Topic 1222 Equation of State: Real Gases: van der Waals and Other
... temperature. If the plot of p ⋅ Vj against p passes through zero at temperature TB , then TB is given by equation (y) [17]. ...
... temperature. If the plot of p ⋅ Vj against p passes through zero at temperature TB , then TB is given by equation (y) [17]. ...
VI. ELECTRONIC SPECTROSCOPY The visible (V) and ultraviolet
... 1801 (Thomas Young): discovery of optical interference (“Whenever two portions of the same light arrive to the eye by different routes, either exactly or very nearly in the same direction, the light becomes most intense when the difference of the two routes is any multiple of a certain length, and l ...
... 1801 (Thomas Young): discovery of optical interference (“Whenever two portions of the same light arrive to the eye by different routes, either exactly or very nearly in the same direction, the light becomes most intense when the difference of the two routes is any multiple of a certain length, and l ...
A completely transparent, adhesively bonded soda-lime
... of solid cast glass blocks that intermix with normal bricks towards the second floor of the building. More specific, the lower ten meters of the facade comprise mainly cast glass blocks. Only at the highest part of this area, the glass blocks intermix with conventional clay bricks in a limited zone un ...
... of solid cast glass blocks that intermix with normal bricks towards the second floor of the building. More specific, the lower ten meters of the facade comprise mainly cast glass blocks. Only at the highest part of this area, the glass blocks intermix with conventional clay bricks in a limited zone un ...
1 Enzymes – Enzyme Mechanism
... provides substrate specificity and catalytic power • Two catalytic modes based on binding properties can each increase reaction rates over 10,000-fold : (1) Proximity effect - collecting and positioning substrate molecules in the active site ...
... provides substrate specificity and catalytic power • Two catalytic modes based on binding properties can each increase reaction rates over 10,000-fold : (1) Proximity effect - collecting and positioning substrate molecules in the active site ...
Enzymes – Enzyme Mechanism
... provides substrate specificity and catalytic power • Two catalytic modes based on binding properties can each increase reaction rates over 10,000-fold : (1) Proximity effect - collecting and positioning substrate molecules in the active site (2) Transition-state (TS) stabilization - transition state ...
... provides substrate specificity and catalytic power • Two catalytic modes based on binding properties can each increase reaction rates over 10,000-fold : (1) Proximity effect - collecting and positioning substrate molecules in the active site (2) Transition-state (TS) stabilization - transition state ...
Theoretical Inversion of Amino Acids (Alanine and Aspartic Acid) by
... Racemization is a chemical reaction and a number of factors influence its rate(5). These include; amino acid structure, the sequence of amino acids in peptides, pH, buffering effects, metallic cations, the presence of water and temperature. To establish a dating method the kinetics and mechanisms of ...
... Racemization is a chemical reaction and a number of factors influence its rate(5). These include; amino acid structure, the sequence of amino acids in peptides, pH, buffering effects, metallic cations, the presence of water and temperature. To establish a dating method the kinetics and mechanisms of ...
Crystal structure, electronic structure and - Research Online
... factor, P = S 2σ , which are functions of thermal conductivity (κ), Seebeck coefficient (S), electrical conductivity (σ), and absolute temperature. The figure of merit increases with thermoelectric efficiency and is achieved by either enhancement of the power factor and/or by decreasing the thermal ...
... factor, P = S 2σ , which are functions of thermal conductivity (κ), Seebeck coefficient (S), electrical conductivity (σ), and absolute temperature. The figure of merit increases with thermoelectric efficiency and is achieved by either enhancement of the power factor and/or by decreasing the thermal ...
Adsorption energy and spin state of first
... is due to the metal-substrate covalent bonding that also implies a polarization of the metal orbitals 共a redistribution of the atomic orbital population兲. In all cases, the metal s orbital combines with the oxygen p orbital perpendicular to the surface resulting in a bonding 共occupied兲 and an antibo ...
... is due to the metal-substrate covalent bonding that also implies a polarization of the metal orbitals 共a redistribution of the atomic orbital population兲. In all cases, the metal s orbital combines with the oxygen p orbital perpendicular to the surface resulting in a bonding 共occupied兲 and an antibo ...
A new, intrinsic, thermal parameter for enzymes reveals true
... effect of temperature on enzyme reactions within the cell. Unlike the Arrhenius activation energy, which is unaffected by the source (“evolved”) temperature of the enzyme, and enzyme stability, which is not necessarily related to activity, Teq is central to the physiological adaptation of an enzyme ...
