A review on shape memory alloys with applications to morphing
... temperature. It consists of a dense arrangement of crystal planes placed with an opposite orientation and with very high relative mobility (resulting in low Young’s modulus and good damping characteristics). When the material is loaded above the yield point in the martensite phase, instead of breaki ...
... temperature. It consists of a dense arrangement of crystal planes placed with an opposite orientation and with very high relative mobility (resulting in low Young’s modulus and good damping characteristics). When the material is loaded above the yield point in the martensite phase, instead of breaki ...
Journal - AIMS Lab
... in a finite size silica chain, they can be divided into three classes of bridging, pairing, and ending sites [Fig. 1(e)]. The structural and mechanical properties of CSHs are provided in Table I. Anomalous tobermorite has the highest Ez and it is due to the existence of Q3 in the crystal which bridg ...
... in a finite size silica chain, they can be divided into three classes of bridging, pairing, and ending sites [Fig. 1(e)]. The structural and mechanical properties of CSHs are provided in Table I. Anomalous tobermorite has the highest Ez and it is due to the existence of Q3 in the crystal which bridg ...
Understanding Damping Techniques for Noise and Vibration Control
... a fluid through orifices that offer a predetermined resistance to highvelocity flow. Many isolation systems use elastomeric materials to provide both the spring force and damping. Some rubbers are capable of achieving useful damping at certain frequencies, although at low frequencies most exhibit lo ...
... a fluid through orifices that offer a predetermined resistance to highvelocity flow. Many isolation systems use elastomeric materials to provide both the spring force and damping. Some rubbers are capable of achieving useful damping at certain frequencies, although at low frequencies most exhibit lo ...
An Optimal Cure Process to Minimize Residual Void and
... (point B) increased to a maximum after four minutes, followed by decreases, before it maintained a constant value after 12 min. The degree of cure for four minutes elapsing at these two sides showed that the wall side was completely solidified due to abrupt curing process on four minutes of curing t ...
... (point B) increased to a maximum after four minutes, followed by decreases, before it maintained a constant value after 12 min. The degree of cure for four minutes elapsing at these two sides showed that the wall side was completely solidified due to abrupt curing process on four minutes of curing t ...
STOICHIOMETRY
... In many industrial processes and laboratory reactions the actual yield obtained is significantly less than the theoretical yield. The relationship between the actual yield and the theoretical yield is called the percentage yield (% yield). ...
... In many industrial processes and laboratory reactions the actual yield obtained is significantly less than the theoretical yield. The relationship between the actual yield and the theoretical yield is called the percentage yield (% yield). ...
UNCONVENTIONAL MACHINING PROCESS
... machinery and readily available cutting tools. Non-traditional processes require expensive equipment and tooling as well as skilled labor, which increases significantly the production cost; From the above it follows that non-traditional processes generally should be employed when 6. there is a need ...
... machinery and readily available cutting tools. Non-traditional processes require expensive equipment and tooling as well as skilled labor, which increases significantly the production cost; From the above it follows that non-traditional processes generally should be employed when 6. there is a need ...
the effects of corrosion on the mechanical properties of aluminium
... aluminum has a huge advantage when compared to other metals, it is not always completely impervious to corrosion. Its protective oxide layer can become unstable when exposed to extreme pH levels. When the environment is highly acidic or basic, breakdown of the protective layer can occur, and its aut ...
... aluminum has a huge advantage when compared to other metals, it is not always completely impervious to corrosion. Its protective oxide layer can become unstable when exposed to extreme pH levels. When the environment is highly acidic or basic, breakdown of the protective layer can occur, and its aut ...
Anisotropy of Elastic Behavior
... • Different crystal systems can be characterized exclusively by their symmetries. Table 7-1 presents the different symmetry operations defining the seven crystal systems. • The seven crystalline systems can be perfectly described by their axes of rotation. For example, a threefold rotation is a rota ...
... • Different crystal systems can be characterized exclusively by their symmetries. Table 7-1 presents the different symmetry operations defining the seven crystal systems. • The seven crystalline systems can be perfectly described by their axes of rotation. For example, a threefold rotation is a rota ...
Chemical Reactions Q3U3
... amount of solute per solution, the solution can not dissolve any more solute Unsaturated solution: The amount of solute dissolved is less than the amount of solute than can be dissolved, the solvent can dissolve more solute Supersaturated solution: Unstable solutions which contain more dissolved ...
... amount of solute per solution, the solution can not dissolve any more solute Unsaturated solution: The amount of solute dissolved is less than the amount of solute than can be dissolved, the solvent can dissolve more solute Supersaturated solution: Unstable solutions which contain more dissolved ...
