College Chemistry
... Dimensional calculations are greatly simplified if a consistent set of units is employed. The three major reference dimensions for mechanics are length, mass, and time, but length can be measured in units of inches, feet, centimeters, meters, etc. Which should be used? The scientific community has m ...
... Dimensional calculations are greatly simplified if a consistent set of units is employed. The three major reference dimensions for mechanics are length, mass, and time, but length can be measured in units of inches, feet, centimeters, meters, etc. Which should be used? The scientific community has m ...
X: Ag, Ca, In, Li, Na, Sn, Sr and Zn
... scanning calorimetry (DSC). These were Mg-In-Sn (5 at. %, 10 at. % Sn), Mg-Sn-Ag (10 at. % Sn, 30 at.% Ag0 and Mg-Sn-Zn (10 at.% Sn). No ternary compounds were found in these three isothermal sections. Iisothermal sections of the Mg-Sn-Ca and Mg-Sn-Sr systems in the Mg-rich region (350 and 415 oC) w ...
... scanning calorimetry (DSC). These were Mg-In-Sn (5 at. %, 10 at. % Sn), Mg-Sn-Ag (10 at. % Sn, 30 at.% Ag0 and Mg-Sn-Zn (10 at.% Sn). No ternary compounds were found in these three isothermal sections. Iisothermal sections of the Mg-Sn-Ca and Mg-Sn-Sr systems in the Mg-rich region (350 and 415 oC) w ...
The evolution of granitic and rhyolitic melts documented
... Quartz provides a detailed chronicle of physicochemical changes in granitic melts. Quartz phenocrysts normally grow in different magma batches, which are repeatedly recharged. The solidified granites and rhyolites therefore comprise mixtures of different phenocryst populations embedded in a microcry ...
... Quartz provides a detailed chronicle of physicochemical changes in granitic melts. Quartz phenocrysts normally grow in different magma batches, which are repeatedly recharged. The solidified granites and rhyolites therefore comprise mixtures of different phenocryst populations embedded in a microcry ...
Chapter 15 Chemical Equilibrium
... 2. For those species for which both the initial and equilibrium concentrations are known, calculate the change in concentration that occurs as the system reaches equilibrium. 3. Use the stoichiometry of the reaction (that is, use the coefficients in the balanced chemical equation) to calculate the c ...
... 2. For those species for which both the initial and equilibrium concentrations are known, calculate the change in concentration that occurs as the system reaches equilibrium. 3. Use the stoichiometry of the reaction (that is, use the coefficients in the balanced chemical equation) to calculate the c ...
Teacher Edition Calculations
... The molecular mass, expressed in grams, is the sum of the molar masses of the atoms making up that molecule. Complete the following table : (Note all molar masses recorded to 4 sig figures) ...
... The molecular mass, expressed in grams, is the sum of the molar masses of the atoms making up that molecule. Complete the following table : (Note all molar masses recorded to 4 sig figures) ...
One-pot aqueous synthesis of cysteine-capped
... generally classified into top-down and bottom-up routes. A conventional bottom-up method is vapor-liquid-solid (VLS) growth (Simon et al. 2010; Wu et al. 2012; Wang et al. 2008; Yang et al. 2013), in which nanowire diameters are controlled by the size of the Au or Bi nanocatalysts. The obtained nano ...
... generally classified into top-down and bottom-up routes. A conventional bottom-up method is vapor-liquid-solid (VLS) growth (Simon et al. 2010; Wu et al. 2012; Wang et al. 2008; Yang et al. 2013), in which nanowire diameters are controlled by the size of the Au or Bi nanocatalysts. The obtained nano ...
Stoichiometry of Formulas and Equations
... hemistry is a practical science. Just imagine how useful it could be to determine the formula of a compound from the masses of its elements or to predict the amounts of substances consumed and produced in a reaction. Suppose you are a polymer chemist preparing a new plastic: how much of this new mat ...
... hemistry is a practical science. Just imagine how useful it could be to determine the formula of a compound from the masses of its elements or to predict the amounts of substances consumed and produced in a reaction. Suppose you are a polymer chemist preparing a new plastic: how much of this new mat ...
© www.CHEMSHEETS.co.uk 17-Jul
... For most bonds (e.g. C-H, C-C, C=O, O-H, etc.) the value for the bond enthalpy is an average taken from a range of molecules as the exact value varies from compound to compound. For some bond enthalpies (e.g. H-H, H-Cl, O=O, etc) the value is exact as only one molecule contains that bond. ...
... For most bonds (e.g. C-H, C-C, C=O, O-H, etc.) the value for the bond enthalpy is an average taken from a range of molecules as the exact value varies from compound to compound. For some bond enthalpies (e.g. H-H, H-Cl, O=O, etc) the value is exact as only one molecule contains that bond. ...
ELECTROCHEMICAL STUDY OF CORROSION PROCESSES IN
... of oxygen. On the other hand, CO processes are dominated by direct molecular processes such as the direct reaction of the metal with aggressive species on one site. EES method is used to differentiate two corrosion mechanisms by postulating that only the electrochemical mechanism gives rise to spont ...