... effect of temperature on enzyme reactions within the cell. Unlike the Arrhenius activation energy, which is unaffected by the source (“evolved”) temperature of the enzyme, and enzyme stability, which is not necessarily related to activity, Teq is central to the physiological adaptation of an enzyme ...
Time-resolved nonlinear optical spectroscopy
... • Multiferroic materials with both polar and magnetic order parameters usually show a relatively low-symmetry crystal structure due to the absence of both time and space inversion symmetries; hence, a strong interaction between the low-lying magnetic and lattice excitations can occur, leading to ri ...
... • Multiferroic materials with both polar and magnetic order parameters usually show a relatively low-symmetry crystal structure due to the absence of both time and space inversion symmetries; hence, a strong interaction between the low-lying magnetic and lattice excitations can occur, leading to ri ...
ENGLISH for ENGINEERS - University of Southampton
... already fully polymerized and exist as solids at room temperature but can be melted and reshaped which eases the manufacture process [2]. The fibres in a PMC provide properties like in-plane tensile strength and flexural strength while the matrix provides support when the material is under compressi ...
... already fully polymerized and exist as solids at room temperature but can be melted and reshaped which eases the manufacture process [2]. The fibres in a PMC provide properties like in-plane tensile strength and flexural strength while the matrix provides support when the material is under compressi ...
Folie 1
... (Examples: change of the height of a weight, expansion of a gas that pushes a piston and raises the weight, or a chemical reaction which e.g. drives an electrical current) The energy of a system is its capacity to do work. When work is done on an otherwise isolated system (e.g. by compressing a ga ...
... (Examples: change of the height of a weight, expansion of a gas that pushes a piston and raises the weight, or a chemical reaction which e.g. drives an electrical current) The energy of a system is its capacity to do work. When work is done on an otherwise isolated system (e.g. by compressing a ga ...
Chapter 15 - College of Engineering WordPress
... Both Tm and Tg increase with increasing chain stiffness Chain stiffness increased by presence of 1. Bulky sidegroups 2. Polar groups or sidegroups 3. Chain double bonds and aromatic chain groups Regularity of repeat unit arrangements – affects Tm only ...
... Both Tm and Tg increase with increasing chain stiffness Chain stiffness increased by presence of 1. Bulky sidegroups 2. Polar groups or sidegroups 3. Chain double bonds and aromatic chain groups Regularity of repeat unit arrangements – affects Tm only ...
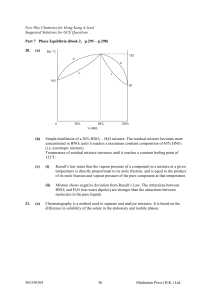
Book 2,Part 7 - GEOCITIES.ws
... The liquid mixture is said to be ideal if the intermolecular attraction between the molecules in the mixture is approximately equal to that in pure A and that in pure B. The liquid mixtures which approximate to ideal behaviour can be separated into their pure components by fractional distillation. T ...
... The liquid mixture is said to be ideal if the intermolecular attraction between the molecules in the mixture is approximately equal to that in pure A and that in pure B. The liquid mixtures which approximate to ideal behaviour can be separated into their pure components by fractional distillation. T ...
Glass transition
The glass–liquid transition or glass transition for short is the reversible transition in amorphous materials (or in amorphous regions within semicrystalline materials) from a hard and relatively brittle state into a molten or rubber-like state. An amorphous solid that exhibits a glass transition is called a glass. Supercooling a viscous liquid into the glass state is called vitrification, from the Latin vitreum, ""glass"" via French vitrifier.Despite the massive change in the physical properties of a material through its glass transition, the transition is not itself a phase transition of any kind; rather it is a laboratory phenomenon extending over a range of temperature and defined by one of several conventions. Such conventions include a constant cooling rate (20 K/min) and a viscosity threshold of 1012 Pa·s, among others. Upon cooling or heating through this glass-transition range, the material also exhibits a smooth step in the thermal-expansion coefficient and in the specific heat, with the location of these effects again being dependent on the history of the material. However, the question of whether some phase transition underlies the glass transition is a matter of continuing research.The glass-transition temperature Tg is always lower than the melting temperature, Tm, of the crystalline state of the material, if one exists.