Elementary Principles of Chemical Processes, 3rd Update Edition
... Education and since 1988 has written the “Random Thoughts” column for that journal. His honors include the R.J. Reynolds Award for Excellence in Teaching, Research, and Extension, the AT&T Foundation Award for Excellence in Engineering Education, the Chemical Manufacturers Association National Catal ...
... Education and since 1988 has written the “Random Thoughts” column for that journal. His honors include the R.J. Reynolds Award for Excellence in Teaching, Research, and Extension, the AT&T Foundation Award for Excellence in Engineering Education, the Chemical Manufacturers Association National Catal ...
Introductory Chemistry
... discussed in the textbook. The instructor, of course, may wish to discuss alternative methods of solution with his or her students. One topic that causes many students concern is the matter of significant figures and the determination of the number of digits to which a solution to a problem should b ...
... discussed in the textbook. The instructor, of course, may wish to discuss alternative methods of solution with his or her students. One topic that causes many students concern is the matter of significant figures and the determination of the number of digits to which a solution to a problem should b ...
9 SHS CH 9 LECTURE shs_ch_9_lecture
... How many moles of NaOH will react with 3.21 moles of sulfuric acid? Step 1. Balanced the chemical equation. Step 2. Write down the given numeric info. If it is not in mole units convert it to moles using the molecular mass as a conversion factor. Step 3. Now convert from the moles of starting substa ...
... How many moles of NaOH will react with 3.21 moles of sulfuric acid? Step 1. Balanced the chemical equation. Step 2. Write down the given numeric info. If it is not in mole units convert it to moles using the molecular mass as a conversion factor. Step 3. Now convert from the moles of starting substa ...
Chapter 1: Matter and Measurements
... nonmetal can combine with other non-metals to form molecular compounds such as those mentioned in Section 2.4 (indeed, a whole field of chemistry, called organic chemistry, is dedicated to the study of such compounds). Carbon can, however, also form ionic compounds such as calcium carbide (used in o ...
... nonmetal can combine with other non-metals to form molecular compounds such as those mentioned in Section 2.4 (indeed, a whole field of chemistry, called organic chemistry, is dedicated to the study of such compounds). Carbon can, however, also form ionic compounds such as calcium carbide (used in o ...
Atmospheric evolution in the Precambrian: Constraints from water
... The significance of the temperature effects depends on the apparent activation energy of mineral dissolution, which has been reported to be affected by solution composition (e.g., Cama et al., 1999). Because the relationship between the apparent activation energy and solution composition is not yet ...
... The significance of the temperature effects depends on the apparent activation energy of mineral dissolution, which has been reported to be affected by solution composition (e.g., Cama et al., 1999). Because the relationship between the apparent activation energy and solution composition is not yet ...
Preparation and reactions of some lower tungsten halides and
... A gradient furnace was used to heat the metal to ...
... A gradient furnace was used to heat the metal to ...
Chem 12 SM Ch5 Review final new ok revised
... releasing about 2.5 times the quantity of energy per gram than methane but the widespread use of hydrogen as a fuel has the limitations of high production cost and difficulty in storage and transport. (b) Answers may vary. Sample answer: Possible discoveries that could make hydrogen a more viable al ...
... releasing about 2.5 times the quantity of energy per gram than methane but the widespread use of hydrogen as a fuel has the limitations of high production cost and difficulty in storage and transport. (b) Answers may vary. Sample answer: Possible discoveries that could make hydrogen a more viable al ...
Answers to SelectedTextbook Questions
... (d) van der Waals equation is a relation between the pressure, temperature and volume of a gas that accounts for the non‐zero size of the gas molecules and the attractive forces between them. (e) Gibbs free energy, G = H − TS, combines enthalpy and entropy to give a quantity which must decreas ...
... (d) van der Waals equation is a relation between the pressure, temperature and volume of a gas that accounts for the non‐zero size of the gas molecules and the attractive forces between them. (e) Gibbs free energy, G = H − TS, combines enthalpy and entropy to give a quantity which must decreas ...