... of oxygen. On the other hand, CO processes are dominated by direct molecular processes such as the direct reaction of the metal with aggressive species on one site. EES method is used to differentiate two corrosion mechanisms by postulating that only the electrochemical mechanism gives rise to spont ...
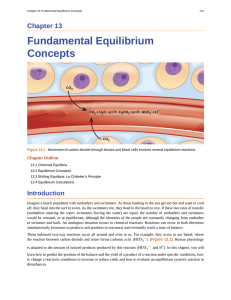
Fundamental Equilibrium Concepts
... Imagine a beach populated with sunbathers and swimmers. As those basking in the sun get too hot and want to cool off, they head into the surf to swim. As the swimmers tire, they head to the beach to rest. If these two rates of transfer (sunbathers entering the water, swimmers leaving the water) are ...
... Imagine a beach populated with sunbathers and swimmers. As those basking in the sun get too hot and want to cool off, they head into the surf to swim. As the swimmers tire, they head to the beach to rest. If these two rates of transfer (sunbathers entering the water, swimmers leaving the water) are ...
10. Solution Guide to Supplementary Exercises
... does not go to completion no matter how long the reaction mixture is heated under reflux. This ...
... does not go to completion no matter how long the reaction mixture is heated under reflux. This ...
- University of Bath Opus
... crystalline mineral with periodic “twinning” planes (across which the dielectric tensor undergoes a mirror flip). From this observation he realised there is a narrow band of wavelengths for which light propagation was prohibited through the planes [Joannopoulos 1995, Biswas 1995]. However, it was no ...
... crystalline mineral with periodic “twinning” planes (across which the dielectric tensor undergoes a mirror flip). From this observation he realised there is a narrow band of wavelengths for which light propagation was prohibited through the planes [Joannopoulos 1995, Biswas 1995]. However, it was no ...
Moles 1 - pedagogics.ca
... and stoichiometry is the study of the ratios in which chemical substances combine. In order to know the exact quantity of each substance that is required to react we need to know the number of atoms, molecules or ions present in a specific amount of that substance. However, the mass of an individual ...
... and stoichiometry is the study of the ratios in which chemical substances combine. In order to know the exact quantity of each substance that is required to react we need to know the number of atoms, molecules or ions present in a specific amount of that substance. However, the mass of an individual ...
Chapter 15: Chemical Equilibrium
... amount of solid present as long as there is some solid present for the system to reach equilibrium. The concentration of a pure solid depends only on the density of the substance, a constant that can be incorporated into the equilibrium constant. 2. Pure liquids and solvents do not appear in equilib ...
... amount of solid present as long as there is some solid present for the system to reach equilibrium. The concentration of a pure solid depends only on the density of the substance, a constant that can be incorporated into the equilibrium constant. 2. Pure liquids and solvents do not appear in equilib ...
b - Gordon State College
... a) If required > available, then B is the limiting reagent and A is the excess reagent. b) If required < available, then B is the excess reagent and A is the limiting reagent. 5) Use the amount of the limiting reagent and the stoichiometry to calculate the amount of any product and the amount of the ...
... a) If required > available, then B is the limiting reagent and A is the excess reagent. b) If required < available, then B is the excess reagent and A is the limiting reagent. 5) Use the amount of the limiting reagent and the stoichiometry to calculate the amount of any product and the amount of the ...
Kinetic investigation of low-pH Fe(II) oxidation and development of a
... costs and operational costs in waste treatment plants where it is generated (Mesa et al., 2004). It is also used in industrial-scale animal feeding operations (AFOs) to treat animal wastes and attaining the same benefits. Biogas production and utilization is constantly increasing, as it represents a ...
... costs and operational costs in waste treatment plants where it is generated (Mesa et al., 2004). It is also used in industrial-scale animal feeding operations (AFOs) to treat animal wastes and attaining the same benefits. Biogas production and utilization is constantly increasing, as it represents a ...
Document
... is 0.113 at 298 K, which corresponds to a standard free-energy change of 5.4 kJ/mol. In a certain experiment, the initial pressures are PN2O4 = 0.453 atm and PNO2 = 0.122 atm. Calculate ΔG for the reaction at these pressures, and predict the direction in which the reaction will proceed spontaneously ...
... is 0.113 at 298 K, which corresponds to a standard free-energy change of 5.4 kJ/mol. In a certain experiment, the initial pressures are PN2O4 = 0.453 atm and PNO2 = 0.122 atm. Calculate ΔG for the reaction at these pressures, and predict the direction in which the reaction will proceed spontaneously ...
Chemistry - A Quantitative Science
... done the same as converting dozens to items. 1.5 doz = (1.5 doz)(12 items.doz-1) = 18 items and 1.5 mol = (1.5 mol)( 6.0x1023 atoms.mol-1) = 9.0x1023 atoms. The mole is used simply because it is much easier to discuss the number of atoms in moles than it is as individual items - 0.10 mol H2O is a mu ...
