AP_chemistry_Summer_Assignment_2014
... temperature is 68˚F. How many moles are present in the flask when the pressure is 1.10 atm and the temperature is 33˚C? 74.On a warm day, an amusement park balloon is filled with 47.8 g He. The temperature is 33˚C and the pressure in the balloon is 2.25 atm. Calculate the volume of the balloon. 75.A ...
... temperature is 68˚F. How many moles are present in the flask when the pressure is 1.10 atm and the temperature is 33˚C? 74.On a warm day, an amusement park balloon is filled with 47.8 g He. The temperature is 33˚C and the pressure in the balloon is 2.25 atm. Calculate the volume of the balloon. 75.A ...
Name - Deans Community High School
... b) Is the forward reaction is exothermic or endothermic. ............................................ 1 c) Gold and platinum both catalyse the reaction. For the forward reaction EA using gold is 30 kJ, while EA using platinum is 40 kJ. i) using different dotted lines add this information to the grap ...
... b) Is the forward reaction is exothermic or endothermic. ............................................ 1 c) Gold and platinum both catalyse the reaction. For the forward reaction EA using gold is 30 kJ, while EA using platinum is 40 kJ. i) using different dotted lines add this information to the grap ...
- Deans Community High School
... 2. Which of the following is not a correct statement about the effect of a catalyst? The catalyst A. provides an alternative route to the products B. lowers the energy which molecules need for successful collisions C. provides energy so that more molecules have successful collisions D. form bonds wi ...
... 2. Which of the following is not a correct statement about the effect of a catalyst? The catalyst A. provides an alternative route to the products B. lowers the energy which molecules need for successful collisions C. provides energy so that more molecules have successful collisions D. form bonds wi ...
Honors Chemistry I
... h. Potassium reacts with water yielding potassium hydroxide and hydrogen gas i. Solutions of silver (I) nitrate and lithium bromide are mixed. (predict the products and balance the equation) j. Solutions of zinc (II) sulfate and sodium phosphate are mixed. (predict the products and balance the equat ...
... h. Potassium reacts with water yielding potassium hydroxide and hydrogen gas i. Solutions of silver (I) nitrate and lithium bromide are mixed. (predict the products and balance the equation) j. Solutions of zinc (II) sulfate and sodium phosphate are mixed. (predict the products and balance the equat ...
chemical bonds - geraldinescience
... Chemical Formulas • A chemical formula is a combination of letters and numbers that shows which elements make up a compound and the number of atoms of each element that are required to make a molecule of a compound. • In a chemical formula, the subscript that appears after the symbol for an element ...
... Chemical Formulas • A chemical formula is a combination of letters and numbers that shows which elements make up a compound and the number of atoms of each element that are required to make a molecule of a compound. • In a chemical formula, the subscript that appears after the symbol for an element ...
Lab 1
... Primary substances, called elements, build all the materials about you. Some look similar, but others look unlike anything else. In this experiment, you will describe the physical properties of elements in a laboratory display and determine the location of elements on a blank periodic table. A. Phys ...
... Primary substances, called elements, build all the materials about you. Some look similar, but others look unlike anything else. In this experiment, you will describe the physical properties of elements in a laboratory display and determine the location of elements on a blank periodic table. A. Phys ...
Experiment 15: Reduction and Oxidation of Organic Compounds
... The reduction of a carbonyl group in an organic compound can be readily accomplished with a metal hydride, such as lithium aluminum hydride or sodium borohydride. While LiAlH4 is the more powerful of the two, capable of reducing aldehydes, ketones, carboxylic acids, esters and amides, NaBH4 is easie ...
... The reduction of a carbonyl group in an organic compound can be readily accomplished with a metal hydride, such as lithium aluminum hydride or sodium borohydride. While LiAlH4 is the more powerful of the two, capable of reducing aldehydes, ketones, carboxylic acids, esters and amides, NaBH4 is easie ...
Batteries are all over the place -- in our cars, our
... hearing aides because of their long life. Lead-acid battery - Lead-acid chemistry is used in automobiles, the electrodes are made of lead and lead-oxide with a strong acidic electrolyte (rechargeable). Nickel-cadmium battery - The electrodes are nickel-hydroxide and cadmium, with potassium-hydroxide ...