The Reactivity of Colloidal Inorganic Nanoparticles
... of a nanostructured material, the atomic surface-to-volume ratio increases. In this process, most of the atoms are preferentially located at the edges and kinks of the particle, which dramatically increases its reactivity. Another consequence of the high surface-to-volume ratio of INPs is their high ...
... of a nanostructured material, the atomic surface-to-volume ratio increases. In this process, most of the atoms are preferentially located at the edges and kinks of the particle, which dramatically increases its reactivity. Another consequence of the high surface-to-volume ratio of INPs is their high ...
Signs of Reaction - Calderglen High School
... IDE means that the compound is made up of the TWO elements obvious from the name. Except: Hydroxide (something + H + 0) and ...
... IDE means that the compound is made up of the TWO elements obvious from the name. Except: Hydroxide (something + H + 0) and ...
The science of chemistry is concerned with the composition
... compressible. Even when quite modest pressures are applied, the volume decreases noticeably. It is also much less dense than the liquid or the solid. At 400°C and ordinary pressures, its density is 3.6 × 10–3 g cm–3 about one four-thousandth that of solid or liquid mercury. A modem chemist would int ...
... compressible. Even when quite modest pressures are applied, the volume decreases noticeably. It is also much less dense than the liquid or the solid. At 400°C and ordinary pressures, its density is 3.6 × 10–3 g cm–3 about one four-thousandth that of solid or liquid mercury. A modem chemist would int ...
The science of chemistry is concerned with the
... compressible. Even when quite modest pressures are applied, the volume decreases noticeably. It is also much less dense than the liquid or the solid. At 400°C and ordinary pressures, its density is 3.6 × 10–3 g cm–3 about one four-thousandth that of solid or liquid mercury. A modem chemist would int ...
... compressible. Even when quite modest pressures are applied, the volume decreases noticeably. It is also much less dense than the liquid or the solid. At 400°C and ordinary pressures, its density is 3.6 × 10–3 g cm–3 about one four-thousandth that of solid or liquid mercury. A modem chemist would int ...
I INTRODUCTION TO MAGNETISM AND MAGNETIC MATERIALS
... they are magnetized and possess large coercive fields. The characterization of soft and hard ferrites is based upon some important parameters like: 1) The residual magnetism (remanence (Mr)), that though materials retains when the external field is removed. 2) The saturation flux or maximum magnetic ...
... they are magnetized and possess large coercive fields. The characterization of soft and hard ferrites is based upon some important parameters like: 1) The residual magnetism (remanence (Mr)), that though materials retains when the external field is removed. 2) The saturation flux or maximum magnetic ...
Synthesis and Characterization of Amorphous and Hybrid Materials
... hydrolysis reaction has been performed with r values ranging from <1 to over 25 depending on the desired polysilicate product, for example, fibers, bulk gel or colloidal particles. From eq. 1, an increased value of r is expected to promote the hydrolysis reaction. (Aelion et al., 1950a, 1950b) found ...
... hydrolysis reaction has been performed with r values ranging from <1 to over 25 depending on the desired polysilicate product, for example, fibers, bulk gel or colloidal particles. From eq. 1, an increased value of r is expected to promote the hydrolysis reaction. (Aelion et al., 1950a, 1950b) found ...
AP Ch 3 Stoichiometry
... • We found that chlorine is the limiting reactant, and 43.8 g of aluminum chloride are produced. 35.0 g Cl2 1 mol Cl2 2 mol Al 71 g Cl2 ...
... • We found that chlorine is the limiting reactant, and 43.8 g of aluminum chloride are produced. 35.0 g Cl2 1 mol Cl2 2 mol Al 71 g Cl2 ...
- Opus
... observed to occur gradually and over temperature ranges in excess of 200oC while the ultimate residual masses could not be correlated with any meaningful stoichiometric component of the precursor molecules. In contrast, and in common with the observations of Tilley and co-workers,7 the onset of deco ...
... observed to occur gradually and over temperature ranges in excess of 200oC while the ultimate residual masses could not be correlated with any meaningful stoichiometric component of the precursor molecules. In contrast, and in common with the observations of Tilley and co-workers,7 the onset of deco ...
Sample Exercise 3.1 Interpreting and Balancing Chemical Equations
... Write the balanced equation for the reaction that occurs when methanol, CH3OH(l), is burned in air. Solution When any compound containing C, H, and O is combusted, it reacts with the O2(g) in air to produce CO2(g) and H2O(g). Thus, the unbalanced equation is CH3OH(l) + O2(g) → CO2(g) + H2O(g) In thi ...
