Chemistry Standards and Frameworks
... 1. The periodic table displays the elements in increasing atomic number and shows how periodicity of the physical and chemical properties of the elements relates to atomic structure. As a basis for understanding this concept: 1. a.: Students know how to relate the position of an element in the perio ...
... 1. The periodic table displays the elements in increasing atomic number and shows how periodicity of the physical and chemical properties of the elements relates to atomic structure. As a basis for understanding this concept: 1. a.: Students know how to relate the position of an element in the perio ...
sch4ureview
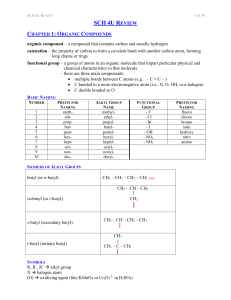
... monomers; two types: addition and condensation monomer – a molecule or compound usually containing carbon and of relatively low molecular weight and simple structure which is capable of conversion to polymers by combination with itself or other similar molecules or compounds dimer – a molecule made ...
... monomers; two types: addition and condensation monomer – a molecule or compound usually containing carbon and of relatively low molecular weight and simple structure which is capable of conversion to polymers by combination with itself or other similar molecules or compounds dimer – a molecule made ...
12 U Chem Review
... monomers; two types: addition and condensation monomer – a molecule or compound usually containing carbon and of relatively low molecular weight and simple structure which is capable of conversion to polymers by combination with itself or other similar molecules or compounds dimer – a molecule made ...
... monomers; two types: addition and condensation monomer – a molecule or compound usually containing carbon and of relatively low molecular weight and simple structure which is capable of conversion to polymers by combination with itself or other similar molecules or compounds dimer – a molecule made ...
Are you ready for S279?
... oxygen decreases. Any chemical reaction leading to oxidation of one substance must be accompanied by the reduction of another substance, and vice versa. The term oxidation is also used more generally to include any reaction in which an atom loses electrons. For example, the change of an iron(II) ion ...
... oxygen decreases. Any chemical reaction leading to oxidation of one substance must be accompanied by the reduction of another substance, and vice versa. The term oxidation is also used more generally to include any reaction in which an atom loses electrons. For example, the change of an iron(II) ion ...
введение в общую introductio to the general ch ведение в общую
... There are many substances that exist as two or more atoms connected together. These combinations are called molecules. A molecule is the smallest part of a substance that has the physical and chemical properties of that substance. Some elements exist in form of molecules. For example, hydrogen and o ...
... There are many substances that exist as two or more atoms connected together. These combinations are called molecules. A molecule is the smallest part of a substance that has the physical and chemical properties of that substance. Some elements exist in form of molecules. For example, hydrogen and o ...
George Facer`s A level Chemistry
... not a species that can be isolated. It changes immediately into the product. An analogy is that the transition state is like the top of a pass going from one valley to another. ...
... not a species that can be isolated. It changes immediately into the product. An analogy is that the transition state is like the top of a pass going from one valley to another. ...
Name: 1) At 1 atmosphere and 298 K, 1 mole of H O(l) molecules
... Using your knowledge of chemistry and the information in the Vapor Pressure of Four Liquids chemistry reference table, which statement concerning propanone and water at 50DC is true? A) Propanone has a lower vapor pressure and weaker intermolecular forces than water. B) Propanone has a lower vapor p ...
... Using your knowledge of chemistry and the information in the Vapor Pressure of Four Liquids chemistry reference table, which statement concerning propanone and water at 50DC is true? A) Propanone has a lower vapor pressure and weaker intermolecular forces than water. B) Propanone has a lower vapor p ...
Chapter 4-5
... of 2.50 M HNO3 (aq) producing 12.38 g of N2O (g). Calculate the % yield of N2O. Solution :4Zn (s) + 10 HNO3 (aq) → 4Zn(NO3)2(aq) + N2O(g) + 5H2O(l) – Calculate moles of each reactant – Then calculate the amount of N2O produced by using in turn all the moles of each reactant. – The reactant giving th ...