... hearing aides because of their long life. Lead-acid battery - Lead-acid chemistry is used in automobiles, the electrodes are made of lead and lead-oxide with a strong acidic electrolyte (rechargeable). Nickel-cadmium battery - The electrodes are nickel-hydroxide and cadmium, with potassium-hydroxide ...
A Guide to Rate of Reactions
... Equilibrium. This is because the underlying theory of each of these is very different. Rate of reaction is also called Chemical Kinetics and deals with how fast a reaction happens. Chemical equilibrium is based on thermodynamics and answers the question: ‘How far does the reaction go?’ Learners are ...
... Equilibrium. This is because the underlying theory of each of these is very different. Rate of reaction is also called Chemical Kinetics and deals with how fast a reaction happens. Chemical equilibrium is based on thermodynamics and answers the question: ‘How far does the reaction go?’ Learners are ...
AP Chemistry Review Preparing for the AP
... Focus on your weakest areas; it is doubtful you can do/know everything. The AP Chemistry Exam is designed so that it is impossible to know absolutely everything on it (in case you haven’t noticed). I might as well place the biggest two at the start – You need to review your incorrect MC from the Pra ...
... Focus on your weakest areas; it is doubtful you can do/know everything. The AP Chemistry Exam is designed so that it is impossible to know absolutely everything on it (in case you haven’t noticed). I might as well place the biggest two at the start – You need to review your incorrect MC from the Pra ...
File
... A) Al2O3 B) BeO C) Na2O D) K2O2 2. What is the chemical formula of iron (III) sulfate? A) FeSO4 B) FeSO3 C) Fe(SO4)3 D) Fe2(SO4)3 3 - 4. An experiment is done to determine the density of copper. A sample of copper is weighed, and has a mass of 36.10 grams. The sample is added to a graduated cylinder ...
... A) Al2O3 B) BeO C) Na2O D) K2O2 2. What is the chemical formula of iron (III) sulfate? A) FeSO4 B) FeSO3 C) Fe(SO4)3 D) Fe2(SO4)3 3 - 4. An experiment is done to determine the density of copper. A sample of copper is weighed, and has a mass of 36.10 grams. The sample is added to a graduated cylinder ...
SPRING 2002 Test 2 1. Which of the following statements is
... B. N2 + 3H2 <=> 2 NH3 C. 2 CO + O2 <=> 2 CO2 D. N2O4 <=> 2 NO2 E. N2 + O2 <=> 2 NO Ans. E 12. Consider the exothermic reaction between N2 and H2 to produce NH3. In order to produce as much NH3 as possible, this reaction should be run at A. low temperature and low pressure B. low temperature and high ...
... B. N2 + 3H2 <=> 2 NH3 C. 2 CO + O2 <=> 2 CO2 D. N2O4 <=> 2 NO2 E. N2 + O2 <=> 2 NO Ans. E 12. Consider the exothermic reaction between N2 and H2 to produce NH3. In order to produce as much NH3 as possible, this reaction should be run at A. low temperature and low pressure B. low temperature and high ...
4_ Chemical reactions
... reaction reactants (starting materials) are converted into products. Consider the reaction in which magnesium oxide reacts with carbon dioxide to form magnesium carbonate. We can represent the above “word description” by a “chemical equation”. Chemical equation: MgO + CO2 → MgCO3 Reactants Product W ...
... reaction reactants (starting materials) are converted into products. Consider the reaction in which magnesium oxide reacts with carbon dioxide to form magnesium carbonate. We can represent the above “word description” by a “chemical equation”. Chemical equation: MgO + CO2 → MgCO3 Reactants Product W ...
College Chemistry 1 Note Guide(free download)
... All college chemistry courses are not organized in the same manner. The American Chemical Society provided recommended content information to members regarding these undergraduate courses, but did not specify the order in which topics were to be addressed. Although Professor Etheridge has included t ...
... All college chemistry courses are not organized in the same manner. The American Chemical Society provided recommended content information to members regarding these undergraduate courses, but did not specify the order in which topics were to be addressed. Although Professor Etheridge has included t ...