... Write the balanced equation for the reaction that occurs when methanol, CH3OH(l), is burned in air. Solution When any compound containing C, H, and O is combusted, it reacts with the O2(g) in air to produce CO2(g) and H2O(g). Thus, the unbalanced equation is CH3OH(l) + O2(g) → CO2(g) + H2O(g) In thi ...
Micromechanics of Cell Walls
... Young’s or elastic modulus and is often given the symbol, E, the constant in Hooke’s Law. The Young’s modulus gives the stiffness of a material, in other words the resistance of a material to elastic deformation and has the same units as stress (Pascals). The higher E is, the more stress is required ...
... Young’s or elastic modulus and is often given the symbol, E, the constant in Hooke’s Law. The Young’s modulus gives the stiffness of a material, in other words the resistance of a material to elastic deformation and has the same units as stress (Pascals). The higher E is, the more stress is required ...
Slide 1
... Write the balanced equation for the reaction that occurs when methanol, CH 3OH(l), is burned in air. Solution When any compound containing C, H, and O is combusted, it reacts with the O 2(g) in air to produce CO2(g) and H2O(g). Thus, the unbalanced equation is CH3OH(l) + O2(g) → CO2(g) + H2O(g) In t ...
... Write the balanced equation for the reaction that occurs when methanol, CH 3OH(l), is burned in air. Solution When any compound containing C, H, and O is combusted, it reacts with the O 2(g) in air to produce CO2(g) and H2O(g). Thus, the unbalanced equation is CH3OH(l) + O2(g) → CO2(g) + H2O(g) In t ...
Ta alloys, studied by X-ray diffraction and molecular dynamics
... The resulting structures were then equilibrated for 1 ns with Berendsen pressure scaling; the initial temperature was set to 300 K. Fig. 2a–c shows the temperature evolution during equilibration for the fcc, bcc and b-Ta lattices, respectively. It can be seen that the temperature does stabilize with ...
... The resulting structures were then equilibrated for 1 ns with Berendsen pressure scaling; the initial temperature was set to 300 K. Fig. 2a–c shows the temperature evolution during equilibration for the fcc, bcc and b-Ta lattices, respectively. It can be seen that the temperature does stabilize with ...
Electrical Effects and Devices/Dielectrics and Insulators
... displays the usual t–1 dependence and the plot of i(t)t versus kTln(nt) yields the distribution of trap depths. Eq. (55.14) represents an approximation, which underestimates the current associated with the shallow traps and overcompensates for the current due to the deep traps. The mobility of the f ...
... displays the usual t–1 dependence and the plot of i(t)t versus kTln(nt) yields the distribution of trap depths. Eq. (55.14) represents an approximation, which underestimates the current associated with the shallow traps and overcompensates for the current due to the deep traps. The mobility of the f ...
Stoichiometry: Calculations with Chemical Formulas and Equations
... Write the balanced equation for the reaction that occurs when methanol,CH3OH(l), is burned in air. When any compound containing C, H, and O is combusted, it reacts with the O2(g) in air to produce CO2(g) and H2O(g). Thus, the unbalanced equation is CH3OH(l) + O2(g) CO2(g) + H2O(g) The C atoms are ...
... Write the balanced equation for the reaction that occurs when methanol,CH3OH(l), is burned in air. When any compound containing C, H, and O is combusted, it reacts with the O2(g) in air to produce CO2(g) and H2O(g). Thus, the unbalanced equation is CH3OH(l) + O2(g) CO2(g) + H2O(g) The C atoms are ...
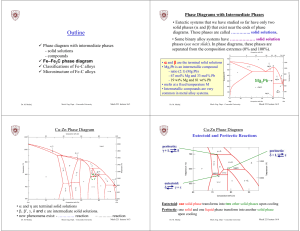
Outline - Concordia University
... • The maximum solubility of C is 0.022 wt% (interstitial solubility) • Soft and relatively easy to deform γ -austenite - solid solution of C in …….. Fe • The maximum solubility of C is 2.14 wt % at 1147°C. • Interstitial lattice positions are much larger than ferrite (higher C%) • Is not stable belo ...
... • The maximum solubility of C is 0.022 wt% (interstitial solubility) • Soft and relatively easy to deform γ -austenite - solid solution of C in …….. Fe • The maximum solubility of C is 2.14 wt % at 1147°C. • Interstitial lattice positions are much larger than ferrite (higher C%) • Is not stable belo ...