... of 2.50 M HNO3 (aq) producing 12.38 g of N2O (g). Calculate the % yield of N2O. Solution :4Zn (s) + 10 HNO3 (aq) → 4Zn(NO3)2(aq) + N2O(g) + 5H2O(l) – Calculate moles of each reactant – Then calculate the amount of N2O produced by using in turn all the moles of each reactant. – The reactant giving th ...
Chemistry I Syllabus 2011-2012
... Classroom Procedures: 1. Enter the classroom quietly, sit in your assigned seat and begin work. 2. Turn in assignments to the appropriate box assigned to your class and in a timely manner. 3. Sharpen pencils only during student work time and only until you hear the sharpener change pitch. 4. No hall ...
... Classroom Procedures: 1. Enter the classroom quietly, sit in your assigned seat and begin work. 2. Turn in assignments to the appropriate box assigned to your class and in a timely manner. 3. Sharpen pencils only during student work time and only until you hear the sharpener change pitch. 4. No hall ...
Equation Writing Information
... On the AP examination you will encounter a question in which you will be required to write net ionic equations for various reactions. In past years, students have been required to choose 5 of 8 reactions. Some of the reactions you will undoubtedly recognize; others you will not! Hopefully, at least ...
... On the AP examination you will encounter a question in which you will be required to write net ionic equations for various reactions. In past years, students have been required to choose 5 of 8 reactions. Some of the reactions you will undoubtedly recognize; others you will not! Hopefully, at least ...
Print out Reviews # 1 through # 17
... 4. Draw the electron dot diagrams for the following elements. (A) argon (B) phosphorus (C) sodium (D) silicon (E) aluminum (F) bromine EOC REVIEW #5 1. How is an element’s outer electron configuration related to its position on the Periodic Table? 2. What are the symbols for all of the elements that ...
... 4. Draw the electron dot diagrams for the following elements. (A) argon (B) phosphorus (C) sodium (D) silicon (E) aluminum (F) bromine EOC REVIEW #5 1. How is an element’s outer electron configuration related to its position on the Periodic Table? 2. What are the symbols for all of the elements that ...
Chapter 2 Introduction to Chemistry
... however, careful measurements show that the total mass of the reactants (coal and oxygen) equals the total mass of the ...
... however, careful measurements show that the total mass of the reactants (coal and oxygen) equals the total mass of the ...
Element Symbol
... substance. The first letter is always capitalized and the second, if there is one, is lower case. ...
... substance. The first letter is always capitalized and the second, if there is one, is lower case. ...
Unit 2
... a) which elements are metals? _____________________________________ b) which elements are nonmetals? ___________________________________ c) which elements are metalloids? ___________________________________ 25) A compound has two boron atoms and six hydrogen atoms. a) what is its empirical formula? ...
... a) which elements are metals? _____________________________________ b) which elements are nonmetals? ___________________________________ c) which elements are metalloids? ___________________________________ 25) A compound has two boron atoms and six hydrogen atoms. a) what is its empirical formula? ...
Unit 2
... a) which elements are metals? _____________________________________ b) which elements are nonmetals? ___________________________________ c) which elements are metalloids? ___________________________________ 25) A compound has two boron atoms and six hydrogen atoms. a) what is its empirical formula? ...
... a) which elements are metals? _____________________________________ b) which elements are nonmetals? ___________________________________ c) which elements are metalloids? ___________________________________ 25) A compound has two boron atoms and six hydrogen atoms. a) what is its empirical formula? ...
Chemical Reactions
... Zn(s) + Cu 2+(aq) Zn2+(aq) + Cu(s) Zn is oxidizing agent Zn(s) Zn2+(aq) + 2 e1Cu 2+(aq) is reducing agent Cu 2+(aq) + 2 e1- Cu(s) ...
... Zn(s) + Cu 2+(aq) Zn2+(aq) + Cu(s) Zn is oxidizing agent Zn(s) Zn2+(aq) + 2 e1Cu 2+(aq) is reducing agent Cu 2+(aq) + 2 e1- Cu(s) ...
Dalton`s Laws worksheet
... Dalton’s Atomic Theory of Matter 1. Which of the following statements is part of Dalton’s atomic theory of matter? a. All atoms are identical b. All atoms of a given element are identical c. All atoms differ from one another d. Atoms of the same element can have a different shape 2. Dalton suggested ...