3.0 Hess`s Law
... • The overall reaction involves the formation rather than the combustion of methane, so the combustion equation for methane is reversed, and its enthalpy changed from negative to positive: CO2(g) + 2H2O(l) → CH4(g) + 2O2(g) ∆H0 = +890.8 kJ ...
... • The overall reaction involves the formation rather than the combustion of methane, so the combustion equation for methane is reversed, and its enthalpy changed from negative to positive: CO2(g) + 2H2O(l) → CH4(g) + 2O2(g) ∆H0 = +890.8 kJ ...
Document
... reasonable effort has been made by the publisher (UCLES) to trace copyright holders, but if any items requiring clearance have unwittingly been included, the publisher will be pleased to make amends at the earliest possible opportunity. University of Cambridge International Examinations is part of t ...
... reasonable effort has been made by the publisher (UCLES) to trace copyright holders, but if any items requiring clearance have unwittingly been included, the publisher will be pleased to make amends at the earliest possible opportunity. University of Cambridge International Examinations is part of t ...
Grade 10 NSC Chemistry Curriculum
... • Define a chemical change as a change in which the chemical nature of the substances involved changes (new chemical substances are formed) • Describe examples of a chemical change that could include - the decomposition of hydrogen peroxide to form water and oxygen; and - the synthesis reaction that ...
... • Define a chemical change as a change in which the chemical nature of the substances involved changes (new chemical substances are formed) • Describe examples of a chemical change that could include - the decomposition of hydrogen peroxide to form water and oxygen; and - the synthesis reaction that ...
Theoretical problems (official version)
... The quantum requirement of the light redox reactions is defined as the average number of light photons (not necessarily integer) needed for the transfer of one electron from a reducing agent to an oxidant. The isolated chloroplasts were irradiated during 2 hours by a monochromatic light (wavelength ...
... The quantum requirement of the light redox reactions is defined as the average number of light photons (not necessarily integer) needed for the transfer of one electron from a reducing agent to an oxidant. The isolated chloroplasts were irradiated during 2 hours by a monochromatic light (wavelength ...
13. transition metal chemistry
... full d-shell.’ Zinc forms only one stable ion, Zn2+ which has an electronic configuration of [Ar] 4so 3d10 i.e. has a full d shell. (1 mark for definition, 1 mark for full explanation of why zinc does not meet the criteria) NOTE IUPAC gives the definition of a transition element as ‘An element whose ...
... full d-shell.’ Zinc forms only one stable ion, Zn2+ which has an electronic configuration of [Ar] 4so 3d10 i.e. has a full d shell. (1 mark for definition, 1 mark for full explanation of why zinc does not meet the criteria) NOTE IUPAC gives the definition of a transition element as ‘An element whose ...
Free Energy I
... Ice melting at 0°C and 1 bar. Ice and water are in equilibrium under these conditions. Adding heat slowly to ice at 0°C will cause it to melt. The process can be undone and the ice reformed by slowly removing the same amount of heat. + heat ...
... Ice melting at 0°C and 1 bar. Ice and water are in equilibrium under these conditions. Adding heat slowly to ice at 0°C will cause it to melt. The process can be undone and the ice reformed by slowly removing the same amount of heat. + heat ...
9. Balancing Equations
... First ask, “how many sodiums on the left, how many sodiums on the right?” Use coefficients to balance each of the atoms left to right. ...
... First ask, “how many sodiums on the left, how many sodiums on the right?” Use coefficients to balance each of the atoms left to right. ...
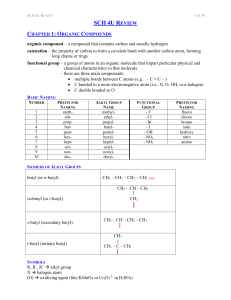
sch4ureview
... monomers; two types: addition and condensation monomer – a molecule or compound usually containing carbon and of relatively low molecular weight and simple structure which is capable of conversion to polymers by combination with itself or other similar molecules or compounds dimer – a molecule made ...
... monomers; two types: addition and condensation monomer – a molecule or compound usually containing carbon and of relatively low molecular weight and simple structure which is capable of conversion to polymers by combination with itself or other similar molecules or compounds dimer – a molecule made ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.