CHAPTER 6 THERMOCHEMISTRY
... stronger (on average) than in the reactants. This occurs generally in exothermic processes. Products have a higher potential energy than reactants when the reactants have the stronger bonds (on average). This is typified by endothermic reactions. ...
... stronger (on average) than in the reactants. This occurs generally in exothermic processes. Products have a higher potential energy than reactants when the reactants have the stronger bonds (on average). This is typified by endothermic reactions. ...
Sample Assessment Materials - Edexcel
... Unit 1: The Core Principles of Chemistry ...........................................................7 Unit 2: Application of Core Principles of Chemistry ............................................ 35 Unit 4: General Principles of Chemistry I ........................................................ ...
... Unit 1: The Core Principles of Chemistry ...........................................................7 Unit 2: Application of Core Principles of Chemistry ............................................ 35 Unit 4: General Principles of Chemistry I ........................................................ ...
Soln Chem 2008Nov(9746)
... In the titration, the reaction is Ba(OH)2.8H2O + 2HCl → BaCl2 + 10H2O The titres were lower than expected suggests that the solution contains less Ba(OH)2 than expected. This is because when the prepared solution was left in an open beaker, some Ba(OH)2 has reacted with CO2(g) in the air to form sol ...
... In the titration, the reaction is Ba(OH)2.8H2O + 2HCl → BaCl2 + 10H2O The titres were lower than expected suggests that the solution contains less Ba(OH)2 than expected. This is because when the prepared solution was left in an open beaker, some Ba(OH)2 has reacted with CO2(g) in the air to form sol ...
1 Ag PO 7.5 10 1.79 10 418.57 mol x gL x M g
... (b) Ksp values assume 100% dissociation into ions. This condition is a reasonable assumption for singly charged species but the assumption does not work well when working with charges greater than +1 or −1. Ion paring becomes a significant factor when ions charges are large and when concentrations a ...
... (b) Ksp values assume 100% dissociation into ions. This condition is a reasonable assumption for singly charged species but the assumption does not work well when working with charges greater than +1 or −1. Ion paring becomes a significant factor when ions charges are large and when concentrations a ...
COMPETITION PTOBLEMS 1
... This publication contains the competition problems from the first twenty International Chemistry Olympiads (ICHO) organized in the years 1968 – 1988. It has been published by the ICHO International Information Centre in Bratislava (Slovakia) on the occasion of the 40th anniversary of this internatio ...
... This publication contains the competition problems from the first twenty International Chemistry Olympiads (ICHO) organized in the years 1968 – 1988. It has been published by the ICHO International Information Centre in Bratislava (Slovakia) on the occasion of the 40th anniversary of this internatio ...
Workshop materials for Class XII
... What new technology can help you? Learn more about computers—use them not only for power points but also for video recording. Do you get good contacts because of your nature that helps you benefit the school What trend do you see in your school how you can take advantage of these— eg: A circular ...
... What new technology can help you? Learn more about computers—use them not only for power points but also for video recording. Do you get good contacts because of your nature that helps you benefit the school What trend do you see in your school how you can take advantage of these— eg: A circular ...
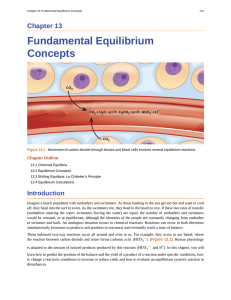
Fundamental Equilibrium Concepts
... equilibrium constants. It should be pointed out that using concentrations in these computations is a convenient but simplified approach that sometimes leads to results that seemingly conflict with the law of mass action. For example, equilibria involving aqueous ions often exhibit equilibrium consta ...
... equilibrium constants. It should be pointed out that using concentrations in these computations is a convenient but simplified approach that sometimes leads to results that seemingly conflict with the law of mass action. For example, equilibria involving aqueous ions often exhibit equilibrium consta ...
Stoichiometry
... One mole of $1 bills stacked one on top of another would reach from the Sun to Pluto and back 7.5 million times. It would take light 9500 years to travel from the bottom to the top of a stack of 1 mole of $1 bills. ...