... Dalton’s Atomic Theory of Matter 1. Which of the following statements is part of Dalton’s atomic theory of matter? a. All atoms are identical b. All atoms of a given element are identical c. All atoms differ from one another d. Atoms of the same element can have a different shape 2. Dalton suggested ...
H2, N2, O2, F2, Cl2, Br2, I2
... 1. Don't forget the 7 diatomic elements, the HOFBrINCl 's, the big 7, the "gens". They are never by themselves in a chemical reaction and they are paired in their elemental form: ...
... 1. Don't forget the 7 diatomic elements, the HOFBrINCl 's, the big 7, the "gens". They are never by themselves in a chemical reaction and they are paired in their elemental form: ...
Students know
... a. Students know the rate of reaction is the decrease in concentration of reactants or the increase in concentration of products with time. b. Students know how reaction rates depend on such factors as concentration, temperature, and pressure. c. Students know the role a catalyst plays in increasing ...
... a. Students know the rate of reaction is the decrease in concentration of reactants or the increase in concentration of products with time. b. Students know how reaction rates depend on such factors as concentration, temperature, and pressure. c. Students know the role a catalyst plays in increasing ...
The Major Classes of Chemical Reactions
... Many reactions take place in aqueous solution, and our first step toward comprehending these reactions is to understand how water acts as a solvent. The role a solvent plays in a reaction depends on its chemical nature. Some solvents play a passive role, dispersing the dissolved substances into indiv ...
... Many reactions take place in aqueous solution, and our first step toward comprehending these reactions is to understand how water acts as a solvent. The role a solvent plays in a reaction depends on its chemical nature. Some solvents play a passive role, dispersing the dissolved substances into indiv ...
6.02 × 1023 molecules = 1 mole
... 1. Given the equation PbO2 → PbO + O2, how many grams of oxygen will be produced if 47.8g of PbO2 decompose to form 44.6g of PbO and oxygen gas? ...
... 1. Given the equation PbO2 → PbO + O2, how many grams of oxygen will be produced if 47.8g of PbO2 decompose to form 44.6g of PbO and oxygen gas? ...
answers to part a of the national high school
... Calculations of this sort are, of course, important in ascertaining how much of an element such as manganese can be extracted from a particular ore, although, it has to be said, that braunite is not commonly used as a source of manganese. Note that the formulae given for minerals often uses the conv ...
... Calculations of this sort are, of course, important in ascertaining how much of an element such as manganese can be extracted from a particular ore, although, it has to be said, that braunite is not commonly used as a source of manganese. Note that the formulae given for minerals often uses the conv ...
PHYSICAL SETTING CHEMISTRY
... Graphite and diamond are two crystalline arrangements for carbon. The crystal structure of graphite is organized in layers. The bonds between carbon atoms within each layer of graphite are strong. The bonds between carbon atoms that connect different layers of graphite are weak because the shared el ...
... Graphite and diamond are two crystalline arrangements for carbon. The crystal structure of graphite is organized in layers. The bonds between carbon atoms within each layer of graphite are strong. The bonds between carbon atoms that connect different layers of graphite are weak because the shared el ...
OKEMOS PUBLIC SCHOOLS
... H-bonding > dipole-dipole > dispersion How would the viscosity of 3 liquids compare if each had a different type of IMF? H-bonding most viscous, dispersion least viscous Write the formula of the given compound. (You may need a polyatomic ion chart) Then, say if the compound is ionic or molecular. _L ...
... H-bonding > dipole-dipole > dispersion How would the viscosity of 3 liquids compare if each had a different type of IMF? H-bonding most viscous, dispersion least viscous Write the formula of the given compound. (You may need a polyatomic ion chart) Then, say if the compound is ionic or molecular. _L ...
Chapters 6 and 17: Chemical Thermodynamics
... (d) If the volume of the combustion container is 10.0 liters, calculate the final pressure in the container when the temperature is changed to 110°C. (Assume no oxygen remains unreacted and that all products are gaseous.) ...
... (d) If the volume of the combustion container is 10.0 liters, calculate the final pressure in the container when the temperature is changed to 110°C. (Assume no oxygen remains unreacted and that all products are gaseous.) ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.