Naming Compounds
... determine the elements involved in the chemical formula (compound)…. Metals and Non- Metals determine the type of compound (Ionic or Molecular) follow the rules outline for Ionic or Molecular ...
... determine the elements involved in the chemical formula (compound)…. Metals and Non- Metals determine the type of compound (Ionic or Molecular) follow the rules outline for Ionic or Molecular ...
Types of Measurement
... 6. A chemical equation uses symbolic language to describe a chemical reaction. 7. Equation means equal numbers of atoms of each element on both sides. 8. Quantities of reactants and products are expressed in moles by using coefficients. ...
... 6. A chemical equation uses symbolic language to describe a chemical reaction. 7. Equation means equal numbers of atoms of each element on both sides. 8. Quantities of reactants and products are expressed in moles by using coefficients. ...
2 - grade11chemistry
... electrons in the outermost energy level are placed as dots on each of the sides. 4. The electrons are placed singly on each side and are then paired on each side until the number of outer electrons has been attained. ...
... electrons in the outermost energy level are placed as dots on each of the sides. 4. The electrons are placed singly on each side and are then paired on each side until the number of outer electrons has been attained. ...
Ch 4 Student.pptx
... • Say you are asked how much CO2 is produced from the combustion of 15.0 moles of octane? • 2 C8H18 (l) + 25 O2 (g) → 16 CO2 (g) + 18 H2O (g) ...
... • Say you are asked how much CO2 is produced from the combustion of 15.0 moles of octane? • 2 C8H18 (l) + 25 O2 (g) → 16 CO2 (g) + 18 H2O (g) ...
chapter_2_2007
... The strength of an acid or base is determined by how completely it will dissociate in water. – Strong acids release almost all of their hydrogen ions into water. – Strong bases release almost all of their hydroxide ions into water. ...
... The strength of an acid or base is determined by how completely it will dissociate in water. – Strong acids release almost all of their hydrogen ions into water. – Strong bases release almost all of their hydroxide ions into water. ...
C. Adding acid shifts the equilibrium to the right
... and properties. Chm.1.2.1 Compare (qualitatively) the relative strengths of ionic, covalent, and metallic bonds. Chm.1.2.2 Infer the type of bond and chemical formula formed between atoms. Chm.1.2.3 Compare inter- and intra- particle forces. Chm.1.2.4 Interpret the name and formula of compounds usin ...
... and properties. Chm.1.2.1 Compare (qualitatively) the relative strengths of ionic, covalent, and metallic bonds. Chm.1.2.2 Infer the type of bond and chemical formula formed between atoms. Chm.1.2.3 Compare inter- and intra- particle forces. Chm.1.2.4 Interpret the name and formula of compounds usin ...
EXAM 3 NAME____KEY –Answers highlighted______(1 pt.) Dr
... Bonus questions (3 points each) 34. Commercial cold packs consist of solid ammonium nitrate and water. NH4NO3 absorbs 3.30 × 102 J of heat per gram dissolved in water. In a coffee-cup calorimeter, 4.40 g NH4NO3 is dissolved in 100.0 g of water at 21.0 °C. What is the final temperature of the solutio ...
... Bonus questions (3 points each) 34. Commercial cold packs consist of solid ammonium nitrate and water. NH4NO3 absorbs 3.30 × 102 J of heat per gram dissolved in water. In a coffee-cup calorimeter, 4.40 g NH4NO3 is dissolved in 100.0 g of water at 21.0 °C. What is the final temperature of the solutio ...
The Chemical Basis of Life
... – Elements are listed in order of their atomic numbers – Elements are designated by standard one or twoletter abbreviations – Elements in the same vertical column often have very similar chemical bonding properties ...
... – Elements are listed in order of their atomic numbers – Elements are designated by standard one or twoletter abbreviations – Elements in the same vertical column often have very similar chemical bonding properties ...
