteorology - The Federation of Galaxy Explorers
... deal with fronts anyway? Well that is where the bad weather is. A cold front is associated with showers and thunder storms. Warm fronts usually bring steady rain. You can see that the clouds lie along the fronts on this weather map. Of course, the weather forecasters have a lot more information abo ...
... deal with fronts anyway? Well that is where the bad weather is. A cold front is associated with showers and thunder storms. Warm fronts usually bring steady rain. You can see that the clouds lie along the fronts on this weather map. Of course, the weather forecasters have a lot more information abo ...
UNIT 5_THE ATMOSPHERE
... How are the clouds formed?: In the areas heated by the Sun, the water evaporates and goes up to the troposphere. In the high part of the troposphere, the water vapour cools down. The cold vapor is condensed in small drops. Those drops form the clouds. There are three basic types of clouds: cirrus, c ...
... How are the clouds formed?: In the areas heated by the Sun, the water evaporates and goes up to the troposphere. In the high part of the troposphere, the water vapour cools down. The cold vapor is condensed in small drops. Those drops form the clouds. There are three basic types of clouds: cirrus, c ...
General Chemistry
... Cg is the solubility of gas, Pg the partial pressure, k = Henry’s law constant. •Carbonated beverages are bottled under PCO2 > 1 atm. As the bottle is opened, PCO2 decreases and the solubility of CO2 decreases. Therefore, bubbles of CO2 escape from solution. ...
... Cg is the solubility of gas, Pg the partial pressure, k = Henry’s law constant. •Carbonated beverages are bottled under PCO2 > 1 atm. As the bottle is opened, PCO2 decreases and the solubility of CO2 decreases. Therefore, bubbles of CO2 escape from solution. ...
equilibrium theory of the kaolinite
... for those on Li+-H~O, are in good q u a l i t a t i v e a g r e e m e n t w i t h t h e conclusions of M a r t i n (1959) a n d J u r i n a k (1963), who s t u d i e d t h e a d s o r p t i o n o f w a t e r v a p o r b y homoionic kaolinite. The e x p e r i m e n t a l l y o b s e r v e d behavior, ...
... for those on Li+-H~O, are in good q u a l i t a t i v e a g r e e m e n t w i t h t h e conclusions of M a r t i n (1959) a n d J u r i n a k (1963), who s t u d i e d t h e a d s o r p t i o n o f w a t e r v a p o r b y homoionic kaolinite. The e x p e r i m e n t a l l y o b s e r v e d behavior, ...
AP Lab - MW of Volatile Liquid - North Allegheny School District
... If a substance behaves as an ideal gas, we can easily calculate its molar mass if we measure the mass m of a volume V of the pure gas at known temperature T and pressure P. The ideal gas law is PV = nRT, where n = moles of gas. But n = m/Mw where Mw equals molar mass. Therefore, Mw = mRT/PV. This me ...
... If a substance behaves as an ideal gas, we can easily calculate its molar mass if we measure the mass m of a volume V of the pure gas at known temperature T and pressure P. The ideal gas law is PV = nRT, where n = moles of gas. But n = m/Mw where Mw equals molar mass. Therefore, Mw = mRT/PV. This me ...
What is meteorology?
... Pressure variations at sea level do not usually exceed 4% of the normal average value (that is 1013 millibar): lower values (up to 900 millibar) can be registered in the eye of tropical cyclones. With the exception of some small local variations, atmospheric pressure and density decrease with altitu ...
... Pressure variations at sea level do not usually exceed 4% of the normal average value (that is 1013 millibar): lower values (up to 900 millibar) can be registered in the eye of tropical cyclones. With the exception of some small local variations, atmospheric pressure and density decrease with altitu ...
Meteorology notes
... the average weather over a long period of time. It generally stays the same from year to year. Climate is described in terms of the average temperature of a region or the average amount of rain or snow a region receives. Climate ...
... the average weather over a long period of time. It generally stays the same from year to year. Climate is described in terms of the average temperature of a region or the average amount of rain or snow a region receives. Climate ...
10 Vapor Pressure - Blue Valley Schools
... above it in the flask. If the stopper is left open, eventually all of the air will be displaced by methanol vapor. Once sealed, equilibrium will be reached between the rate of evaporation and the rate of condensation. At this point, the vapor pressure of the liquid is equal to the partial pressure o ...
... above it in the flask. If the stopper is left open, eventually all of the air will be displaced by methanol vapor. Once sealed, equilibrium will be reached between the rate of evaporation and the rate of condensation. At this point, the vapor pressure of the liquid is equal to the partial pressure o ...
Solutions!
... Shows the relationship of grams of solute that may be dissolved at various temperatures. ...
... Shows the relationship of grams of solute that may be dissolved at various temperatures. ...
Material and Energy Balances CHEN 2120 Outline Specific
... • “Designed” to take into account ‘PV’ work for open systems • Example: If the specific internal energy of a gas at 300 K and 1 atm is 4200 J/mol, and the specific molar volume at the same temperature and pressure is 25.34 L/mol, the specific enthalpy of the gas is: ...