... done the same as converting dozens to items. 1.5 doz = (1.5 doz)(12 items.doz-1) = 18 items and 1.5 mol = (1.5 mol)( 6.0x1023 atoms.mol-1) = 9.0x1023 atoms. The mole is used simply because it is much easier to discuss the number of atoms in moles than it is as individual items - 0.10 mol H2O is a mu ...
Copyright 2010 Scott R
... Na(H3BNMe2BH3), in tetrahydrofuran produces the new complex Th(H3BNMe2BH3)4. The thorium center forms bonds with fifteen hydrogen atoms; accordingly, this is the first example of a fifteen-coordinate atom of any kind. As determined by both single crystal X-ray and single crystal neutron diffraction ...
... Na(H3BNMe2BH3), in tetrahydrofuran produces the new complex Th(H3BNMe2BH3)4. The thorium center forms bonds with fifteen hydrogen atoms; accordingly, this is the first example of a fifteen-coordinate atom of any kind. As determined by both single crystal X-ray and single crystal neutron diffraction ...
synthesis and properties of v3+ analogues of jarosite-group
... The reaction vessel was sealed with a ground glass lid, and the contents of the vessel were stirred using a magnetic stirring bar. Reagent-grade VCl3 was dissolved in distilled water to give a 0.4 M V3+ solution. The as-prepared solution was filtered to remove the small amount of insoluble matter th ...
... The reaction vessel was sealed with a ground glass lid, and the contents of the vessel were stirred using a magnetic stirring bar. Reagent-grade VCl3 was dissolved in distilled water to give a 0.4 M V3+ solution. The as-prepared solution was filtered to remove the small amount of insoluble matter th ...
Cliffs Notes
... Acquisitions Editor: Sherry Gomoll Technical Editor: Christopher Bushee Production Proofreader: Joel K. Draper Hungry Minds Indianapolis Production Services ...
... Acquisitions Editor: Sherry Gomoll Technical Editor: Christopher Bushee Production Proofreader: Joel K. Draper Hungry Minds Indianapolis Production Services ...
The science of chemistry is concerned
... amount relative to some critical minimum will control growth and reproduction of any species of plant or animal life. When a group of organisms runs out of that essential limiting reagent, the chemical reactions needed for growth and reproduction must stop. Vitamins, protein, and other nutrients are ...
... amount relative to some critical minimum will control growth and reproduction of any species of plant or animal life. When a group of organisms runs out of that essential limiting reagent, the chemical reactions needed for growth and reproduction must stop. Vitamins, protein, and other nutrients are ...
CALCITE – AMPHIBOLE – CLINOPYROXENE ROCK
... parageneses. Sr-enrichment is generally characteristic of primary calcite in carbonatites worldwide; secondary or late-stage calcite invariably shows some depletion in Sr that is in some cases accompanied by enrichment in Mn (e.g., Pouliot 1970, Sokolov 1985, Clarke et al. 1992, Dawson et al. 1996, ...
... parageneses. Sr-enrichment is generally characteristic of primary calcite in carbonatites worldwide; secondary or late-stage calcite invariably shows some depletion in Sr that is in some cases accompanied by enrichment in Mn (e.g., Pouliot 1970, Sokolov 1985, Clarke et al. 1992, Dawson et al. 1996, ...
Chapter 15
... of the reaction products is physically separated from a reaction mixture as a gas (the reaction of a metal carbonate with acid, for example, Figure 15.X), a single reaction arrow is used. The Equilibrium State In Chapter 12, we described the nature of a dynamic equilibrium between a liquid and its v ...
... of the reaction products is physically separated from a reaction mixture as a gas (the reaction of a metal carbonate with acid, for example, Figure 15.X), a single reaction arrow is used. The Equilibrium State In Chapter 12, we described the nature of a dynamic equilibrium between a liquid and its v ...
Chapter 15: Chemical Equilibrium
... The equilibrium arrow (Æ) is used to indicate that a reaction is reversible. NO2(g) + CO(g) Æ NO(g) + CO2(g) There are occasions when the use of an equilibrium arrow is not appropriate. For example, when hydrogen and oxygen react to form water vapor (Figure 15.X), product formation is very strongly ...
... The equilibrium arrow (Æ) is used to indicate that a reaction is reversible. NO2(g) + CO(g) Æ NO(g) + CO2(g) There are occasions when the use of an equilibrium arrow is not appropriate. For example, when hydrogen and oxygen react to form water vapor (Figure 15.X), product formation is very strongly ...
Crystallization

Crystallization is the (natural or artificial) process of formation of solid crystals precipitating from a solution, melt or more rarely deposited directly from a gas. Crystallization is also a chemical solid–liquid separation technique, in which mass transfer of a solute from the liquid solution to a pure solid crystalline phase occurs. In chemical engineering crystallization occurs in a crystallizer. Crystallization is therefore an aspect of precipitation, obtained through a variation of the solubility conditions of the solute in the solvent, as compared to precipitation due to chemical reaction.