6-1 Endothermic and Exothermic Reactions
... someone take note of the initial temperature. SLOWLY add ACID TO WATER. Take temperature again. Typical temperature change will be from around 22o C to 93o C. The equation for this reaction is: H2O (l) + H2SO4 (aq) = H2O (l) + H2SO4 (aq) (diluted) The amount of heat in Joules can be calculated as fo ...
... someone take note of the initial temperature. SLOWLY add ACID TO WATER. Take temperature again. Typical temperature change will be from around 22o C to 93o C. The equation for this reaction is: H2O (l) + H2SO4 (aq) = H2O (l) + H2SO4 (aq) (diluted) The amount of heat in Joules can be calculated as fo ...
Cumulative Review, entire quarter
... atoms combined to make that particular compound (NH4)2 C2O4 is the formula for ammonium oxalate, There are 2 nitrogens, 8 hydrogens, 2 carbons and 4 oxygens in one molecule. The nitrogen and hydrogen occur in two groups of 4 hydrogens and one nitrogen The carbons and oxygens occur as one group of 2 ...
... atoms combined to make that particular compound (NH4)2 C2O4 is the formula for ammonium oxalate, There are 2 nitrogens, 8 hydrogens, 2 carbons and 4 oxygens in one molecule. The nitrogen and hydrogen occur in two groups of 4 hydrogens and one nitrogen The carbons and oxygens occur as one group of 2 ...
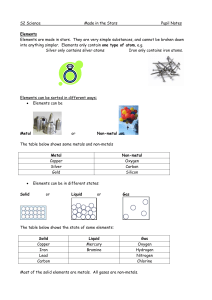
Made in the Stars Notes
... at room temperature except for mercury, which is a liquid. Non-metal solids are usually brittle (they break easily). Non-metals can be solids, liquids or gases at room temperature. Non-metals usually have low melting and boiling points. They are poor conductors of electricity. The exception is graph ...
... at room temperature except for mercury, which is a liquid. Non-metal solids are usually brittle (they break easily). Non-metals can be solids, liquids or gases at room temperature. Non-metals usually have low melting and boiling points. They are poor conductors of electricity. The exception is graph ...
Chemistry in Biology
... COMPOUNDS II. Composition of Matter A. Elements—pure substances that cannot be broken down into simpler kinds of matter • Made of one type of atom • More than 100 elements (92 naturally occurring) • 90% of the mass of an organism is composed of 4 elements (oxygen, carbon, hydrogen, and nitrogen) • E ...
... COMPOUNDS II. Composition of Matter A. Elements—pure substances that cannot be broken down into simpler kinds of matter • Made of one type of atom • More than 100 elements (92 naturally occurring) • 90% of the mass of an organism is composed of 4 elements (oxygen, carbon, hydrogen, and nitrogen) • E ...
Wizard Test Maker
... conductor of electricity? A 3 A KCl B 7 B C6H12O6 C 8 C CO2 D 11 D CO 33. When ethylene glycol (an antifreeze) is added to water, what happens to the boiling point of the water? A It decreases, and the freezing point ...
... conductor of electricity? A 3 A KCl B 7 B C6H12O6 C 8 C CO2 D 11 D CO 33. When ethylene glycol (an antifreeze) is added to water, what happens to the boiling point of the water? A It decreases, and the freezing point ...
Balancing Equations
... Chemical reactions occur when bonds (between the electrons of atoms) are formed or broken Chemical reactions involve changes in the chemical composition of matter the making of new materials with new ...
... Chemical reactions occur when bonds (between the electrons of atoms) are formed or broken Chemical reactions involve changes in the chemical composition of matter the making of new materials with new ...
AP Syllabus
... ends during the last week of May. In order to increase class time and meet the demands of the course we also meet before or after school for an hour each week. Extended lab time is also available once a week. We do this during zero periods for those that do not have the optional zero period course, ...
... ends during the last week of May. In order to increase class time and meet the demands of the course we also meet before or after school for an hour each week. Extended lab time is also available once a week. We do this during zero periods for those that do not have the optional zero period course, ...