... • “Designed” to take into account ‘PV’ work for open systems • Example: If the specific internal energy of a gas at 300 K and 1 atm is 4200 J/mol, and the specific molar volume at the same temperature and pressure is 25.34 L/mol, the specific enthalpy of the gas is: ...
FREE Sample Here
... played by water vapor, which is a source of precipitation and latent heat energy as well as being the most important greenhouse gas, are given particular attention. Current concern over increasing concentrations of another constituent, carbon dioxide, and its possible effect on global climate are al ...
... played by water vapor, which is a source of precipitation and latent heat energy as well as being the most important greenhouse gas, are given particular attention. Current concern over increasing concentrations of another constituent, carbon dioxide, and its possible effect on global climate are al ...
Classical Thermodynamics I: Sublimation of Solid Iodine
... The test tube containing iodine in the solid and vapor states must also contain air or nitrogen at about 1 atm to provide pressure broadening of the extremely sharp and intense absorption lines of the rotational fine structure (which can be individually resolved only by special techniques of laser s ...
... The test tube containing iodine in the solid and vapor states must also contain air or nitrogen at about 1 atm to provide pressure broadening of the extremely sharp and intense absorption lines of the rotational fine structure (which can be individually resolved only by special techniques of laser s ...
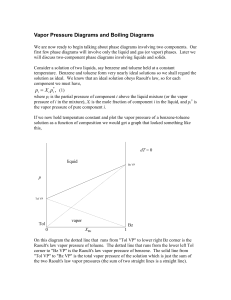
Vapor Pressure Diagrams and Boiling Diagrams
... As we heat the liquid it will begin to boil when the temperature reaches the temperature of point "a." The first vapor to come off has the composition shown at point "b." Capture the vapor, condense it, and heat it up. The new liquid will boil at point "c" giving a vapor with composition at point "d ...
... As we heat the liquid it will begin to boil when the temperature reaches the temperature of point "a." The first vapor to come off has the composition shown at point "b." Capture the vapor, condense it, and heat it up. The new liquid will boil at point "c" giving a vapor with composition at point "d ...
Review of heat and water movement in field soils
... there has never been a satisfactory comparison of water vapor ¯uxes measured in the ®eld with theory. At least two factors have led to this, ®rst, most of the experimental work has been laboratory oriented with steady state boundary conditions imposed and second, there have been relatively few ®eld ...
... there has never been a satisfactory comparison of water vapor ¯uxes measured in the ®eld with theory. At least two factors have led to this, ®rst, most of the experimental work has been laboratory oriented with steady state boundary conditions imposed and second, there have been relatively few ®eld ...
Lecture Packet#1
... 2. Vertical structure of atmospheric pressure & temperature 3. Types of weather & climate in the atmosphere ...
... 2. Vertical structure of atmospheric pressure & temperature 3. Types of weather & climate in the atmosphere ...
notes09
... Physically, the concept of partial pressure means that each gas component acts as if it were the only gas in the container. Even though air is made up of N2, O2, Ar, CO2, etc, for engineering purposes, we model air as a single component when dealing with humid air calculations. Accordingly, consider ...
... Physically, the concept of partial pressure means that each gas component acts as if it were the only gas in the container. Even though air is made up of N2, O2, Ar, CO2, etc, for engineering purposes, we model air as a single component when dealing with humid air calculations. Accordingly, consider ...
Intro to the Atmosphere
... The Stratosphere and Ozone Layer Above the troposphere is the stratosphere, where air flow is mostly horizontal. The thin ozone layer in the upper stratosphere has a high concentration of ozone, a particularly reactive form of oxygen. This layer is primarily responsible for absorbing the ultraviole ...
... The Stratosphere and Ozone Layer Above the troposphere is the stratosphere, where air flow is mostly horizontal. The thin ozone layer in the upper stratosphere has a high concentration of ozone, a particularly reactive form of oxygen. This layer is primarily responsible for absorbing the ultraviole ...
Water vapor

Water vapor, or water vapour or aqueous vapor, is the gaseous phase of water. It is one state of water within the hydrosphere. Water vapor can be produced from the evaporation or boiling of liquid water or from the sublimation of ice. Unlike other forms of water, water vapor is invisible. Under typical atmospheric conditions, water vapor is continuously generated by evaporation and removed by condensation. It is lighter than air and triggers convection currents that can lead to clouds.Water vapor is a relatively common atmospheric constituent, present even in the solar atmosphere as well as every planet in the Solar System and many astronomical objects including natural satellites, comets and even large asteroids. Likewise the detection of extrasolar water vapor would indicate a similar distribution in other planetary systems. Water vapor is significant in that it can be indirect evidence supporting the presence of extraterrestrial liquid water in the case of some planetary mass objects.Being a component of Earth's hydrosphere and hydrologic cycle, it is particularly abundant in Earth's atmosphere where it is also a potent greenhouse gas along with other gases such as carbon dioxide and methane. Use of water vapor, as steam, has been important to humans for cooking and as a major component in energy production and transport systems since the industrial revolution.