... One mole of $1 bills stacked one on top of another would reach from the Sun to Pluto and back 7.5 million times. It would take light 9500 years to travel from the bottom to the top of a stack of 1 mole of $1 bills. ...
Chemistry.of Organic Compounds
... in this text considerable emphasis is placed on the explanation of physical properties and on the mechanism of organic reactions. Where such material is given, an effort has been made to keep the discussion as simple as possible even at the risk of being quantitatively inaccurate, for "except ye utt ...
... in this text considerable emphasis is placed on the explanation of physical properties and on the mechanism of organic reactions. Where such material is given, an effort has been made to keep the discussion as simple as possible even at the risk of being quantitatively inaccurate, for "except ye utt ...
Chapter 4 - Chemistry
... Strategy: Recall that strong acids and strong bases are strong electrolytes. They are completely ionized in solution. An ionic equation will show strong acids and strong bases in terms of their free ions. Weak acids and weak bases are weak electrolytes. They only ionize to a small extent in solution ...
... Strategy: Recall that strong acids and strong bases are strong electrolytes. They are completely ionized in solution. An ionic equation will show strong acids and strong bases in terms of their free ions. Weak acids and weak bases are weak electrolytes. They only ionize to a small extent in solution ...
Ozone decomposition
... When the ozone concentration exceeds 15–20% it has a blue color. At atmospheric pressure and temperature of 161.3 K, the ozone becomes fluid and is of deep blue color. It cures at 80.6 K by acquiring a dark purple color (Lunin et al., 1998). Ozone is explosive in all three physical conditions. Work ...
... When the ozone concentration exceeds 15–20% it has a blue color. At atmospheric pressure and temperature of 161.3 K, the ozone becomes fluid and is of deep blue color. It cures at 80.6 K by acquiring a dark purple color (Lunin et al., 1998). Ozone is explosive in all three physical conditions. Work ...
College Chemistry
... Dimensional calculations are greatly simplified if a consistent set of units is employed. The three major reference dimensions for mechanics are length, mass, and time, but length can be measured in units of inches, feet, centimeters, meters, etc. Which should be used? The scientific community has m ...
... Dimensional calculations are greatly simplified if a consistent set of units is employed. The three major reference dimensions for mechanics are length, mass, and time, but length can be measured in units of inches, feet, centimeters, meters, etc. Which should be used? The scientific community has m ...
Stoichiometric Calculations
... about the relative volumes of gas reactants and products. For the next problem, we will not write out all of the ratios ahead of time. Sample Problem #4 How many liters of SO2 will be produced when 6 liters of O2 are reacted as shown below? CS2 (g) + 3 O2 (g)® CO2 (g) + 2 SO2 (g) ...
... about the relative volumes of gas reactants and products. For the next problem, we will not write out all of the ratios ahead of time. Sample Problem #4 How many liters of SO2 will be produced when 6 liters of O2 are reacted as shown below? CS2 (g) + 3 O2 (g)® CO2 (g) + 2 SO2 (g) ...
X Science Practice Paper - Brilliant Public School Sitamarhi
... Q 35 (a) Name two edible acids. (b) Classify aqueous solution of carbon dioxide as acid or base. (c)Which acid will produce large number of H + ions and why? (i) aq. solution of acetic acid (ii) aq. solution of hydrochloric acid Marks (3) Q 36 Represent the following by a balanced chemical reaction: ...
... Q 35 (a) Name two edible acids. (b) Classify aqueous solution of carbon dioxide as acid or base. (c)Which acid will produce large number of H + ions and why? (i) aq. solution of acetic acid (ii) aq. solution of hydrochloric acid Marks (3) Q 36 Represent the following by a balanced chemical reaction: ...
Electrolysis of water
Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current being passed through the water.This technique can be used to make hydrogen fuel (hydrogen gas) and breathable oxygen; though currently most industrial methods make hydrogen fuel from natural gas instead.