Effects of surface charge density and distribution on the
... 4.66×4.22×3.49 nm. Channel walls perpendicular to the z axis were formed by four [111] oriented layers of Si atoms in a diamond crystal structure, each wall being 0.39 nm thick. Periodic boundary conditions were applied in the x and y directions. The size of the simulation cell in the z direction wa ...
... 4.66×4.22×3.49 nm. Channel walls perpendicular to the z axis were formed by four [111] oriented layers of Si atoms in a diamond crystal structure, each wall being 0.39 nm thick. Periodic boundary conditions were applied in the x and y directions. The size of the simulation cell in the z direction wa ...
Sahand University of Technology
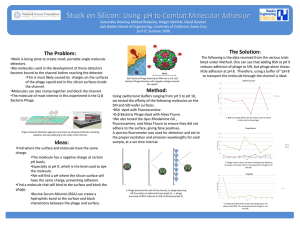
... 1. It must wet the surfaces, that is it must spread and make a contact angle approaching zero. Intimate contact is required between the molecules of the adhesive and the atoms and molecules in the surface. When applied the adhesive will be a liquid of relatively low viscosity. 2. The adhesive must t ...
... 1. It must wet the surfaces, that is it must spread and make a contact angle approaching zero. Intimate contact is required between the molecules of the adhesive and the atoms and molecules in the surface. When applied the adhesive will be a liquid of relatively low viscosity. 2. The adhesive must t ...
Surface tension
Surface tension is the elastic tendency of liquids which makes them acquire the least surface area possible. Surface tension is an important property that markedly influences many ecosystems. Surface tension is responsible, for example, when an object or insect (e.g. water striders) that is denser than water is able to float or run along the water surface.At liquid-air interfaces, surface tension results from the greater attraction of water molecules to each other (due to cohesion) than to the molecules in the air (due to adhesion). The net effect is an inward force at its surface that causes water to behave as if its surface were covered with a stretched elastic membrane. Because of the relatively high attraction of water molecules for each other, water has a high surface tension (72.8 millinewtons per meter at 20 °C) compared to that of most other liquids. Surface tension is an important factor in the phenomenon of capillarity.Surface tension has the dimension of force per unit length, or of energy per unit area. The two are equivalent—but when referring to energy per unit of area, people use the term surface energy—which is a more general term in the sense that it applies also to solids and not just liquids.In materials science, surface tension is used for either surface stress or surface free energy.