CH 4 Notes
... The neutral Ca has lost two electrons to 2 H1+ to become Ca2+ We say Ca has been oxidized to Ca2+ When a substance gains electrons, it undergoes reduction: 2 Ca (s) + O2 (g) ---> 2 CaO (s) In this reaction the neutral O2 has gained electrons from the Ca to become O2- in CaO. We say O2 has ...
... The neutral Ca has lost two electrons to 2 H1+ to become Ca2+ We say Ca has been oxidized to Ca2+ When a substance gains electrons, it undergoes reduction: 2 Ca (s) + O2 (g) ---> 2 CaO (s) In this reaction the neutral O2 has gained electrons from the Ca to become O2- in CaO. We say O2 has ...
AP Chem Equations - Speedway High School
... Phosphorus halides react with water to produce an acid of phosphorus (phosphorous acid or phosphoric acid) and a hydrohalic acid. The oxidation number of the phosphorus remains the same in both ...
... Phosphorus halides react with water to produce an acid of phosphorus (phosphorous acid or phosphoric acid) and a hydrohalic acid. The oxidation number of the phosphorus remains the same in both ...
WRITING AP EQUATIONS AP equation sets are found in the free
... Phosphorus halides react with water to produce an acid of phosphorus (phosphorous acid or phosphoric acid) and a hydrohalic acid. The oxidation number of the phosphorus remains the same in both ...
... Phosphorus halides react with water to produce an acid of phosphorus (phosphorous acid or phosphoric acid) and a hydrohalic acid. The oxidation number of the phosphorus remains the same in both ...
Midterm Review 2017
... an element is the element’s ability to 1) react with an acid 2) react with oxygen 3) form a compound with chlorine 4) form an aqueous solution 59) Which element forms an ionic compound when it reacts with lithium? 1) K ...
... an element is the element’s ability to 1) react with an acid 2) react with oxygen 3) form a compound with chlorine 4) form an aqueous solution 59) Which element forms an ionic compound when it reacts with lithium? 1) K ...
File
... Copper is often stolen to sell for scrap, if cables are stolen this can cause problems with trains or electrical supply or motorway signalling. Both are made of copper, the anode is impure copper but the cathode is pure copper Copper (II) sulphate solution is the electrolyte. The Anode loses mass as ...
... Copper is often stolen to sell for scrap, if cables are stolen this can cause problems with trains or electrical supply or motorway signalling. Both are made of copper, the anode is impure copper but the cathode is pure copper Copper (II) sulphate solution is the electrolyte. The Anode loses mass as ...
Test #1 Study Guide
... the mass of an electron was about 200 times lighter than a hydrogen atom. Ernest Rutherford – Through his gold foil experiment in which he shot particles through a piece of gold foil and recorded where these particles ended up, he disproved the plum-pudding model of the atom and proposed nuclear t ...
... the mass of an electron was about 200 times lighter than a hydrogen atom. Ernest Rutherford – Through his gold foil experiment in which he shot particles through a piece of gold foil and recorded where these particles ended up, he disproved the plum-pudding model of the atom and proposed nuclear t ...
Chapter 3 - Warren County Schools
... – Composed of two or more different elements in distinct whole number ratios – Ex: Water, H2O – Ex: Salt, NaCl ...
... – Composed of two or more different elements in distinct whole number ratios – Ex: Water, H2O – Ex: Salt, NaCl ...
Class Notes
... body is water.) Also, when reacting ionic compounds, they are more easily mixed in an aqueous form than in their natural solid state. Therefore, it makes sense that many reactions take place in aqueous environments. When an ionic compound dissolves in water to make an aqueous solution, the crystalli ...
... body is water.) Also, when reacting ionic compounds, they are more easily mixed in an aqueous form than in their natural solid state. Therefore, it makes sense that many reactions take place in aqueous environments. When an ionic compound dissolves in water to make an aqueous solution, the crystalli ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.